EQUILIBRIUM REACTION RATES ENERGY Chemical energy of a

EQUILIBRIUM REACTION RATES + ENERGY

Chemical energy of a substance = potential + kinetic energy. • Kinetic energy = doing energy (mechanical) • Potential energy = stored energy

What these energies result from • • • Attractions between electrons and protons Repulsion between nuclei Repulsion between electrons Movement of electrons Vibration and rotation of nuclei

Enthalpy • Is the Chemical energy of a substance is • sometimes also called its HEAT CONTENT • given the sign H
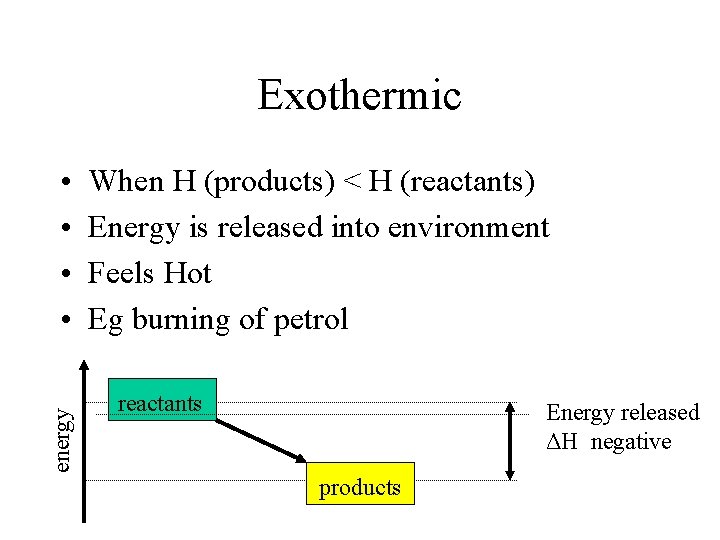
Exothermic energy • • When H (products) < H (reactants) Energy is released into environment Feels Hot Eg burning of petrol reactants Energy released ΔH negative products
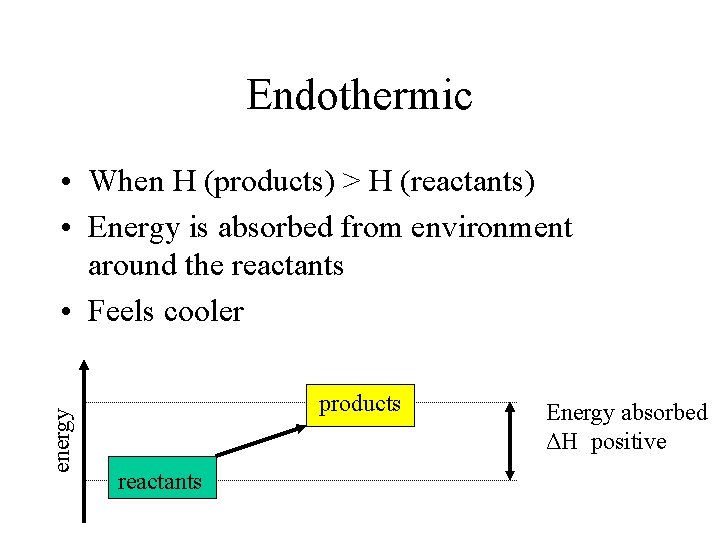
Endothermic energy • When H (products) > H (reactants) • Energy is absorbed from environment around the reactants • Feels cooler products reactants Energy absorbed ΔH positive

ΔH – Delta H • ΔH = energy (products) – energy (reactants) • The energy released or absorbed in a chemical reaction is called the HEAT OF REACTION • This heat of reaction is the difference in the enthalpy’s of the products and reactants (hence ΔH)

Examples • Exothermic reactions – Burning of fuels including food • Endothermic reactions – Photosynthesis (absorbs energy from sunlight to convert H 2 O and CO 2 into glucose and O 2

Thermochemical equations • Show the energy released or absorbed durin a chemical reaction • Energy is measured in Joules (J) or kilo joules (k. J) • The heat of reaction (ΔH ) has the units j mol– 1 of k. J mol– 1 • The energy is therefore related to the number of moles as given by the equation
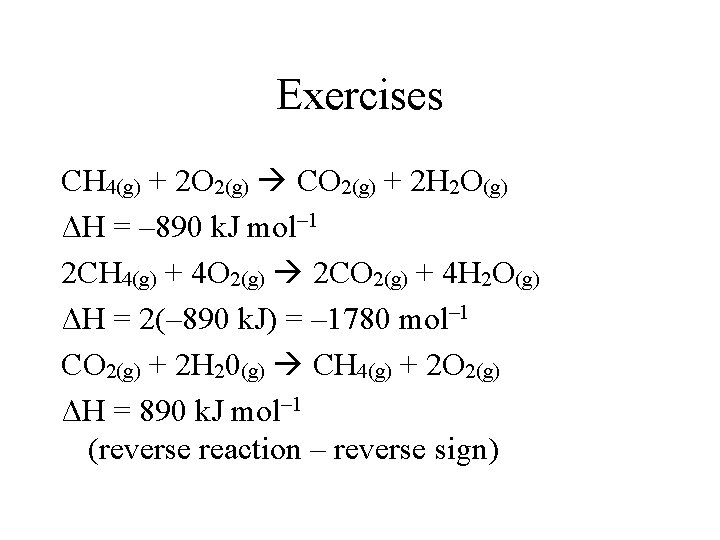
Exercises CH 4(g) + 2 O 2(g) CO 2(g) + 2 H 2 O(g) ΔH = – 890 k. J mol– 1 2 CH 4(g) + 4 O 2(g) 2 CO 2(g) + 4 H 2 O(g) ΔH = 2(– 890 k. J) = – 1780 mol– 1 CO 2(g) + 2 H 20(g) CH 4(g) + 2 O 2(g) ΔH = 890 k. J mol– 1 (reverse reaction – reverse sign)

Chemical bonds during a chemical reaction • Bonds in the reactants must first be broken – This requires energy to be absorbed • New bonds are created in the products – This requires energy to be released

Activation energy • The energy required to break bonds in the reactants so that the reaction can proceed.
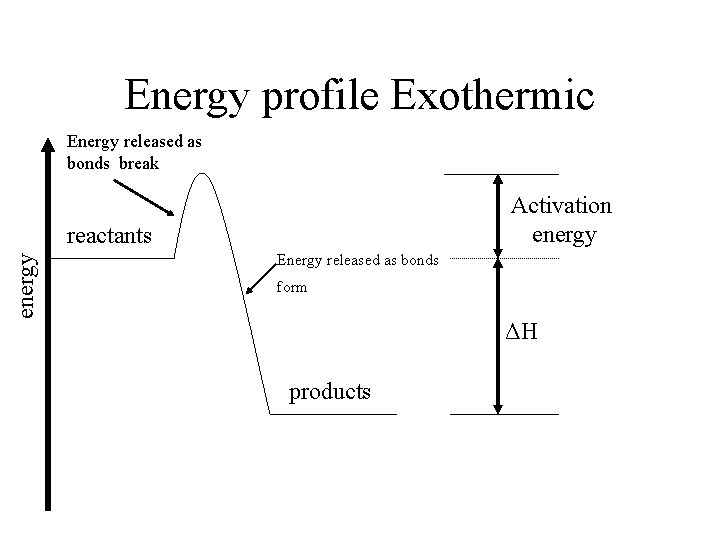
Energy profile Exothermic Energy released as bonds break Activation energy reactants Energy released as bonds form ΔH products
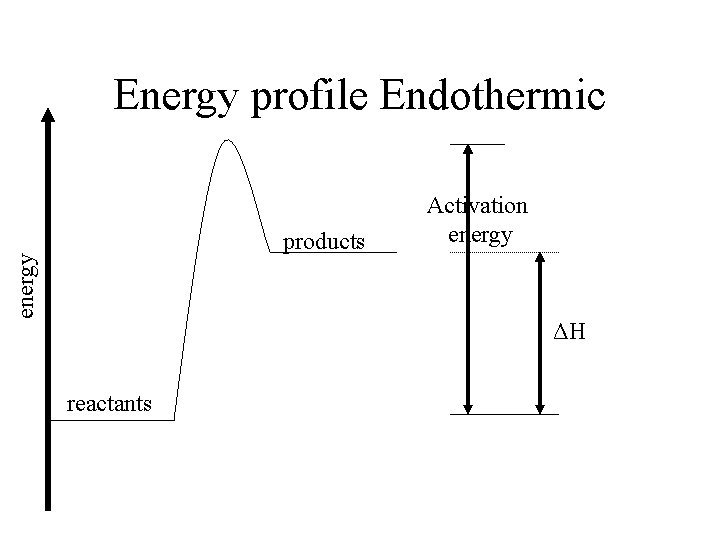
Energy profile Endothermic energy products Activation energy ΔH reactants

Factors affecting rate of a reaction • Increasing the surface area of solids • Increasing concentration of reactants or pressure of gases • Increasing temperature • Adding a catalyst

Factors affecting rate of a reaction • Increasing the surface area of solids – Breaking reactant into smaller pices increases the surface area – More particles are present on the surface – The greater number of particles exposed allows for greater number of collisions with other reactant particles

Factors affecting rate of a reaction • Increasing concentration of reactants or pressure of gases – A greater number of particles moving in a given volume of solution will mean a greater chance of collision between particles

Factors affecting rate of a reaction • Increasing temperature – As temperature increases the average speed and kinetic energy of particles increases – This results in a greater chance of collisions occurring

A Catalyst • A substance that will increase the rate of a chemical reaction without being consumed in that reaction - 2 Types • Homogenous – Same state as the reactants eg Atmospheric reactions • Heterogeneous – Different state to the reactants – Easier to separate out at the conclusion of reaction

How catalysts work Haber process – N 2(g) + 3 H 2(g) 2 NH 3 (g) ΔH = – 91 k. J mol – 1 – Catalysed with powdered Iron – N 2 and H 2 absorb onto surface of Fe – As they absorb, bonds within them break – N and H molecules now readily form NH 3 molecules and move away from Fe
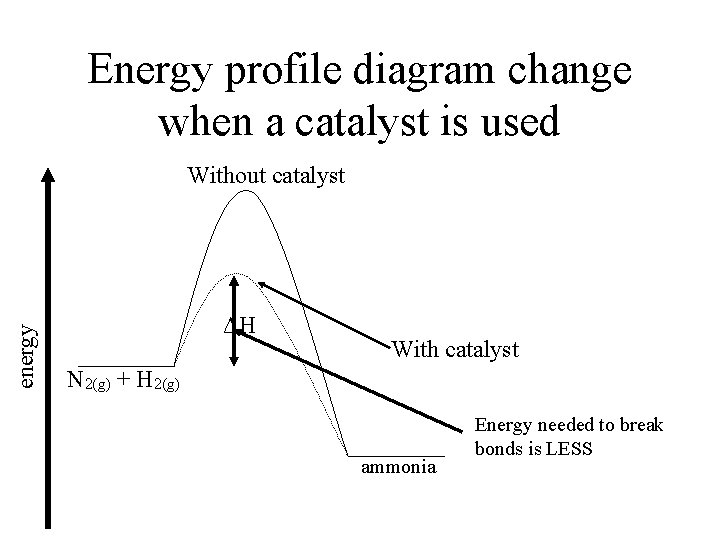
Energy profile diagram change when a catalyst is used energy Without catalyst ΔH With catalyst N 2(g) + H 2(g) ammonia Energy needed to break bonds is LESS
- Slides: 21