Equilibrium Reactions are reversible ZA B C D

Equilibrium

Reactions are reversible ZA + B C + D ( forward) ZC + D A + B (reverse) Z Initially there is only A and B so only the forward reaction is possible Z As C and D build up, the reverse reaction speeds up while the forward reaction slows down. Z Eventually the rates are equal.
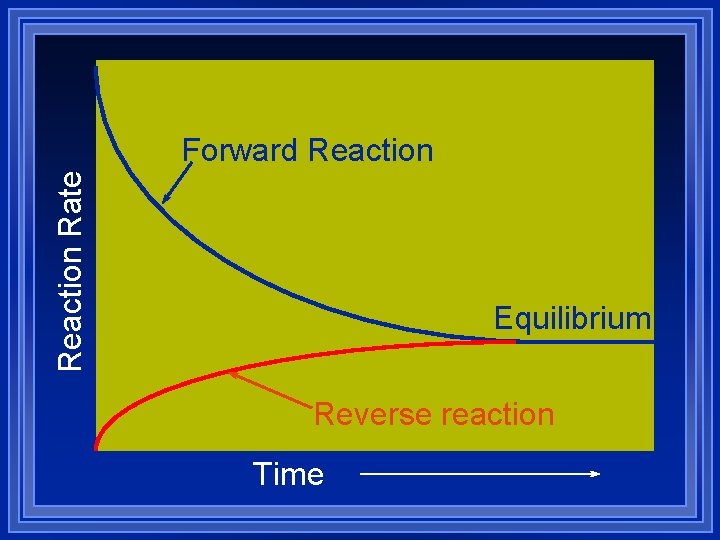
Reaction Rate Forward Reaction Equilibrium Reverse reaction Time

What is equal at Equilibrium? Z Rates are equal. Z Concentrations are not. Z Rates are determined by concentrations and activation energy. Z The concentrations of products and reactants do not change: aat equilibrium. aor if the reaction is verrrry slooooow.

Law of Mass Action Z For any reaction Z j. A + k. B Z K = [C]l[D]m [A]j[B]k l. C + m. D [PRODUCTS]power [REACTANTS]power Z K is called the equilibrium constant. Z is how we indicate a reversible reaction

Playing with K Z If we write the reaction in reverse, l. C + m. D j. A + k. B Z Then the new equilibrium constant is K’ = [A]j[B]k = 1/K [C]l[D]m

Playing with K Z If we multiply the equation by a constant (n), nj. A + nk. B nl. C + nm. D Z Then the equilibrium constant is nl[D]nm l[D]m)n [C] ( [C] K’ = [A]nj[B]nk = ([A] j[B]k)n n K =

The units for K Z NONE! Z BUT, you have to make sure you use the correct units throughout your calculation of K.

K is CONSTANT Z At any specific temperature. Z Temperature affects rate! Z The equilibrium concentrations don’t have to be the same, only K. Z Equilibrium position is a set of concentrations at equilibrium. Z There an unlimited number of K’s.

Equilibrium Constant One for each Temperature

Calculate K Z N 2 + 3 H 2 Z Initial 2 NH 3 At Equilibrium [N 2]0 =1. 000 M [N 2] = 0. 921 M [H 2]0 =1. 000 M [H 2] = 0. 763 M [NH 3]0 =0 M [NH 3] = 0. 157 M

Calculate K Z N 2 + 3 H 2 Z Initial 2 NH 3 At Equilibrium [N 2]0 = 0 M [N 2] = 0. 399 M [H 2]0 = 0 M [H 2] = 1. 197 M [NH 3]0 = 1. 000 M [NH 3] = 0. 203 M Z K is the same for a given reaction, no matter what the amount of starting materials!

Equilibrium and Pressure Z Some reactions are gaseous. Z Ideal Gas Law: PV = n. RT Z P = (n/V)RT Z P = CRT, where C is a concentration in moles/liter (M). Z C = M = P/RT

Equilibrium and Pressure Z 2 SO 2(g) + O 2(g) Z KP = (PSO 3)2 2 SO 3(g) (PSO 2)2 (PO 2) Z KC [SO 3]2 = [SO 2]2 [O 2] = (PSO 3/RT)2 (PSO 2/RT)2 (PO 2/RT)

Equilibrium and Pressure Z Z Z K K = = (PSO 3/RT)2 (PSO 2/RT)2 (PO 2/RT) (PSO 3)2 (1/RT)2 (PSO 2)2(PO 2) (1/RT)3 K = Kp (1/RT)2 = Kp RT (1/RT)3

General Equation Z j. A + k. B l. C + m. D Z Kp= (PC)l (PD)m= (CCx. RT)l (CDx. RT)m (PA)j (PB)k (CAx. RT)j(CBx. RT)k Z Kp= (CC)l (CD)m x (RT)l+m (CA)j(CB)k x (RT)j+k Z Kp = K (RT)(l+m)-(j+k) = K (RT)Dn Z Dn=(l+m)-(j+k)=Change in moles of gas

Homogeneous Equilibria Z So far every example dealt with reactants and products where all were in the same phase. Z We can use K in terms of either concentration or pressure. Z Units depend on reaction.

Heterogeneous Equilibria Z If the reaction involves pure solids or pure liquids, the concentration of the solid or the liquid doesn’t change. Z As long as they are not used up we can leave them out of the equilibrium expression. Z For example:
![For Example Z H 2(g) + I 2(s) Z K = [HI]2 2 HI(g) For Example Z H 2(g) + I 2(s) Z K = [HI]2 2 HI(g)](http://slidetodoc.com/presentation_image_h/0b26b815e1d12b1f53428900f5782fae/image-19.jpg)
For Example Z H 2(g) + I 2(s) Z K = [HI]2 2 HI(g) [H 2][I 2] Z But the concentration of I 2(s) does not change, so it doesn’t affect K. Z K= [HI]2 [ H 2]

Write the equilibrium constant for the heterogeneous reaction

The Reaction Quotient Z Tells you the direction the reaction will go to reach equilibrium Z Calculated the same as the equilibrium constant, but for a system not at equilibrium Z Q = [Products]0 coefficient [Reactants]0 coefficient Z Compare value to equilibrium constant

What Q tells us Z If Q<K : Not enough products : Shift to right Z If Q>K : Too many products : Shift to left Z If Q=K system is at equilibrium

Example Z for the reaction Z 2 NOCl(g) 2 NO(g) + Cl 2(g) Z K = 1. 55 x 10 -5 M at 35ºC Z In an experiment 0. 10 mol NOCl, 0. 0010 mol NO(g) and 0. 00010 mol Cl 2 are mixed in 2. 0 L flask. Z Which direction will the reaction proceed to reach equilibrium?

Solving Equilibrium Problems Z Given the starting concentrations and one equilibrium concentration. Z Use stoichiometry to figure out other concentrations and K. Z Learn to create a table of initial and final conditions.

Z Consider the following reaction at 600ºC Z 2 SO 2(g) + O 2(g) 2 SO 3(g) Z In a certain experiment 2. 00 mol of SO 2, 1. 50 mol of O 2 and 3. 00 mol of SO 3 were placed in a 1. 00 L flask. At equilibrium 3. 50 mol of SO 3 were found to be present. Calculate Z The equilibrium concentrations of O 2 and SO 2, K and KP

Z Consider the same reaction at 600ºC 2 SO 2(g) + O 2(g) 2 SO 3(g) Z In a different experiment. 500 mol SO 2 and. 350 mol SO 3 were placed in a 1. 000 L container. When the system reaches equilibrium 0. 045 mol of O 2 are present. Z Calculate the final concentrations of SO 2 and SO 3 and K

Solving Equilibrium Problems

Type 1 Problems with large K K > 100

What if you’re not given equilibrium concentration? Z The size of K will determine what approach to take. Z First let’s look at the case of a LARGE value of K ( >100). Z Allows us to make simplifying assumptions.

Example Z H 2(g) + I 2(g) 2 HI(g) Z K = 7. 1 x 102 at 25ºC Z Calculate the equilibrium concentrations if a 5. 00 L container initially contains 15. 8 g of H 2 294 g I 2. Z [H 2]0 = (15. 8 g/2. 02)/5. 00 L = 1. 56 M Z [I 2]0 = (294 g/253. 8)/5. 00 L = 0. 232 M Z [HI]0 = 0

Z Q= 0<K so more product will be formed. Z Set up table of initial, final and change in concentrations. Z Assumption since K is large- reaction will almost go to completion. Z Stoichiometry tells us I 2 is LR, it will be smallest at equilibrium let it be x
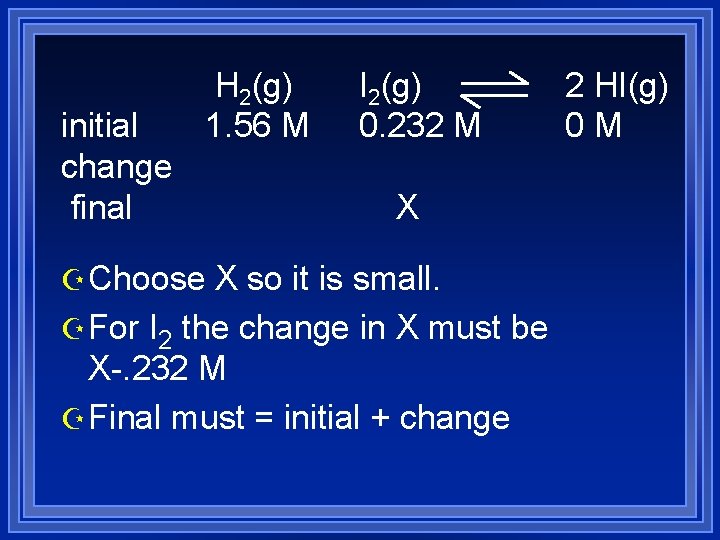
initial change final H 2(g) 1. 56 M I 2(g) 0. 232 M X Z Choose X so it is small. Z For I 2 the change in X must be X-. 232 M Z Final must = initial + change 2 HI(g) 0 M

initial change final H 2(g) 1. 56 M I 2(g) 0. 232 M X-0. 232 M X 2 HI(g) 0 M Z Using to stoichiometry we can find Z Change in H 2 = X-0. 232 M Z Change in HI = -twice change in H 2 Z Change in HI = 0. 464 -2 X
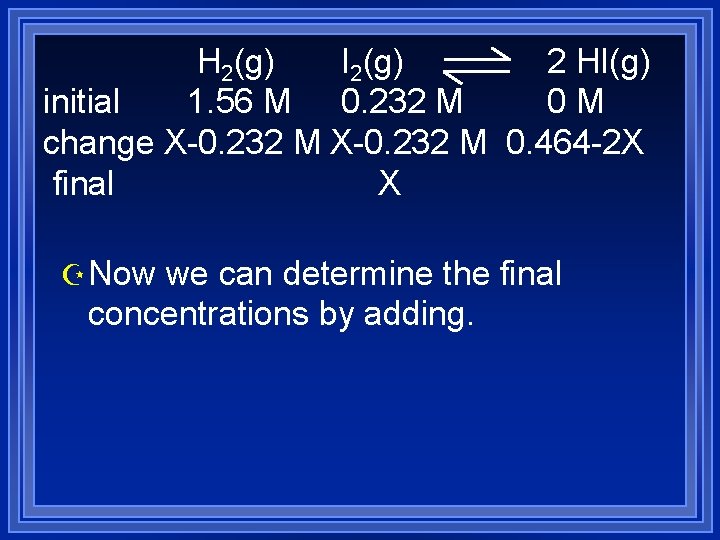
H 2(g) I 2(g) 2 HI(g) initial 1. 56 M 0. 232 M 0 M change X-0. 232 M 0. 464 -2 X final X Z Now we can determine the final concentrations by adding.

H 2(g) I 2(g) initial 1. 56 M 0. 232 M change X-0. 232 M final 1. 328+X X 2 HI(g) 0 M 0. 464 -2 X Z Now plug these values into the equilibrium expression Z K = (0. 464 -2 X)2 = 7. 1 x 102 (1. 328+X)(X)

Why we chose X (0. 464 -2 X)2 = 7. 1 x 102 (1. 328+X)(X) Z Since X is going to be small, we can ignore it in relation to 0. 464 and 1. 328 Z So we can rewrite the equation Z 7. 1 x 102 = (0. 464)2 ZK = (1. 328)(X) Z Makes the algebra easy

H 2(g) I 2(g) initial 1. 56 M 0. 232 M change X-0. 232 M final 1. 328+X X 2 HI(g) 0 M 0. 464 -2 X Z When we solve for X we get 2. 3 x 10 -4 Z So we can find the other concentrations Z I 2 = 2. 3 x 10 -4 M Z H 2 = 1. 328 M Z HI = 0. 464 M

Checking the assumption Z The rule of thumb is that if the value of X is less than 5% of the smallest concentration, our assumption was valid. Z If not we would have had to use the quadratic equation Z More on this later. Z Our assumption was valid.

Practice Z For the reaction Cl 2 + O 2 2 Cl. O(g) K = 156 Z In an experiment 0. 100 mol Cl. O, 1. 00 mol O 2 and 0. 0100 mol Cl 2 are mixed in a 4. 00 L flask. Z If the reaction is not at equilibrium, which way will it shift? Z Calculate the equilibrium concentrations.

At an elevated temperature, the reaction: has a value of Keq = 1. 01 x 103. If 0. 234 mol IBr is placed in a 1. 00 L. flask and allowed to reach equilibrium, what is the equilibrium concentration in M. of I 2?

Type 2 Problems with small K K<. 01

Process is the same Z Set up table of initial, change, and final concentrations. Z Choose X to be small. Z For this case it will be a product. Z For a small K the product concentration is small.

For example Z For the reaction 2 NOCl Z K= 1. 6 x 10 -5 2 NO +Cl 2 Z If 1. 20 mol NOCl, 0. 45 mol of NO and 0. 87 mol Cl 2 are mixed in a 1 L container Z What are the equilibrium concentrations Z Q = [NO]2[Cl 2] = (0. 45)2(0. 87) = 0. 15 M [NOCl]2 (1. 20)2
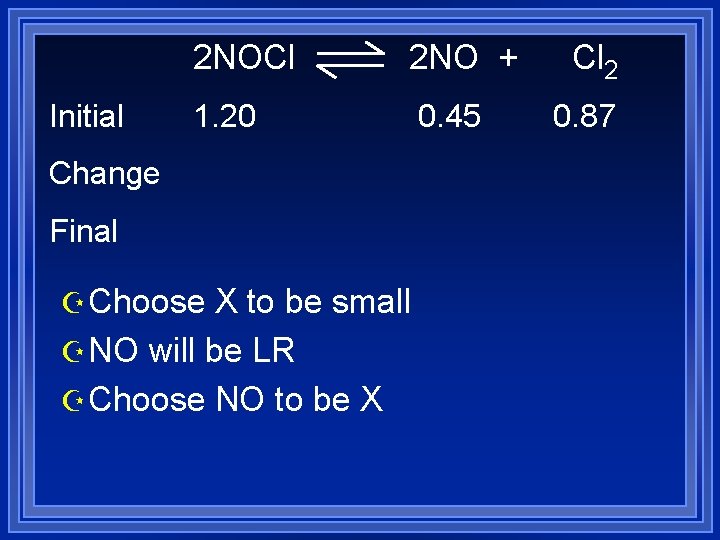
Initial 2 NOCl 2 NO + 1. 20 0. 45 Change Final Z Choose X to be small Z NO will be LR Z Choose NO to be X Cl 2 0. 87
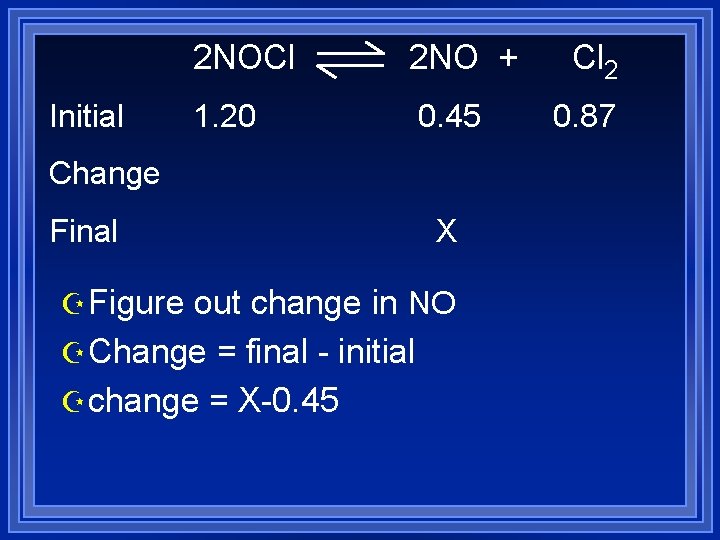
Initial 2 NOCl 2 NO + 1. 20 0. 45 Change Final X Z Figure out change in NO Z Change = final - initial Z change = X-0. 45 Cl 2 0. 87

Initial 2 NOCl 2 NO + 1. 20 0. 45 Change Final Cl 2 0. 87 X-. 45 X Z Now figure out the other changes Z Use stoichiometry Z Change in Cl 2 is 1/2 change in NO Z Change in NOCl is - change in NO

2 NOCl 2 NO + Initial 1. 20 0. 45 Change 0. 45 -X X-. 45 0. 5 X -. 225 Final l Now X we can determine final concentrations l Add Cl 2 0. 87

2 NOCl 2 NO + Initial 1. 20 0. 45 Change 0. 45 -X X-. 45 0. 5 X -. 225 Final 1. 65 -X l Now X Cl 2 0. 87 0. 5 X +0. 645 we can write equilibrium constant l K = (X)2(0. 5 X+0. 645) (1. 65 -X)2 l Now we can test our assumption X is small ignore it in + and -

2 NOCl 2 NO + Initial 1. 20 0. 45 Change 0. 45 -X X-. 45 0. 5 X -. 225 Final 1. 65 -X X Cl 2 0. 87 0. 5 X +0. 645 = (X)2(0. 645) = 1. 6 x 10 -5 (1. 65)2 l X= 8. 2 x 10 -3 l Figure out final concentrations l. K

2 NOCl 2 NO + Initial 1. 20 0. 45 Change 0. 45 -X X-. 45 0. 5 X -. 225 Final 1. 65 -X l [NOCl] X Cl 2 0. 87 0. 5 X +0. 645 = 1. 64 l [Cl 2] = 0. 649 l Check assumptions l. 0082/. 649 = 1. 2 % OKAY!!!

Practice Problem Z For the reaction 2 Cl. O(g) Cl 2 (g) + O 2 (g) Z K = 6. 4 x 10 -3 Z In an experiment 0. 100 mol Cl. O(g), 1. 00 mol O 2 and 1. 00 x 10 -2 mol Cl 2 are mixed in a 4. 00 L container. ZWhat are the equilibrium concentrations?

Type 3 Mid-range K’s. 01<K<100

No Simplification Z Choose X to be small. Z Can’t simplify so we will have to solve the quadratic (we hope) Z H 2(g) + I 2 (g) 2 HI(g) K=38. 6 Z What is the equilibrium concentrations if 1. 800 mol H 2, 1. 600 mol I 2 and 2. 600 mol HI are mixed in a 2. 000 L container?

Problems Involving Pressure Z Solved exactly the same, with same rules for choosing X depending on KP Z For the reaction N 2 O 4(g) 2 NO 2(g) KP =. 131. What are the equilibrium pressures if a flask initially contains 1. 000 atm N 2 O 4?

Le Châtelier’s Principle Z If a stress is applied to a system at equilibrium, the position of the equilibrium will shift to reduce the stress. Z 3 Types of stress a. Concentration a. Pressure a. Temperature

Change amounts of reactants and/or products Z Adding product makes Q>K Z Removing reactant makes Q>K Z Adding reactant makes Q<K Z Removing product makes Q<K Z Determine the effect on Q, will tell you the direction of shift

Change Pressure Z By changing volume Z System will move in the direction that has the least moles of gas. Z Because partial pressures (and concentrations) change, a new equilibrium must be reached. Z System tries to minimize the moles of gas if volume is reduced Z And visa versa

Change in Pressure Z By adding an inert gas Z Partial pressures of reactants and product are not changed Z No effect on equilibrium position

Change in Temperature Z Affects the rates of both the forward and reverse reactions. Z Doesn’t just change the equilibrium position, changes the equilibrium constant. Z The direction of the shift depends on whether it is exo- or endothermic

Exothermic Z DH<0 Z Releases heat Z Think of heat as a product Z Raising temperature push toward reactants. Z Shifts to left.

Endothermic Z DH>0 Z Produces heat Z Think of heat as a reactant Z Raising temperature push toward products. Z Shifts to right.
- Slides: 61