Equilibrium Reversible Reactions Reversible reactions can proceed in

Equilibrium

Reversible Reactions • Reversible reactions can proceed in both directions: • Reactants → Products • Products → Reactants Ca. CO 3 → Ca. O + CO 2 → Ca. CO 3 • A double arrow is used to show that a reaction is reversible • Ca. CO 3 Ca. O + CO 2
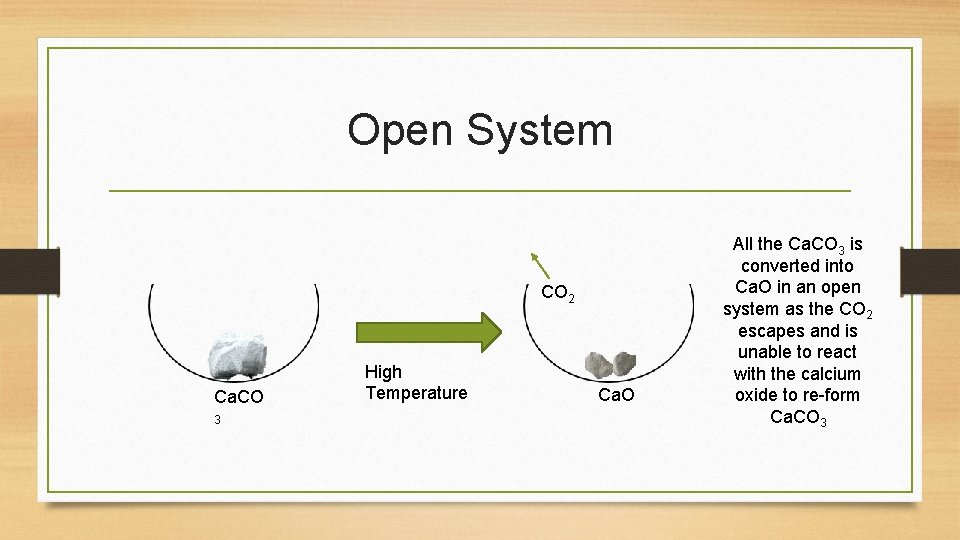
Open System CO 2 Ca. CO 3 High Temperature Ca. O All the Ca. CO 3 is converted into Ca. O in an open system as the CO 2 escapes and is unable to react with the calcium oxide to re-form Ca. CO 3
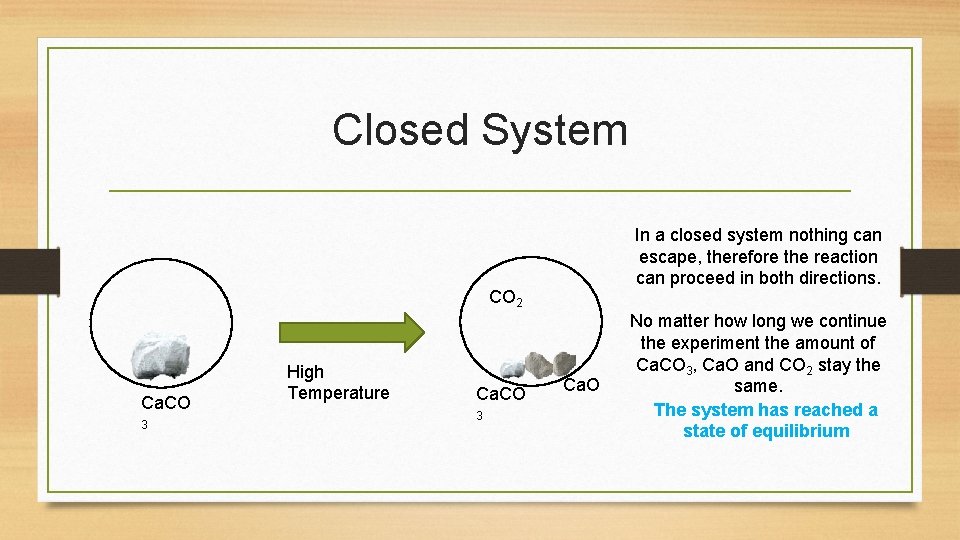
Closed System In a closed system nothing can escape, therefore the reaction can proceed in both directions. CO 2 Ca. CO 3 High Temperature Ca. CO 3 Ca. O No matter how long we continue the experiment the amount of Ca. CO 3, Ca. O and CO 2 stay the same. The system has reached a state of equilibrium

Dynamic Equilibrium • When a reaction reaches dynamic equilibrium the concentrations of reactants and products are constant • This is because the rate of the forward reaction is equal to the rate of the reverse reaction • Equilibrium does not necessarily mean that reactants and products are present in equal concentrations

Physical Equilibrium • Chemical equilibrium involves different substances as reactants and products. • Equilibrium between two phases of the same substance is called physical equilibrium because the changes that occur are physical processes H 2 O(l) H 2 O(g)
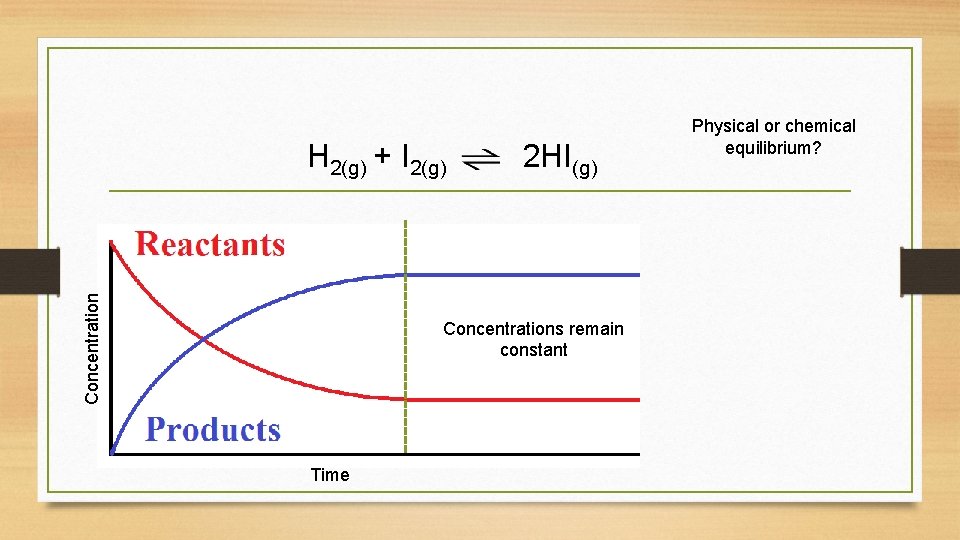
Concentration H 2(g) + I 2(g) 2 HI(g) Concentrations remain constant Time Physical or chemical equilibrium?
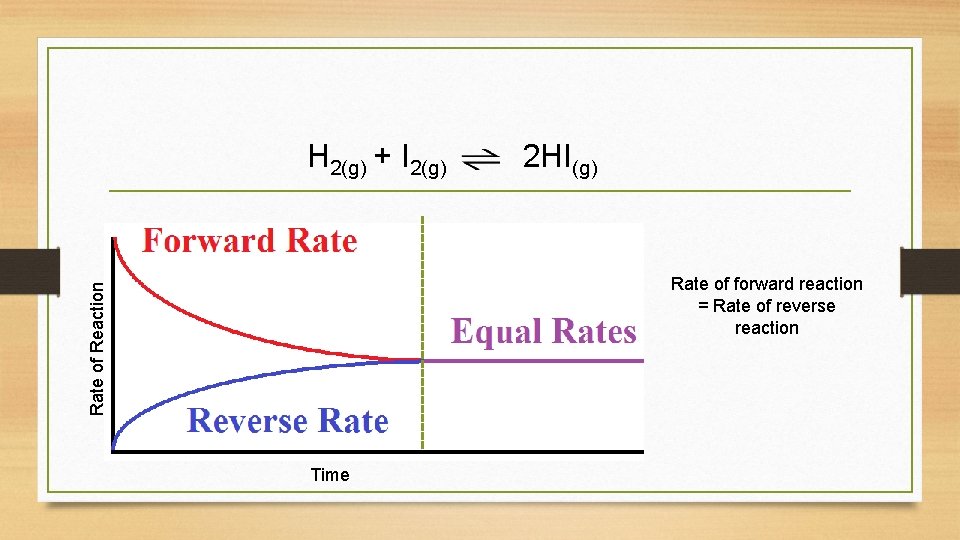
H 2(g) + I 2(g) 2 HI(g) Rate of Reaction Rate of forward reaction = Rate of reverse reaction Time

Ca. CO 3 Ca. O + CO 2 Equilibrium Characteristics • Macroscopic properties are constant: concentrations of reactants and products. • Rate of forward reaction is equal to rate of reverse reaction • Equilibrium can only be attained in a closed system • All species in the chemical equation are present in the equilibrium mixture • Equilibrium can be attained from either direction. Concentrations at equilibrium depend on initial concentrations of reactants

Position of Equilibrium • The position of equilibrium refers to the concentrations of reactants and products present at equilibrium. [Reactant s] [Product s] In this reaction the position of the equilibrium lies to the right Alternatively, the position can be described as favouring reactants or favouring products. [Reactant s] [Products ] In this reaction the position of the equilibrium lies to the left

Le Chatelier’s Principle • If a system at equilibrium is subjected to some change (stress), the position of equilibrium will shift in order to minimize the effect of the change. • Le Chatelier’s principle allows us to predict in which direction the position of equilibrium will shift.

The effect of concentration • An equilibrium exists between the following four substances: A + 2 B C + D • What would happen if you increased [A]? • According to Le Chatelier, the position of equilibrium will move in such a way as to counteract the change. Therefore, the position of equilibrium will shift to the right.

• What would happen if reactants or products were selectively removed from the equilibrium mixture? A + 2 B C + D C is removed as soon as it forms (decrease in [C]) A is removed (decrease in [A]) D is removed as soon as it forms (decrease in [D]) B is removed (decrease in [B]) Equilibrium shifts to the right Equilibrium shifts to the left

The effect of temperature • In order to determine the effect of temperature on an equilibrium we need to know whether the forward/reverse reaction is endothermic or exothermic. Reactants → Products + = A + 2 B C + D∆H = A + 2 B − 250 k. J/mol C + D∆H = +250 k. J/mol exothermic = endothermic Heat + Reactants → Products Suppose the system reaches equilibrium at 700 K and the temperature is increased to 800 K The equilibrium will shift to counteract the increase in temperature, meaning that it will favor the endothermic direction. In the example above the equilibrium would shift to the left

• How will the position of equilibrium change if the temperature is increased? ∆H forward reaction: − 40 k. J/mol NO(g) + NO 2(g) N 2 O 3(g) CH 4(g) + H 2 O(g) CO(g) + 3 H 2(g) +206 k. J/mol CO(g) + 2 H 2(g) CH 3 OH(g) − 90 k. J/mol When temperature is increased the equilibrium will shift in the endothermic direction in order to use the energy

• How will the position of equilibrium change if the temperature is decreased? 2 HI(g) H 2(g) + I 2(g) N 2(g) + 3 H 2(g) H 2 O(l) 2 NH 3(g) H+(aq) + OH-(aq) ∆H forward reaction: +10 k. J/mol − 92 k. J/mol − 95 k. J/mol When temperature is decreased the equilibrium will shift in the exothermic direction in order to produce heat

The effect of pressure/volume • If a reaction involves a change in the number of gas molecules, an increase in pressure results in the position of equilibrium shifting in the direction that gives a decrease in the number of gas molecules. The pressure of (2) is higher as the greater number of particles results in more collisions with the walls of the container

A + 2 B C + D • Assume that all the reactants/products are in the gas state. • What would happen if the pressure of the system is increased? The equilibrium would shift to the right as there are fewer gas molecules on the product side of the equation. 2 SO 3(g) What would happen if the pressure of 2 SO 2(g) + O 2(g) the system was decreased? The equilibrium would shift to the right as the number of gas molecules is greater on the product side of the equation
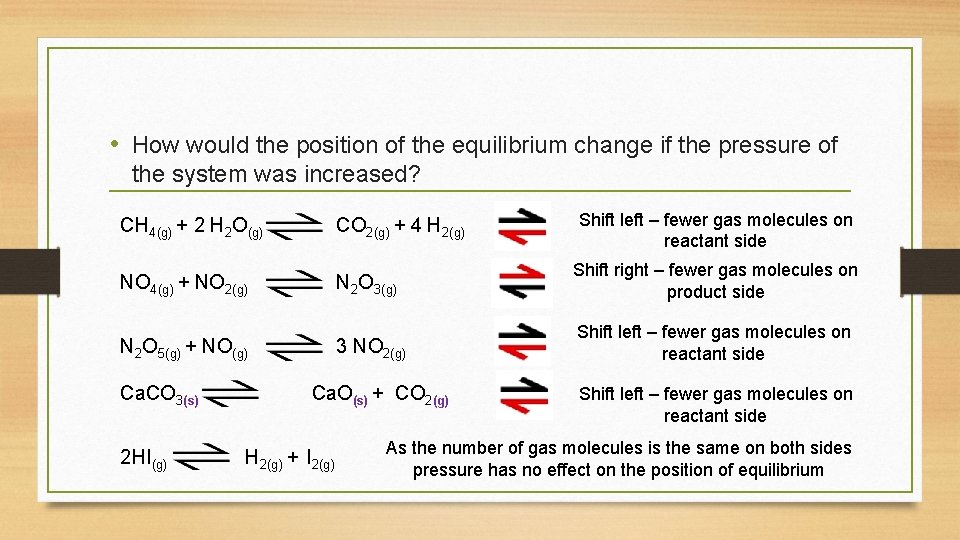
• How would the position of the equilibrium change if the pressure of the system was increased? Shift left – fewer gas molecules on reactant side CH 4(g) + 2 H 2 O(g) CO 2(g) + 4 H 2(g) NO 4(g) + NO 2(g) N 2 O 3(g) Shift right – fewer gas molecules on product side 3 NO 2(g) Shift left – fewer gas molecules on reactant side N 2 O 5(g) + NO(g) Ca. CO 3(s) Ca. O(s) + CO 2(g) 2 HI(g) H 2(g) + I 2(g) Shift left – fewer gas molecules on reactant side As the number of gas molecules is the same on both sides pressure has no effect on the position of equilibrium
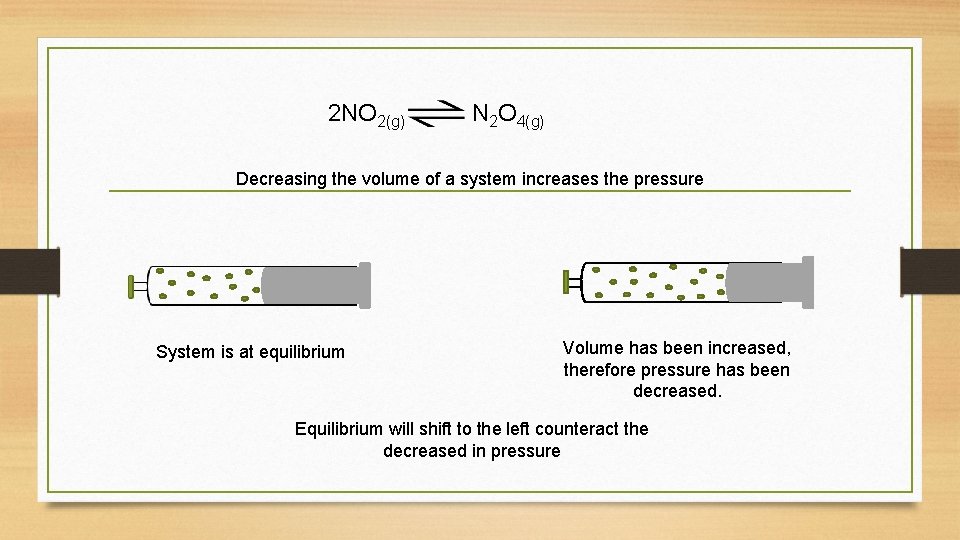
2 NO 2(g) N 2 O 4(g) Decreasing the volume of a system increases the pressure System is at equilibrium Volume has been increased, therefore pressure has been decreased. Equilibrium will shift to the left counteract the decreased in pressure

The effect of a catalyst • Recall that a catalyst increases the rate of reaction by lowering the activation energy • A catalysts lowers the activation energy for the forward reaction and the reverse reaction to the same extent • Therefore, a catalyst does not effect the position of an equilibrium • Adding a catalyst simply causes an equilibrium to be reached sooner
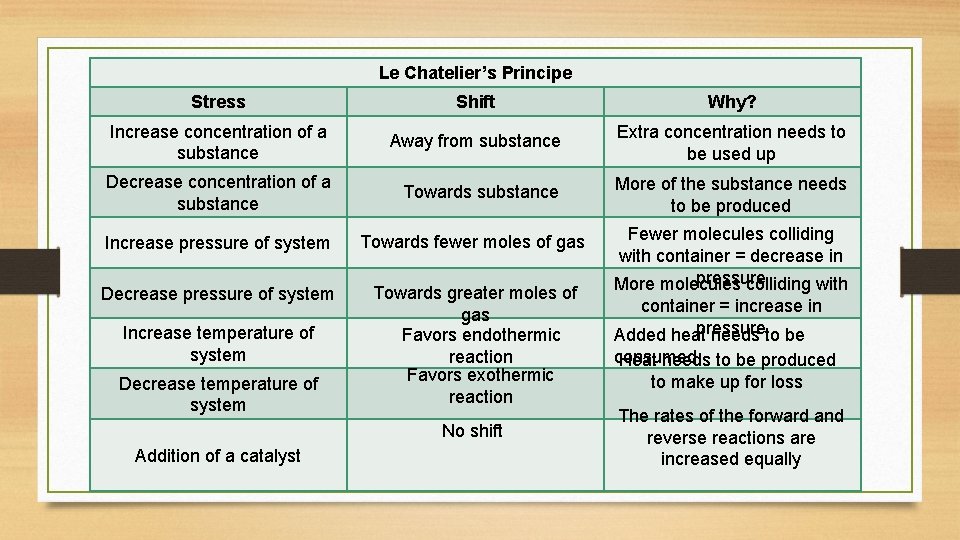
Le Chatelier’s Principe Stress Shift Why? Increase concentration of a substance Away from substance Extra concentration needs to be used up Decrease concentration of a substance Towards substance More of the substance needs to be produced Increase pressure of system Towards fewer moles of gas Decrease pressure of system Towards greater moles of gas Favors endothermic reaction Favors exothermic reaction Increase temperature of system Decrease temperature of system No shift Addition of a catalyst Fewer molecules colliding with container = decrease in pressure More molecules colliding with container = increase in pressure Added heat needs to be consumed Heat needs to be produced to make up for loss The rates of the forward and reverse reactions are increased equally

Solubility & p. H Pb. I 2(s) 2+ Pb + 2 I (aq) Dissolution Precipitation How can a change in p. H effect the position of equilibrium? Decreasing p. H = increasing [H+] Increasing p. H = increasing [OH-]
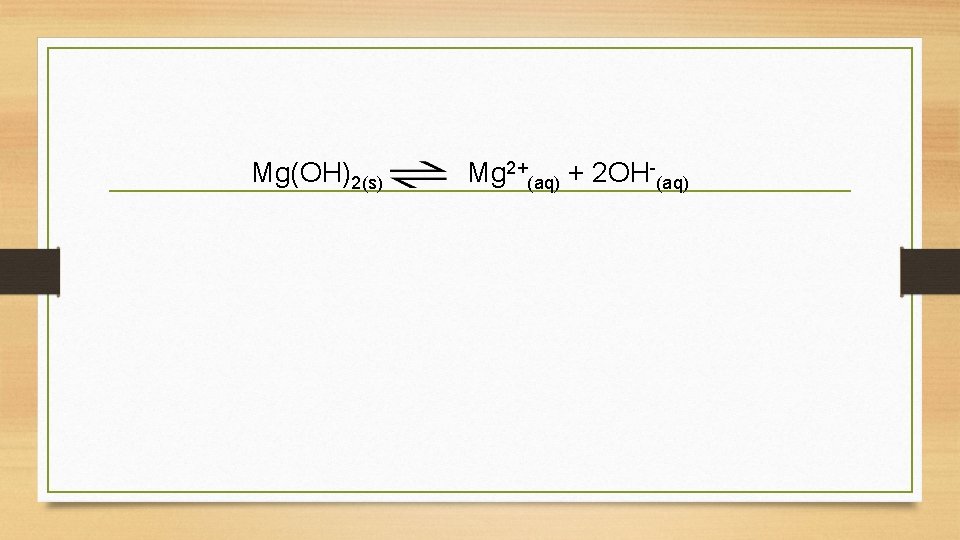
Mg(OH)2(s) Mg 2+(aq) + 2 OH-(aq)

The Equilibrium Constant • Based on the molar concentrations of all the different reaction species at equilibrium, we can define a quantity called the equilibrium constant (Kc) • Kc can also be written as Keq • The subscript c stands for concentration in mol/L (molarity, M) • Kc describes the molar concentrations at equilibrium for a specific temperature (usually 25°C) • The equilibrium constant can help us understand whether the reaction tends to have a higher concentration of products or reactants at equilibrium. • We can also use Kc to determine if the reaction is already at equilibrium.

a. A + b. B c. C + d. D • Given the reaction above the equilibrium constant can be written as follows: [C]c [D]d Kc = a [A] [B]b Products Reactants Upper case = molar concentration Lower case (exponent) = coefficient from balanced chemical equation Solids and liquids are not included. (g) (aq) The magnitude of Kc tells us whether an equilibrium reaction favors the products or reactants If Kc > 1 then the reaction favors the products If Kc > 10 the reaction proceeds almost totally towards If Kc < 1 then the reaction favors the reactantsproducts If Kc < 0. 1 the reaction hardly proceeds at all towards products

CO(g) + 3 H 2(g) CH 4(g) + H 2 O(g) • Calculate Kc at 25°C given the following concentrations at equilibrium: [CO] = 0. 850 M [H 2] = 1. 333 M 0. 286 M 27. 7 x 0. 286 = 3. 935 0. 850 x (1. 333)3 [CH 4] = 27. 70 M [H 2 O] = Kc is greater than 1, therefore the reaction favours the forwards reaction Kc = The formula above can be rearranged to find an unknown concentration at equilibrium when other concentrations are known

Equilibrium Constant Units • The units are calculated from the expression of products over reactants N 2 O 3(g) ↔ NO(g) + NO 2(g) • Examples: Expressio n: (mol dm-3) [NO] [NO 2] [N 2 O 3] H 2(g) + I 2(g) ↔ 2 HI(g) Recall that units concentration are mol dm-3 [HI]2 [H 2] [I 2] Units = mol dm-3 (mol dm-3)2 (mol dm-3) (mol dm 3) No units

4 NO(g) + 6 H 2 O(g) ↔ 4 NH 3(g) + 5 O 2(g) • A shortcut can be used to determine the units using the total powers for the top/bottom of the expression. [NH 3]4 [O 2]5 [NO]4 [H 2 O]6 ∆n = total power on top − total power on bottom (4 + 5) (4 + 6) ∆n = 9 − 10 = − 1 Units of Kc = (mol dm-3)-1 (mol)-1 = mol-1 (dm-3)-1 = dm 3 = mol-1 dm 3

Practice a) b) c) d) CO(g) + 3 H 2(g) ↔ CH 4(g) + H 2 O(g) 2 H 2(g) + O 2(g) ↔ 2 H 2 O(g) 2 NO(g) + O 2(g) ↔ 2 NO 2(g) 3 Ba. Cl 2(aq) + Al 2(SO 4)3(aq) ↔ 2 Al. Cl 3(aq) + 3 Ba. SO 4(s)

H 2(g) + I 2(g) ↔ 2 HI(g) • 5. 00 mol H 2 and 3. 00 mol I 2 are added to a 10. 0 dm 3 vessel and allowed to reach equilibrium at 1100 K. At equilibrium there were 0. 43 mol I 2 present in the reaction mixture. Calculate the value of Kc H 2(g) + I 2(g) ↔ 2 HI(g) Initial 5. 00 3. 00 0. 00 moles ? 0. 43 Eq moles ? The coefficient ratio from the balanced chemical equation can be used to find the moles of all species. Moles can then be converted to molarity (moles/volume)

CO(g) + 3 H 2(g) CH 4(g) + H 2 O(g) • Calculate the concentration of H 2 O at equilibrium [CO] = 0. 370 M [H 2] = 0. 580 M 3. 935 = [CH 4] [H 2 O] [CO] [H 2]3 [CH 4] = 11. 74 M [H 2 O] = ? [CO] [H 2]3 [H 2 O] = 3. 935 [CH ] 4 x = 0. 024 M

The Reaction Quotient • To determine whether a system has reached equilibrium, chemists use a Quantity called the reaction Quotient (Q). • The expression for the reaction quotient is the same as the equilibrium constant expression • The difference is the values for the reaction quotient expression can be measured at anytime, regardless of whether the system is at equilibrium

N 2 O 4(g) 2 NO 2(g) • Dissociation of dinitrogen tetroxide to nitrogen dioxide. • The direction of a reversible reaction to reach equilibrium depends on the magnitudes of Q (Qc) and Kc • Kc is a constant at a given temperature, however Q varies according to the concentrations of reactants and products present • If Q = Kc the system has reached equilibrium • If Q < Kc the reaction proceeds to the right to reach equilibrium • If Q > Kc the reaction proceeds to the left to reach equilibrium
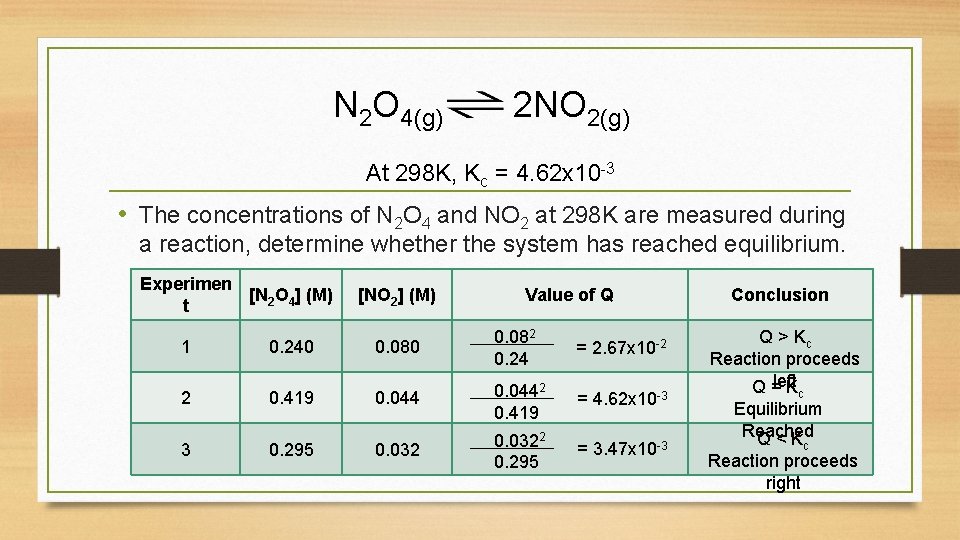
N 2 O 4(g) 2 NO 2(g) At 298 K, Kc = 4. 62 x 10 -3 • The concentrations of N 2 O 4 and NO 2 at 298 K are measured during a reaction, determine whether the system has reached equilibrium. Experimen [N 2 O 4] (M) t 1 0. 240 [NO 2] (M) Value of Q 0. 080 0. 082 0. 24 = 2. 67 x 10 -2 = 4. 62 x 10 -3 = 3. 47 x 10 -3 2 0. 419 0. 0442 0. 419 3 0. 295 0. 0322 0. 295 Conclusion Q > Kc Reaction proceeds left Q = K c Equilibrium Reached Q < K c Reaction proceeds right
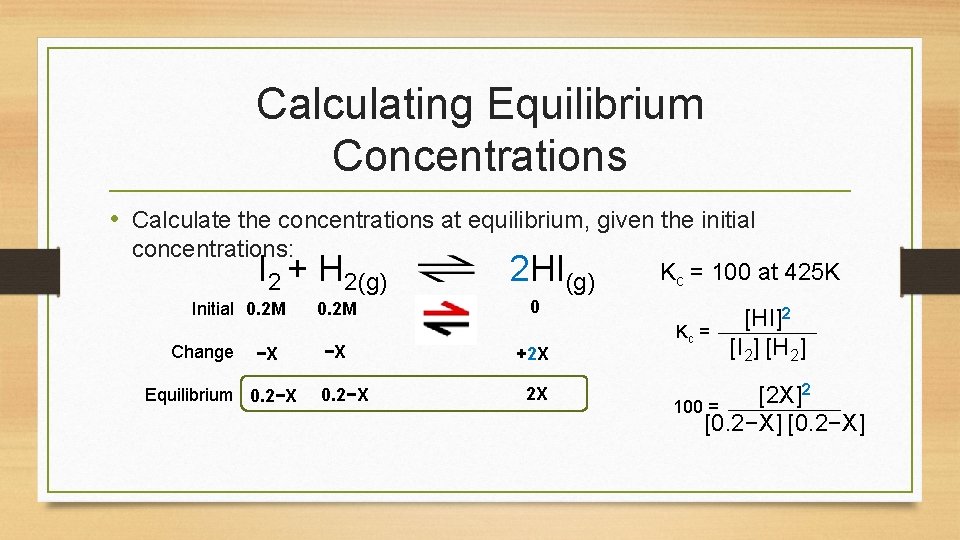
Calculating Equilibrium Concentrations • Calculate the concentrations at equilibrium, given the initial concentrations: I 2 + H 2(g) Initial 0. 2 M Change −X Equilibrium 0. 2−X 0. 2 M −X 0. 2−X 2 HI(g) Kc = 100 at 425 K 0 +2 X 2 X Kc = [HI]2 [I 2] [H 2] 2 [2 X] 100 = [0. 2−X]
![[2 X]2 100 = [0. 2−X] 2 X = 2 − 10 X √ [2 X]2 100 = [0. 2−X] 2 X = 2 − 10 X √](http://slidetodoc.com/presentation_image_h/e14249f576ef51ea47fb4ccea49864d7/image-39.jpg)
[2 X]2 100 = [0. 2−X] 2 X = 2 − 10 X √ 100 =√ [2 X]2 [0. 2−X]2 12 X = 2 X = 0. 167 10 = 1 [2 X] [0. 2−X] Cross multiply

I 2 + H 2(g) Initial 0. 2 M Change −X Equilibrium 0. 2−X 2 HI(g) 0. 2 M 0 −X +2 X 0. 2−X 2 X 0. 2 − 0. 167 = 0. 033 M 2 x 0. 167 = 0. 334 M X = 0. 167

Calculating Equilibrium Concentrations • The concentrations of each species are measured in the following system: I 2 + H 2(g) 0. 00623 M 0. 00414 M 0. 02242 • First we calculate Q 0. 00623 x 0. 00414 2 HI(g) 0. 0224 M = 19. 5 Kc = 54. 3 at 430°C Q < Kc Reaction proceeds right
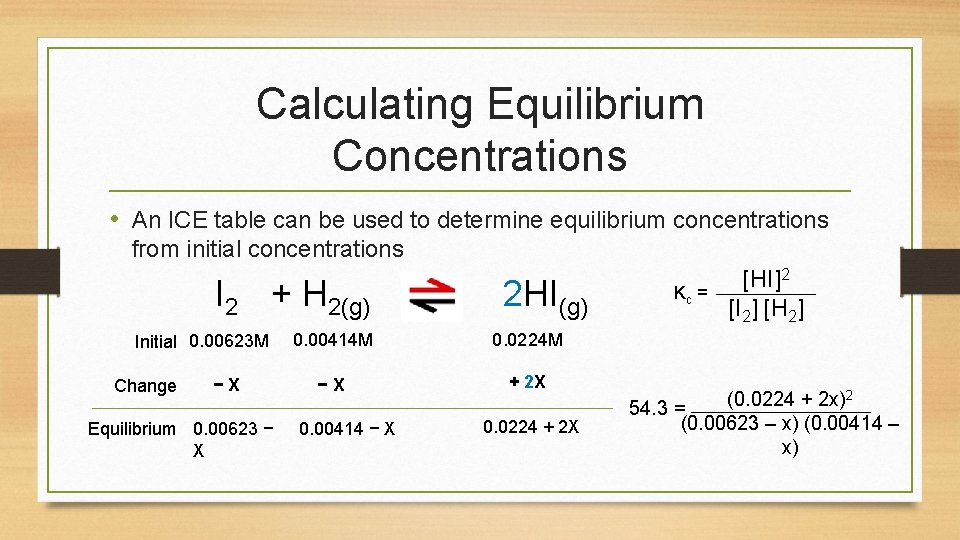
Calculating Equilibrium Concentrations • An ICE table can be used to determine equilibrium concentrations from initial concentrations I 2 + H 2(g) Initial 0. 00623 M Change − X Equilibrium 0. 00623 − X 2 HI(g) 0. 00414 M 0. 0224 M − X + 2 X 0. 00414 − X 0. 0224 + 2 X Kc = [HI]2 [I 2] [H 2] 2 (0. 0224 + 2 x) 54. 3 = (0. 00623 – x) (0. 00414 – x)

2 (0. 0224 + 2 x) 54. 3 = (0. 00623 – x) (0. 00414 – x) • Cannot take square root, need to use the quadratic method • FOIL the numerators and denominators First Outside Inner Last Don’t forget to carry the signs

(0. 0224 + 2 x) 54. 3 = (0. 00623 – x) (0. 00414 – x) 2 Numerators (0. 0224 + 2 X) F O I L 0. 0224 x 0. 00224 = 5. 02 x 10 -4 0. 0224 x 2 X = 0. 0448 X 2 X x 2 X = 4 X 2 5. 02 x 10 -4 + 0. 0448 X + 4 X 2 Denominators (0. 00623 – x) (0. 00414 – x) F O I L 0. 00623 x 0. 00414 = -5 2. 58 x 10 0. 00623 x −X = − 0. 00623 X −X x 0. 00414 = − 0. 00414 X −X x −X = X 2 2. 58 x 10 -5 − 0. 00623 X − 0. 00414 X + X 2

5. 02 x 10 -4 + 0. 0448 X + 54. 3 =2. 58 x 10 -5 − 0. 00623 X − 0. 00414 X + X 2 4 X 2 • Combine terms and cross multiply • Arrange in quadratic equation format 54. 3 = 1 0 = 8. 99 x 10 -4 − 0. 653 X + 50. 3 X 2 2 0 = 50. 3 X − 0. 653 X + 8. 99 x 10 4 a b c 5. 02 x 10 -4 + 0. 0896 X + 4 X 2 0. 0448 X + 0. 0448 X 2. 58 x 10 -5 − 0. 0104 X + X 2 (− 0. 00623 X) + (− 0. 00414 X) 5. 02 x 10 -4 +0. 0896 X +4 X 2 + (− 5. 02 x 10 -4) + (− 0. 0896 X) + (− 4 X 2 ) + =1. 401 x 10 -3 − 0. 563 X + (− 5. 02 x 10 54. 3 X 2 -4) + (− 0. 0896 X) + (− 4 X 2 )

0 = 50. 3 X 2 − 0. 653 X + 8. 99 x 10 -4 c a b X = 0. 653 ±√(-0. 653 x -0. 653) − 4(50. 3)(8. 99 x 104) 2 x 50. 3 X = 0. 0114 M or I 2 H 2(g) Equilibrium 0. 00623 − : X 0. 00467 M + 0. 00414 − X 0. 00258 M 0. 00156 M 2 HI(g) 0. 0224 + 2 X 0. 0255 M a = 50. 3 b = − 0. 653 c = 8. 99 x 10 -4

Equilibrium Pressure • The equilibrium constant, Kp, describes the ratio of product and reactant concentrations at equilibrium in terms of partial pressures. • For a gas-phase reaction, a. A(g) + b. B(g) ↔ c. C(g) + d. D(g), the expression for Kp is • Kp is related to the equilibrium constant in terms of molar concentration, Kc, by the equation below: Δn=mol of product gas−mol of reactant gas

• Recall that the units of P depend on the gas constant used Δn=mol of product gas−mol of reactant gas

• At temperature T, what is Kp for this reaction? P(N 2 O 5) = 2. 00 atm P(O 2) = 0. 296 atm P(NO 2) = 1. 70 atm

• If Kc for this reaction is 4. 5 x 104 at 400 K, what is the equilibrium constant, Kp, at the same temperature? Δn = mol of gas products − mol of gas reactants

Finding Kp from total pressure • Assume that initially there is no hydrogen or oxygen gas present. As the reaction proceeds to equilibrium the total pressure increases by 2. 10 atm Daltons Law

atm
- Slides: 52