Equilibrium Nothing ever changes Does it 352021 1

Equilibrium Nothing ever changes. Does it? 3/5/2021 1

Phase Equilibrium l The laws of thermodynamics determine equilibrium between phases. l The fundamental fact of phase equilibrium is that at equilibrium the chemical potential of any substance must have the same value in all phases in which that substance appears. 3/5/2021 2

Phase Equilibrium l Phase: ¡a portion of a system or an entire system wherein the intensive properties do not change abruptly as a function of position. ¡One of the many homogeneous portions of a heterogeneous system 3/5/2021 A heterogeneous system 3
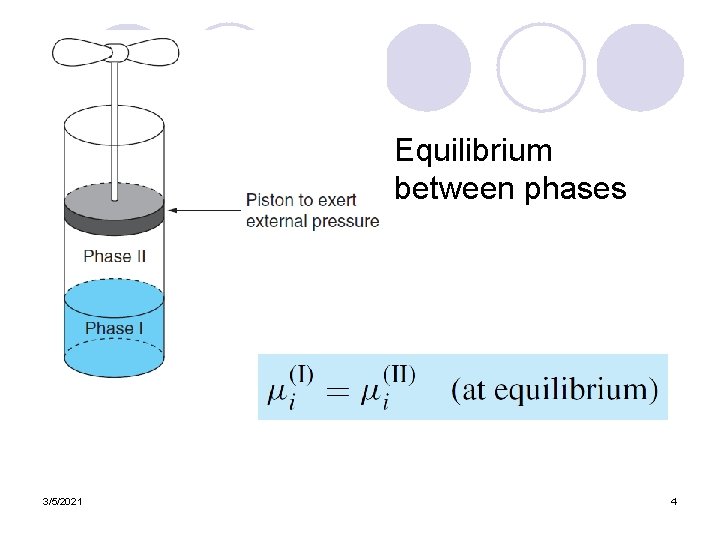
Equilibrium between phases 3/5/2021 4
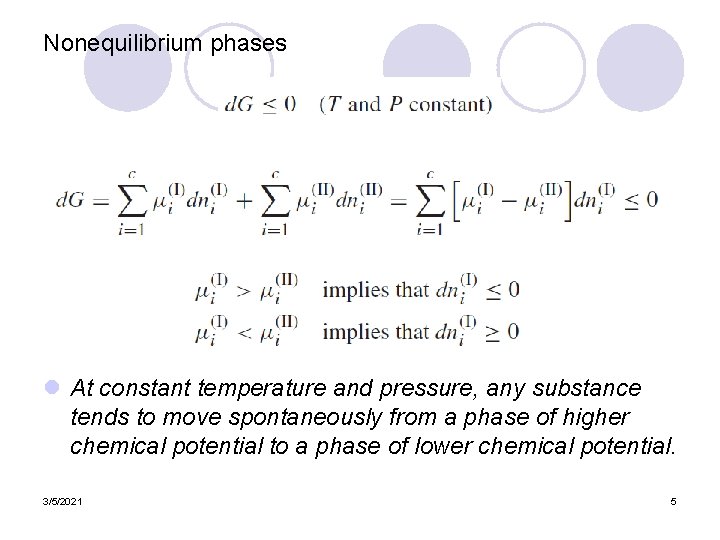
Nonequilibrium phases l At constant temperature and pressure, any substance tends to move spontaneously from a phase of higher chemical potential to a phase of lower chemical potential. 3/5/2021 5

Gibbs Phase Rule l The Gibbs phase rule gives the number of independent intensive variables in a simple system that can have a number of phases and components. l The equilibrium thermodynamic state of a single-phase simple system with c components is specified by the values of c + 2 thermodynamic variables, at least one of which must be an extensive variable. l All other equilibrium variables are dependent variables. 3/5/2021 6

l The coexistence of two phases in equilibrium implies that: l This means that the two intensive variables needed to describe the state of a system are now related and no longer independent. 3/5/2021 7

Thermodynamic Cat says: The Gibbs phase rule is one of the most elegant deductions of the whole of chemical thermodynamics and one of the truly great generalizations of the physical sciences. • Where f is the: • Degrees of freedom • Variance • the number of independent intensive variables in a simple system that can have a number of phases and components. 3/5/2021 8

But how do we count the number of components? Is number of components = number of chemical species? Actually, NO! 3/5/2021 9

l Components ¡Chemically independent species ¡The number of components is equal to the number of chemical species present minus the number of relations that constrain the amounts of the species l Relations due to chemical equilibrium; l a relation due to a requirement of electrical neutrality (which we always assume to exist); l relations due to the way the system was prepared (such as a specification that two substances are in their stoichiometric ratio) Wise sensei, is this true? 3/5/2021 my Son, 10
Example: l Determine the number of components in: ¡An aqueous solution containing Na+, Cl−, and Br−. ¡An aqueous solution containing Na+, K+, Li+, Cl−, and Br−. ¡A gaseous system containing NO 2 and N 2 O 4 at chemical equilibrium with each other. ¡An aqueous solution containing Ca 2+ ions and Cl− ions. 3, 5, 1, 2 3/5/2021 11

Phase equilibria in one-component systems l f = 1 – 1 + 2 = 2 (one component, one phase) ¡ Temperature and pressure can both be independent and any point in a plot of the T-P graph can represent a possible intensive state of the system l f = 1 – 2 + 2 = 1 (one component, two phases) ¡ The pressure must be a function of the temperature, and is represented by a coexistence curve. l f = 1 – 3 + 2 = 0 (one component, three phases) ¡ This three-phase state is represented by a triple point at which three coexistence curves intersect. 3/5/2021 12

Examples: l A system composed of solid sucrose in equilibrium with an aqueous solution of sucrose. l SO 2, SO 3, and O 2 in a one-phase gaseous system, with the chemical reaction among these substances at equilibrium l Ice and liquid water. l CO, O 2, and CO 2 in a single gas phase, with no catalyst present so that the chemical reaction cannot equilibrate, and with each substance added separately. l An aqueous solution of acetic acid. Make a list of the major species present in the solution. 3/5/2021 13
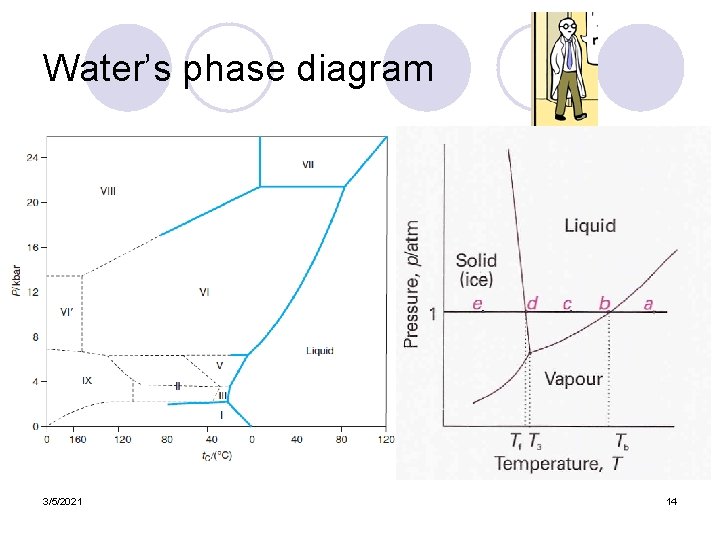
Water’s phase diagram 3/5/2021 14
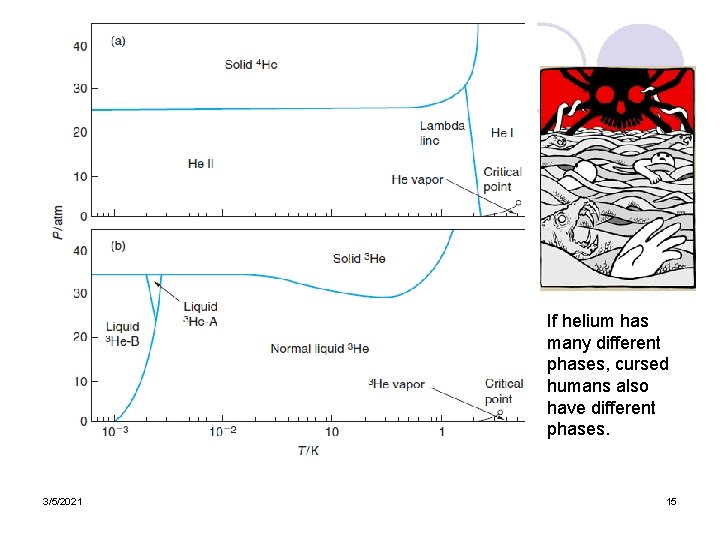
If helium has many different phases, cursed humans also have different phases. 3/5/2021 15

Clapeyron equation 3/5/2021 16

Example l Estimate the pressure on a system of liquid and solid water if the equilibrium melting temperature is equal to − 0. 100°C. The density of ice is 0. 917 g/cm 3, the density of liquid water is 1. 000 g/cm 3, and the molar enthalpy change of fusion is 6008 J/mol. 14. 3 atm l Integrate the Clapeyron equation for a solid–solid or liquid–solid phase transition under the assumption that ΔVm is constant and that ΔHm(T) = ΔHm(T 1) + ΔCP, m(T − T 1) where ΔCP, m is constant. 3/5/2021 17

Clausius-Clapeyron Equation l The Clausius–Clapeyron equation is obtained by integrating the Clapeyron equation in the case that one of the two phases is a vapor (gas) and the other is a condensed phase (liquid or solid). l We make two approximations: ¡ (1) that the vapor is an ideal gas, and ¡ (2) that the molar volume of the condensed phase is negligible compared with that of the vapor (gas) phase. ¡ These are both good approximations 3/5/2021 18
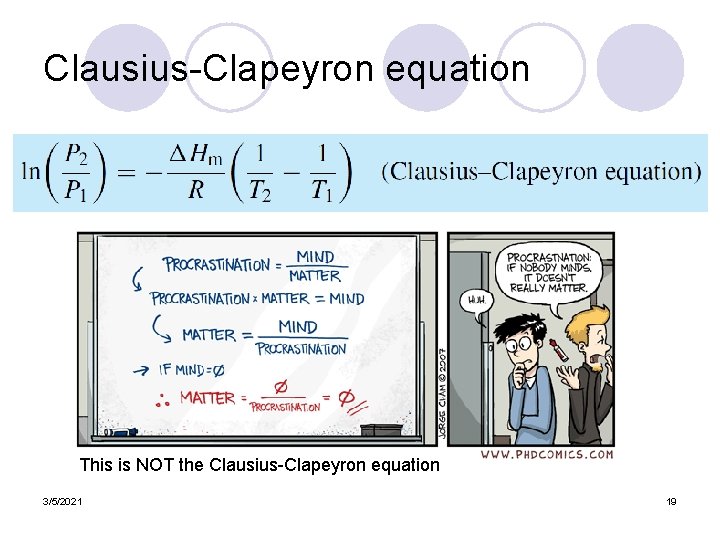
Clausius-Clapeyron equation This is NOT the Clausius-Clapeyron equation 3/5/2021 19

Example l Calculate the enthalpy change of vaporization of water given the following values: ¡ Vapor pressure of water at 25. 0°C = 23. 756 torr ¡ Vapor pressure of water at 100. 0°C = 760. 0 torr 3/5/2021 20
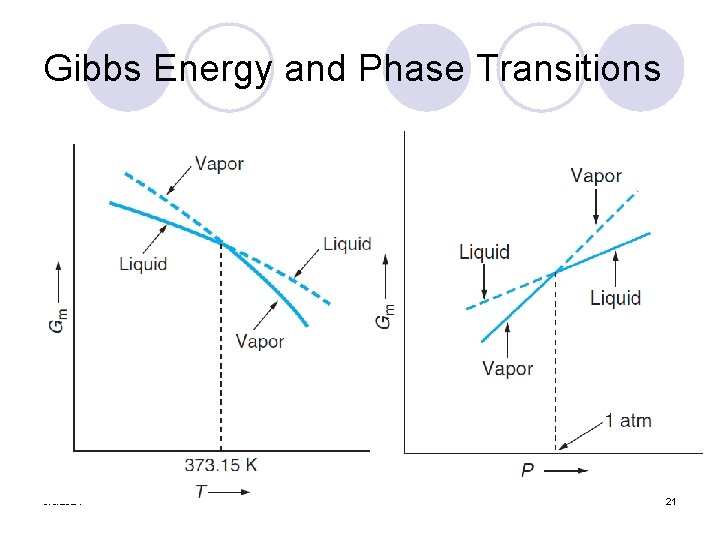
Gibbs Energy and Phase Transitions 3/5/2021 21
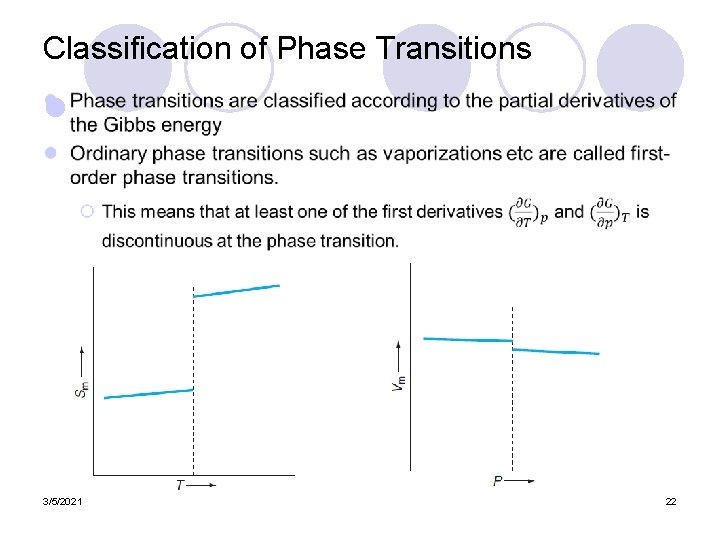
Classification of Phase Transitions l 3/5/2021 22
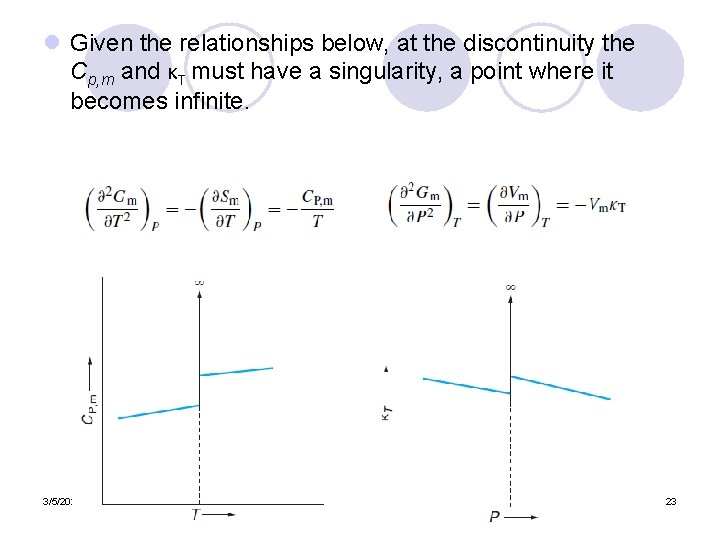
l Given the relationships below, at the discontinuity the Cp, m and κT must have a singularity, a point where it becomes infinite. 3/5/2021 23

Second order phase transitions l ¡ A transition wherein both of the first derivatives of the Gibbs energy are continuous but at least one of the second derivatives is discontinuous. ¡ The order of a phase transition must be determined experimentally through careful measurements of the isothermal compressibility and heat capacity at the phase transition. ¡ An example of a second-order phase transition is the transition between normal and superconducting states 3/5/2021 24
Other kinds of transitions l ¡Paramagnetic-toferromagnetic transitions in some magnetic materials ¡Order-disorder transitions in certain alloys such as beta brass 3/5/2021 25
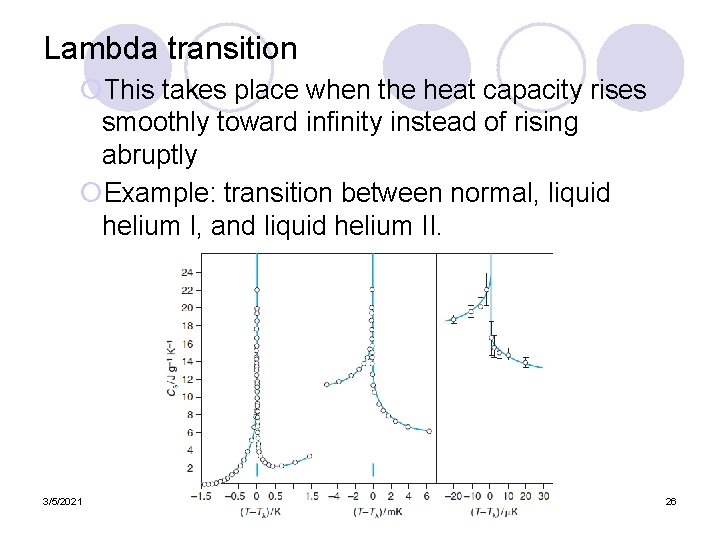
Lambda transition ¡This takes place when the heat capacity rises smoothly toward infinity instead of rising abruptly ¡Example: transition between normal, liquid helium I, and liquid helium II. 3/5/2021 26

Surfaces ¡Many of thermodynamic equations discussed are valid only when surface contributions to the energy can be neglected. ¡Although this is ordinarily an excellent approximation, there is a significant contribution to the energy of a liquid by the surface of the phase in the case of a small droplet or a liquid in a small capillary tube. ¡Many products and reactions involve surface effects, such as colloids, biological cell membranes, lubrication, corrosion, adhesion, detergency, lubrication, and electrochemical cell reactions. 3/5/2021 27

Energy attributed to a surface ¡The surface contribution to the energy of a liquid is primarily due to intermolecular attractions. ¡Although this is ordinarily an excellent approximation, there is a significant contribution to the energy of a liquid by the surface of the phase in the case of a small droplet or a liquid in a small capillary tube. ¡Many products and reactions involve surface effects, such as colloids, biological cell membranes, lubrication, corrosion, adhesion, detergency, lubrication, and electrochemical cell reactions. 3/5/2021 28

Nanoscale Thermodynamics ¡Thermodynamic properties change as objects become smaller, significantly so in nanoscale. ¡Melting point and enthalpy of fusion decreases as the size of a nanoparticle decreases. ¡Thermodynamics becomes less and less applicable as particle sizes become smaller. 3/5/2021 29

Nanoparticles • In nanoparticles and other nanoscale objects, their properties begin to depend more and more on surface effects, electrostatic interactions, molecular interactions, and even quantum mechanics. From http: //cfpub. epa. gov/ncer_abstracts/index. cfm/fuseaction/display. abstract. Detail/abstract/7136/report/F http: //cheed. nus. edu. sg/~cheleejy/Gallery. html 30
Nanoparticles From http: //upload. wikimedia. org/wikipedia/commons/6/6 b/Colloidal. Gold_aq. png http: //www. topnews. in/files/stained-glass. jpg Buzea, 2007 31

Nanoparticles: Fabrication l l Gas phase methods Vapor deposition synthesis Mechanical processes Liquid phase methods From http: //cheed. nus. edu. sg/~cheleejy/Gallery. html http: //www. ptl. ethz. ch/research/res_top_FSP http: //www. manmadediamondinfo. com/cvd. shtml http: //www. cuttingtoolssite. com/2011/02/25/ball-milling-2/ 32

Nanoparticles: Applications From http: //www-ibmc. u-strasbg. fr/ict/vectorisation/nanotubes_eng. shtml http: //www. voyle. net/Extra%202005%20 Images/21 -01 -2005 -2. jpg http: //www. pharmacy. ac. uk/uploads/pics/Drug_Delivery_Polymer. jpg http: //www. bridgat. com/files/anti. UV_agent. jpg http: //ic. tweakimg. net/ext/i/imagenormal/1265797330. jpeg 33
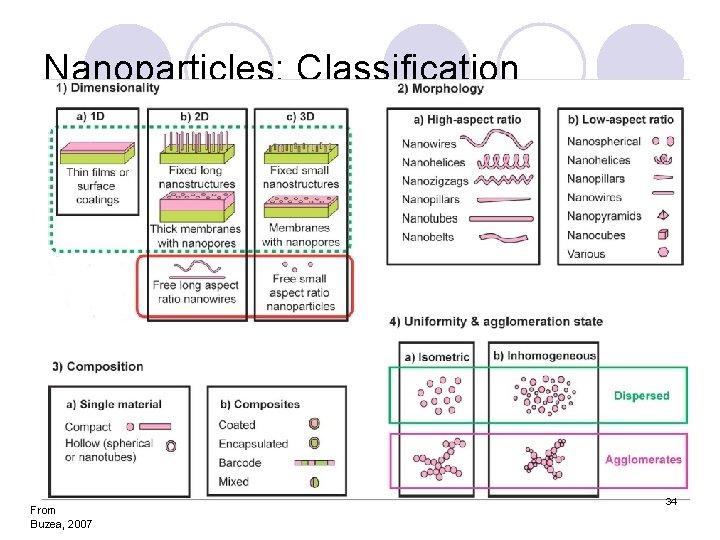
Nanoparticles: Classification From Buzea, 2007 34

Quantum dots l Inorganic semiconductor nanocrystals. l Have many possible applications due to their optical properties. ¡ In solar cells, biosensors and bioimaging, LEDs, quantum computing
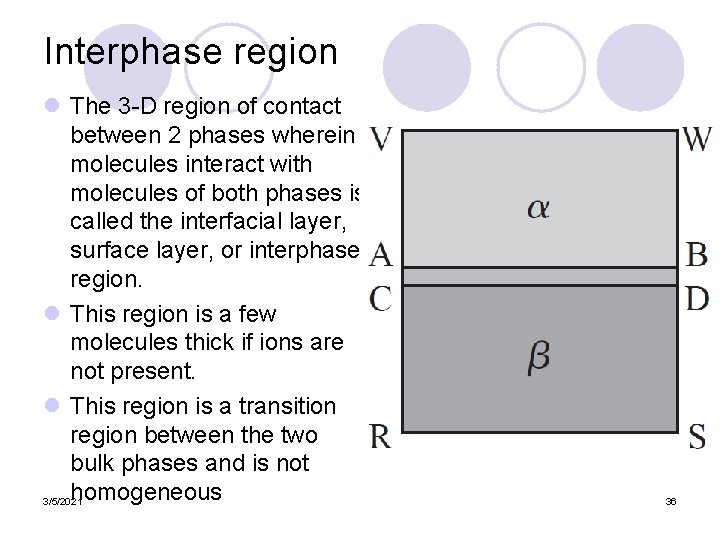
Interphase region l The 3 -D region of contact between 2 phases wherein molecules interact with molecules of both phases is called the interfacial layer, surface layer, or interphase region. l This region is a few molecules thick if ions are not present. l This region is a transition region between the two bulk phases and is not homogeneous 3/5/2021 36

Colloids ¡A colloid or disperse phase is a dispersion of small particles of one material in another. ¡This means typically around less than 500 nm particles. ¡In general, colloidal particles are aggregates of numerous atoms or molecules. ¡Kinds of colloids l. Sol – solid in liquid l. Aerosol – liquid in gas l. Emulsion – liquid in liquid 3/5/2021 37
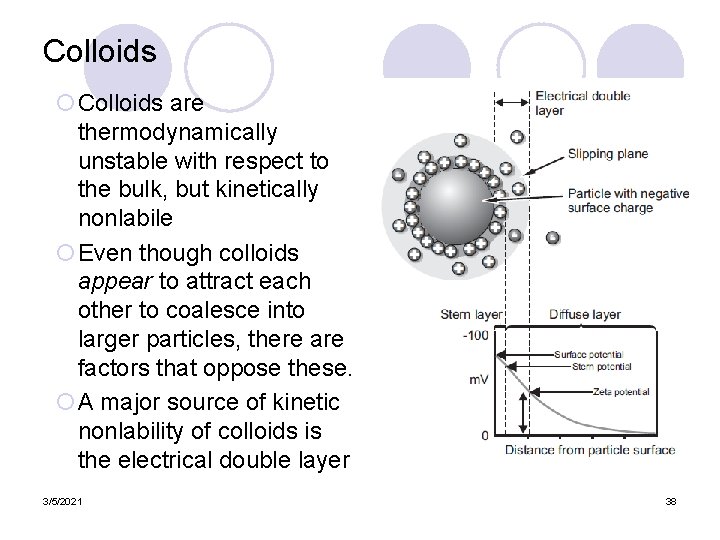
Colloids ¡Colloids are thermodynamically unstable with respect to the bulk, but kinetically nonlabile ¡Even though colloids appear to attract each other to coalesce into larger particles, there are factors that oppose these. ¡A major source of kinetic nonlability of colloids is the electrical double layer 3/5/2021 38
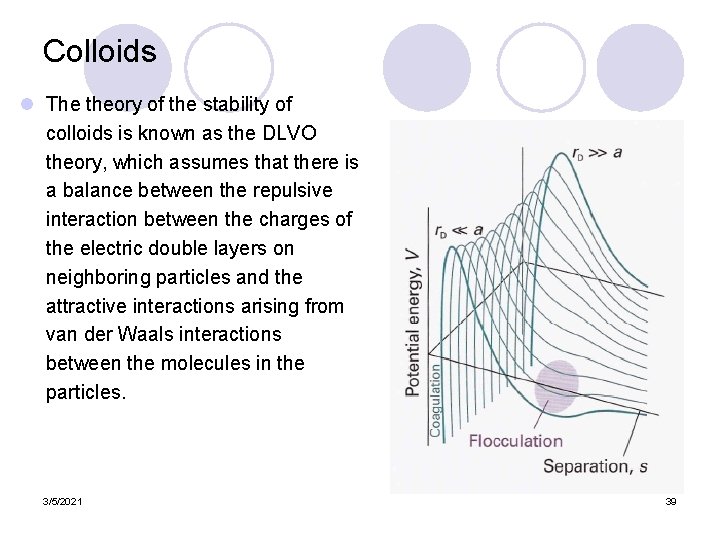
Colloids l The theory of the stability of colloids is known as the DLVO theory, which assumes that there is a balance between the repulsive interaction between the charges of the electric double layers on neighboring particles and the attractive interactions arising from van der Waals interactions between the molecules in the particles. 3/5/2021 39

l The Gibbs phase rule allows phase diagrams to be understood. l The Clausius and Clausius–Clapeyron equations govern the curves in phase diagrams. l Thermodynamics allows analysis of the stability of phases in systems. l Surface effects must be included in a complete thermodynamic treatment, but are usually negligible. 3/5/2021 40

Chemical Equilibrium 1. The principles of thermodynamics determine the state of chemical equilibrium for any reaction. 2. The equilibrium constant expression of elementary chemistry is equal to a constant at constant temperature when it is expressed in terms of activities. 3. The principle of Le Chatelier can predict how a chemical system at equilibrium responds to changes in temperature, pressure, or amounts of substances. 4. The coupling of biochemical reactions can be understood through thermodynamics and the use of postulated mechanisms. 3/5/2021 41

Zerg at chemical equilibrium. 3/5/2021 Zumdahl Chapter 2 42

3/5/2021 43

At constant T and P… ξ 3/5/2021 = extent of reaction 44

We now have the expression of G for our reaction system as a function of the extent of reaction at constant T and P 3/5/2021 45

Want equilibrium? Join the Marines! 3/5/2021 46

3/5/2021 47

l Where activity can be defined as: l The activity is a dimensionless quantity that is equal to unity if the substance is in its standard state. 3/5/2021 48
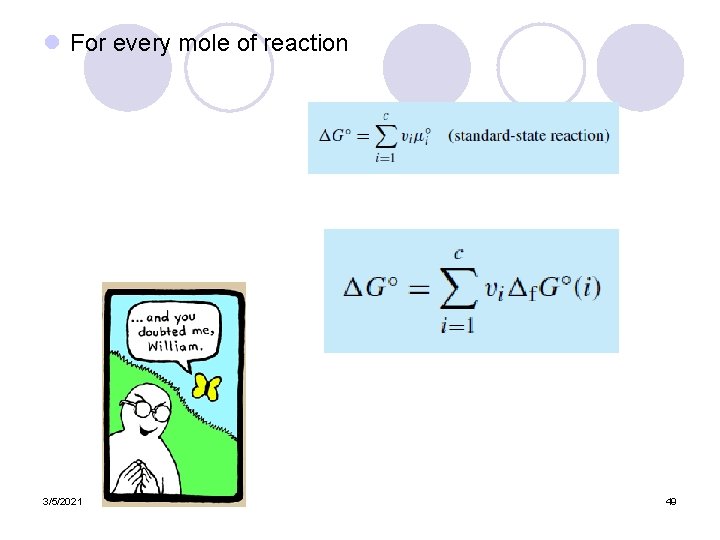
l For every mole of reaction 3/5/2021 49

Gibbs Energy Change at a definite composition l Q is called the activity quotient. The factors for the reactants have negative exponents. 3/5/2021 50
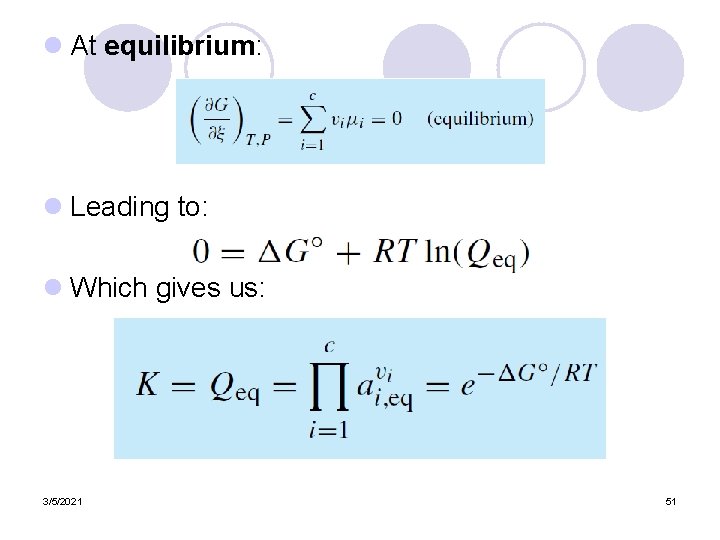
l At equilibrium: l Leading to: l Which gives us: 3/5/2021 51
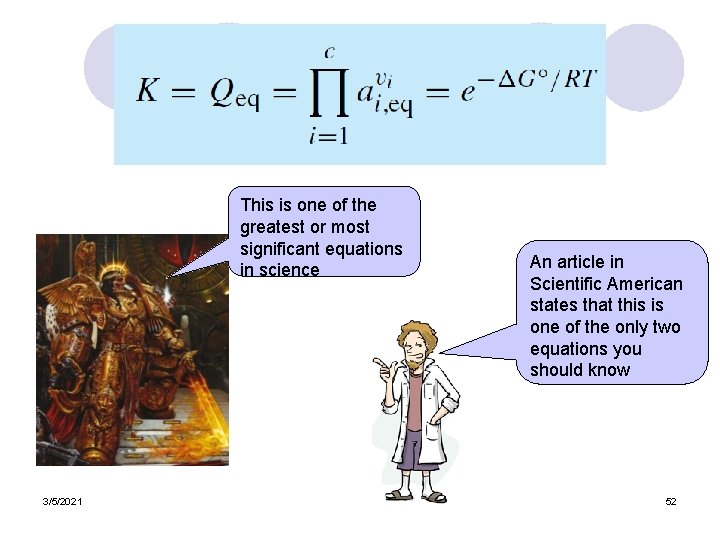
This is one of the greatest or most significant equations in science 3/5/2021 An article in Scientific American states that this is one of the only two equations you should know 52

Reactions involving gases and pure solids or liquids l We know that l So for an ideal gas reaction: l We define this as the pressure equilibrium constant, or Kp 3/5/2021 53

Example: l Consider the reaction: 0 = 2 NO 2(g) − N 2 O 4(g) ¡ Calculate the value of ΔG◦ at 298. 15 K. 4. 729 k. J/mol ¡ Calculate the value of K at 298. 15 K. 0. 148 ¡ Calculate the equilibrium pressure of a system that initially consists of 1. 000 mol of N 2 O 4 and that is confined in a fixed volume of 24. 46 L at 298. 15 K. Assume ideal gases. 1. 174 atm ¡ Δf. G◦ (NO 2) = 51. 258 k. J/mol ¡ Δf. G◦ (N 2 O 4) = 97. 787 k. J/mol l HINT: It is always the case that when a quadratic equation is solved in a chemistry problem, only one of the two roots corresponds to a physically 3/5/2021 possible situation. 54
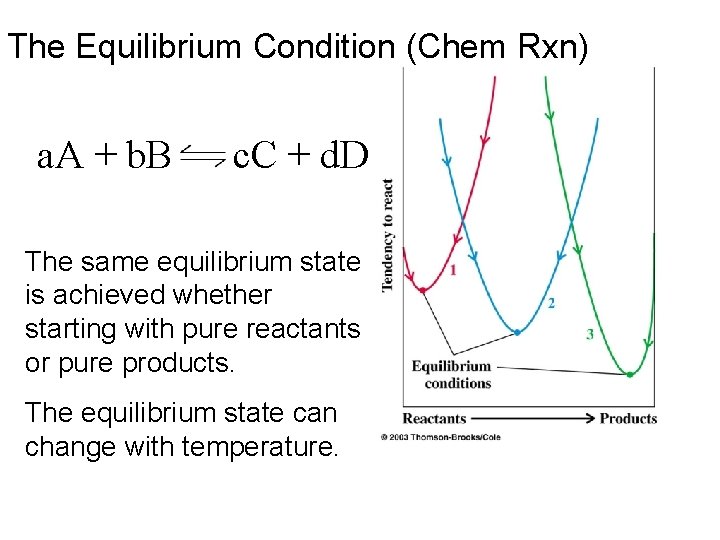
The Equilibrium Condition (Chem Rxn) a. A + b. B c. C + d. D The same equilibrium state is achieved whether starting with pure reactants or pure products. The equilibrium state can change with temperature.
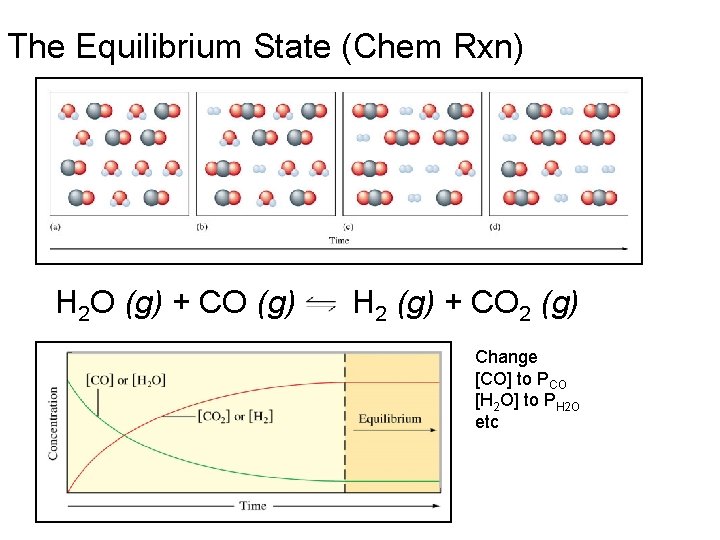
The Equilibrium State (Chem Rxn) H 2 O (g) + CO (g) H 2 (g) + CO 2 (g) Change [CO] to PCO [H 2 O] to PH 2 O etc

Chemical Reactions and Equilibrium As the equilibrium state is approached, the forward and backward rates of reaction approach equality. At equilibrium the rates are equal, and no further net change occurs in the partial pressures of reactants or products. Fundamental characteristics of equilibrium states: 1. No macroscopic evidence of change. 2. Reached through spontaneous processes. 3. Show a dynamic balance of forward and backward processes. 4. Same regardless of the direction from which they are approached. 5. No change over time.
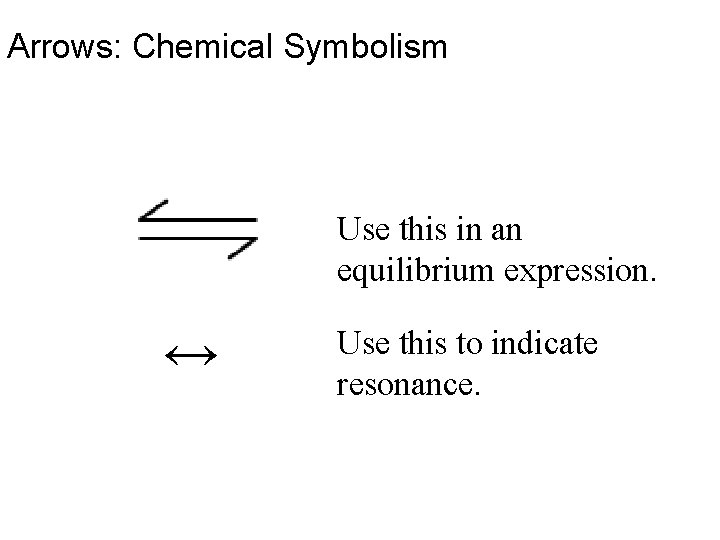
Arrows: Chemical Symbolism Use this in an equilibrium expression. ↔ Use this to indicate resonance.

Chemical Reactions and Equilibrium The equilibrium condition for every reaction can be described in a single equation in which a number, the equilibrium constant (K) of the reaction, equals an equilibrium expression, a function of properties of the reactants and products. H 2 O(l) H 2 O(g) @ 25 o. C K = 0. 03126 H 2 O(l) H 2 O(g) @ 30 o. C K = 0. 04187 Temperature (o. C) 15. 0 17. 0 19. 0 21. 0 23. 0 25. 0 30. 0 50. 0 Vapor Pressure (atm) 0. 01683 0. 01912 0. 02168 0. 02454 0. 02772 0. 03126 0. 04187 0. 1217

Law of Mass Action (1) Partial pressures and concentrations of products appear in the numerator and those of the reactants in the denominator. Each is raised to a power equal to its coefficient in the balanced chemical equation. a. A + b. B d. D c. C +

Law of Mass Action (2) 1. Gases enter equilibrium expressions as partial pressures, in atmospheres. E. g. , PCO 2 2. Dissolved species enter as concentrations, in molarity (M) moles per liter. E. g. , [Na+] 3. Pure solids and pure liquids are represented in equilibrium expressions by the number 1 (unity); a solvent taking part in a chemical reaction is represented by unity, provided that the solution is dilute. E. g. , I 2(s) ↔ I 2(aq) [I 2 (aq) ] = K

Activities The concept of Activity (i-th component) = ai = Pi / P reference H 2 O (l) H 2 O (g) Kp = P H 2 O PH 2 O =K @ 25 o. C Kp = 0. 03126 atm Pref is numerically equal to 1 Pref K = 0. 03126 The convention is to express all pressures in atmospheres and to omit factors of Pref because their value is unity. An equilibrium constant K is a pure number.
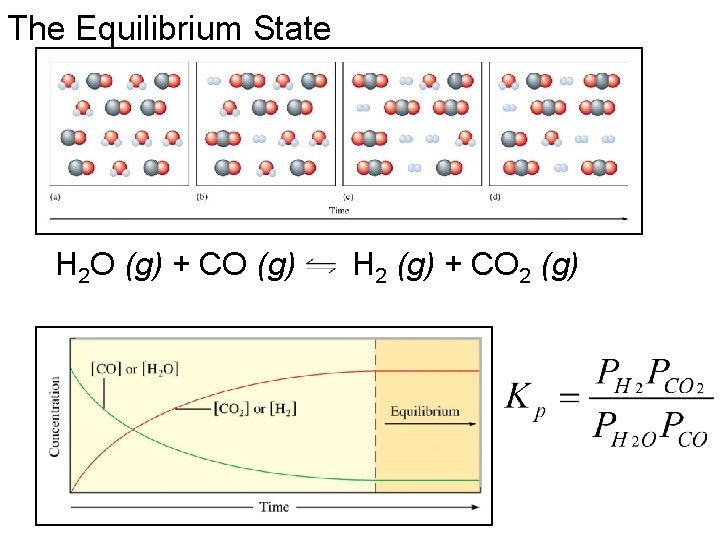
The Equilibrium State H 2 O (g) + CO (g) H 2 (g) + CO 2 (g)

The Equilibrium Expressions a. A + b. B d. D c. C + In a chemical reaction in which a moles of species A and b moles of species B react to form c moles of species C and d moles of species D, The partial pressures at equilibrium are related through K = Pc. CPd. D/Pa. APb. B
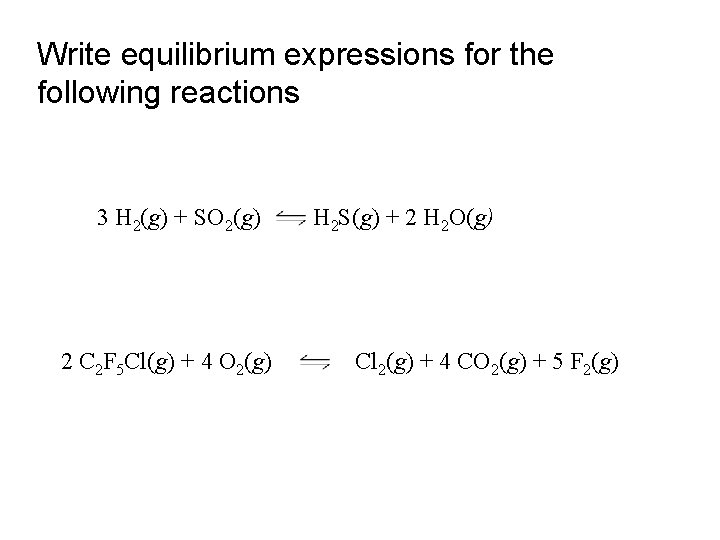
Write equilibrium expressions for the following reactions 3 H 2(g) + SO 2(g) 2 C 2 F 5 Cl(g) + 4 O 2(g) H 2 S(g) + 2 H 2 O(g) Cl 2(g) + 4 CO 2(g) + 5 F 2(g)
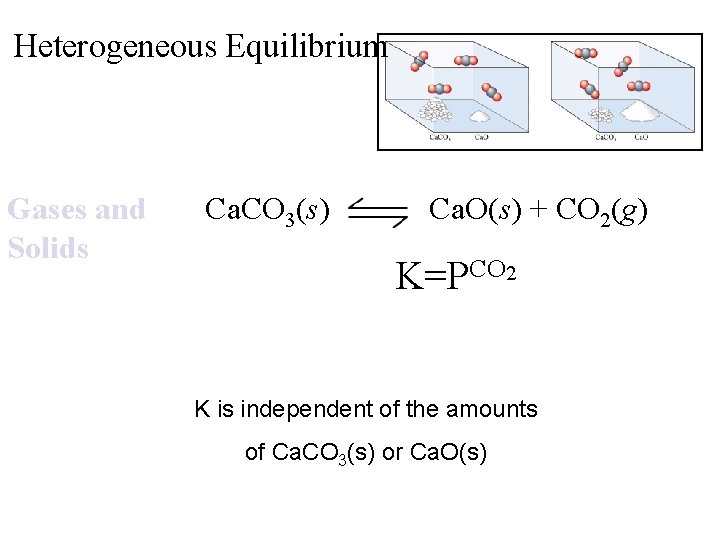
Heterogeneous Equilibrium Gases and Solids Ca. CO 3(s) Ca. O(s) + CO 2(g) K=PCO 2 K is independent of the amounts of Ca. CO 3(s) or Ca. O(s)

Heterogeneous Equilibrium Liquids H 2 O(l) H 2 O(g) K=PH 2 O Solutions I 2(s) I 2(aq) K=[I 2]

Relationships Among the K’s of Related Reactions #1: The equilibrium constant for a reverse reaction is always the reciprocal of the equilibrium constant for the corresponding forward reaction. a. A + b. B #1 #2 c. C + d. D 2 H 2 (g) + O 2 (g) 2 H 2 O (g) versus c. C + d. D 2 (P ) H O 2 2 H 2 O (g) (PH 2)2(PO 2) = K 1 2 H 2 (g) + O 2 (g) (PH 2)2(PO 2) (PH 2 O)2 = K 2 a. A + b. B K 1 = 1/K 2

Relationships Among the K’s of Related Reactions # 2: When the coefficients in a balanced chemical equation are all multiplied by a constant factor, the corresponding equilibrium constant is raised to a power equal to that factor. #1 #3 2 H 2 (g) + O 2 (g) H 2 (g) + ½ O 2 (g) (PH 2 O) (PH 2)(PO 2)½ 2 H 2 O (g) Rxn 1 H 2 O (g) = K 3 (PH 2 O)2 (PH 2)2(PO 2) = K 1 Rxn 3 = Rxn 1 times 1/2 K 3 = K 1½

Relationships Among the K’s of Related Reactions # 3: when chemical equations are added to give a new equation, their equilibrium constants are multiplied to give the equilibrium constant associated with the new equation. 2 Br. Cl (g) ↔ Br 2 (g) + Cl 2 (g) (PBr 2)(PCl 2) (PBr. Cl wrong arrow Br 2 (g) + I 2 (g) ↔ 2 IBr (g) wrong arrow )2 = K 1 = 0. 45 @ 25 o. C (PIBr)2 = K 2 = 0. 051 @ 25 o. C (PBr 2) (PI 2) 2 Br. Cl (g) + I 2 (g) ↔ 2 IBr (g) + Cl 2(g) wrong arrow (PBr 2)(PCl 2) (PIBr)2 (PBr. Cl)2 X (PBr 2) (PI 2) = K 1 K 2 = K 3 = K 1 K 2 = (0. 45)(0. 051) =0. 023 @ 25 o. C

Calculating Equilibrium Constants Consider the equilibrium 4 NO 2(g)↔ 2 N 2 O(g) + 3 O 2(g) The three gases are introduced into a container at partial pressures of 3. 6 atm (for NO 2), 5. 1 atm (for N 2 O), and 8. 0 atm (for O 2) and react to reach equilibrium at a fixed temperature. The equilibrium partial pressure of the NO 2 is measured to be 2. 4 atm. Calculate the equilibrium constant of the reaction at this temperature, assuming that no competing reactions occur. 4 NO 2(g) ↔ 2 N 2 O(g) + 3 O 2(g) initial partial pressure (atm) change in partial pressure (atm) equilibrium partial pressure (atm)

Calculate the equilibrium constant of the reaction at this temperature, assuming that no competing reactions occur. 4 NO 2(g) ↔ 2 N 2 O(g) + 3 O 2(g) initial partial pressure (atm) 3. 6 5. 1 8. 0 change in partial pressure (atm) – 4 x +2 x +3 x equilibrium partial pressure (atm) 2. 4 5. 1 + 2 x 8. 0 + 3 x 5. 1 + 2(0. 3 atm) = 5. 7 atm N 2 O 3. 6 – 4 x = 2. 4 atm NO 2; x = 0. 3 atm 8. 0 + 3(0. 3 atm) = 8. 9 atm O 2 (PN 2 O)2(PO 2)3 K= = (PNO 2)4

The compound Ge. WO 4(g) forms at high temperature in the reaction 2 Ge. O (g) + W 2 O 6(g) ↔ 2 Ge. WO 4(g) Some Ge. O (g) and W 2 O 6 (g) are mixed. Before they start to react, their partial pressures both equal 1. 000 atm. After their reaction at constant temperature and volume, the equilibrium partial pressure of Ge. WO 4(g) is 0. 980 atm. Assuming that this is the only reaction that takes place, (a) determine the equilibrium partial pressures of Ge. O and W 2 O 6, and (b) determine the equilibrium constant for the reaction. initial partial pressure (atm) 2 Ge. O (g) + W 2 O 6 (g) ↔ 2 Ge. WO 4(g) 1. 000 0 change in partial pressure (atm) equilibrium partial pressure (atm) – 2 x –x +2 x

(a) determine the equilibrium partial pressures of Ge. O and W 2 O 6, and (b) determine the equilibrium constant for the reaction. initial partial pressure (atm) change in partial pressure (atm) 2 Ge. O(g) + W 2 O 6(g) ↔ 2 Ge. WO 4(g) 1. 000 0 – 2 x equilibrium partial pressure (atm) 1. 000 – 2 x –x 1. 000 – x +2 x 0. 980 0 + 2 x = 0. 980 atm Ge. WO 4; 1. 000 – 2(0. 490) = 0. 020 atm Ge. O x = 0. 490 atm 1. 000 – 0. 490 = 0. 510 atm W 2 O 6 (PGe. WO 4)2 K = (P )2(P = ) Ge. O W 2 O 6

l Skip Solving quadratic equations l Will utilize approximation method ¡Systems that have small equilibrium constants. ¡Assume “x” (the change in concentration) is small (less than 5%) of the initial concentration.

A vessel holds pure CO (g) at a pressure of 1. 282 atm and a temperature of 354 K. A quantity of nickel is added, and the partial pressure of CO (g) drops to an equilibrium value of 0. 709 atm because of the reaction Ni (s) + 4 CO (g) ↔ Ni(CO)4 (g) Compute the equilibrium constant for this reaction at 354 K. Ni (s) + 4 CO (g) ↔ Ni(CO)4 (g) Construct an “ICE” table initial partial pressure (atm) change in partial pressure (atm) equilibrium partial pressure (atm) At equil. Pco= P CO (atm) P Ni(CO)4 (atm) 1. 282 -4 x 0. 709 0 +1 x x x = PNi(CO)4 =

Equilibrium Calculations At a particular temperature, K = 2. 0 x 10 -6 mol/L for the reaction 2 CO 2 (g) 2 CO (g) + O 2 (g) If 2. 0 mol CO 2 is initially placed into a 5. 0 -L vessel, calculate the equilibrium concentrations of all species. 2 CO 2 (g) initial partial pressure (mol/L) 0. 4 change in partial pressure (mol/L) – 2 x equilibrium partial pressure (mol/L) 0. 4 -2 x 2 CO (g) + O 2 (g) 0 +2 x 2 x 0 +1 x 1 x

At a particular temperature, K = 2. 0 x 10 -6 mol/L for the reaction 2 CO 2 (g) 2 CO (g) + O 2 (g) If 2. 0 mol CO 2 is initially placed into a 5. 0 -L vessel, calculate the equilibrium concentrations of all species. 2 CO 2 (g) initial partial pressure (mol/L) 0. 4 change in partial pressure (mol/L) – 2 x equilibrium partial pressure (mol/L) 0. 4 -2 x 2 CO (g) + O 2 (g) 0 +2 x 2 x 0 +1 x 1 x

At a particular temperature, K = 2. 0 x 10 -6 mol/L for the reaction 2 CO 2 (g) 2 CO (g) + O 2 (g) If 2. 0 mol CO 2 is initially placed into a 5. 0 -L vessel, calculate the equilibrium concentrations of all species. 2 CO 2 (g) initial partial pressure (mol/L) 0. 4 change in partial pressure (mol/L) – 2 x equilibrium partial pressure (mol/L) 0. 4 -2 x 2 CO (g) + O 2 (g) 0 +2 x 2 x 0 +1 x 1 x

Non-Equilibrium Conditions: The Reaction Quotient (1) wrong arrow K (the Equilibrium Constant) uses equilibrium partial pressures Q (the reaction quotient) uses prevailing partial pressures, not necessarily at equilibrium
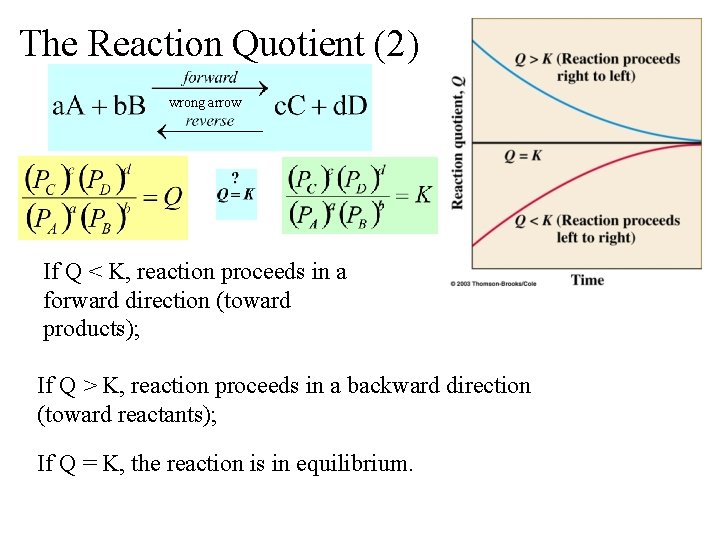
The Reaction Quotient (2) wrong arrow If Q < K, reaction proceeds in a forward direction (toward products); If Q > K, reaction proceeds in a backward direction (toward reactants); If Q = K, the reaction is in equilibrium.

The equilibrium constant for the reaction P 4(g) ↔ 2 P 2(g) is 1. 39 at 400 o. C. Suppose that 2. 75 mol of P 4(g) and 1. 08 mol of P 2(g) are mixed in a closed 25. 0 L container at 400 o. C. Compute Q(init) (the Q at the moment of mixing) and state the direction in which the reaction proceeds. K = 1. 39 @ 400 o. C; n. P 4(init) = 2. 75 mol; n. P 2(init) = 1. 08 mol PP 4(init) = n. P 4(init)RT/V = [(2. 75 mol)(0. 08206 atm L mol-1 K-1)(273. 15+400 o. C)]/(25. 0 L) = 6. 08 atm PP 2(init) = n. P 2(init)RT/V = [(1. 08 mol)(0. 08206 atm L mol-1 K-1)(273. 15+400 o. C)]/(25. 0 L) = 2. 39 atm Q=

Henri Louis Le Châtelier (1850 -1936) Highlights ¡ 1884 Le Chatelier's Principle: A system in equilibrium that is subjected to a stress reacts in a way that counteracts the stress ¡ If a chemical system at equilibrium experiences a change in concentration, temperature or total pressure the equilibrium will shift in order to minimize that change. ¡ Industrial chemist involved with industrial efficiency and labor-management relations Moments in a Life ¡ Le Chatelier was named "chevalier" (knight) of the Légion d'honneur in 1887, decoration established by Napoléon Bonaparte in 1802. Challenge my moustache? I think not!

Effects of External Stresses on Equilibria: Le Châtelier’s Principle A system in equilibrium that is subjected to a stress reacts in a way that counteracts the stress. Le Châtelier’s Principle provides a way to predict the response of an equilibrium system to an external perturbation, such as… 1. Effects of Adding or Removing Reactants or Products 2. Effects of Changing the Volume (or Pressure) of the System 3. Effects of Changing the Temperature

Effects of Adding or Removing Reactants or Products PCl 5(g) PCl 3(g) + Cl 2(g) K = 11. 5 @ 300 o. C = Q add extra PCl 5(g) add extra PCl 3(g) remove some PCl 5(g) remove some PCl 3(g) A system in equilibrium that is subjected to a stress reacts in a way that counteracts the stress. In this case adding or removing reactants or products

Effects of Changing the Volume of the System PCl 5(g) 1 mole PCl 3(g) + Cl 2(g) 1+1 = 2 moles Let’s decrease the volume of the reaction container Less room : : less amount (fewer moles) Shifts reaction to restore equilibrium Let’s increase the volume of the reaction container More room : : more amount (greater moles) Shifts reaction to restore equilibrium A system in equilibrium that is subjected to a stress reacts in a way that counteracts the stress. In this case a change in volume
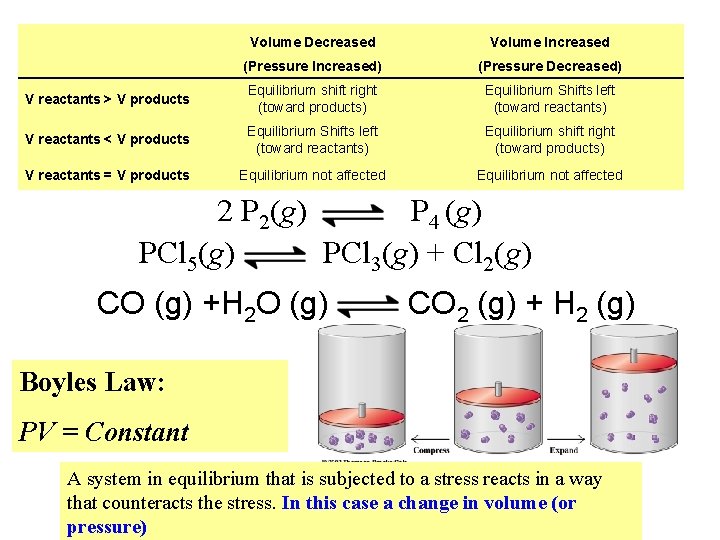
Volume Decreased Volume Increased (Pressure Increased) (Pressure Decreased) V reactants > V products Equilibrium shift right (toward products) Equilibrium Shifts left (toward reactants) V reactants < V products Equilibrium Shifts left (toward reactants) Equilibrium shift right (toward products) V reactants = V products Equilibrium not affected 2 P 2(g) P 4 (g) PCl 5(g) PCl 3(g) + Cl 2(g) CO (g) +H 2 O (g) CO 2 (g) + H 2 (g) Boyles Law: PV = Constant A system in equilibrium that is subjected to a stress reacts in a way that counteracts the stress. In this case a change in volume (or pressure)

Effects of Changing the Temperature Endothermic: heat is aborbed by a reaction Reactants + heat gives Products Exothermic: heat is liberated by a reaction Reactants gives Products + heat A system in equilibrium that is subjected to a stress reacts in a way that counteracts the stress. In this case a change in temperature

Effects of Changing the Temperature Endothermic: absorption of heat by a reaction Reactants + heat gives Products Let’s increase the temperature of the reaction, what direction does the equilibrium reaction shift Let’s decrease the temperature of the reaction

Effects of Changing the Temperature Exothermic: heat liberated by a reaction Reactants Products + heat Let’s increase the temperature of the reaction Let’s decrease the temperature of the reaction A system in equilibrium that is subjected to a stress reacts in a way that counteracts the stress. In this case a change in temperature
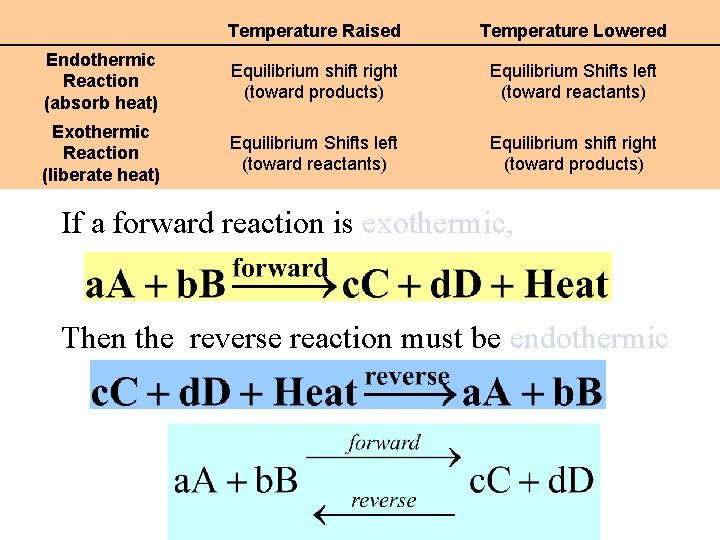
Temperature Raised Temperature Lowered Endothermic Reaction (absorb heat) Equilibrium shift right Equilibrium Shifts left (toward products) (toward reactants) Exothermic Reaction (liberate heat) Equilibrium Shifts left Equilibrium shift right (toward reactants) (toward products) If a forward reaction is exothermic, Then the reverse reaction must be endothermic

Driving Reactions to Completion/ Increasing Yield Industrial Synthesis of Ammonia (Haber) N 2 (g) + 3 H 2 (g) ↔ 2 NH 3 (g) Forward reaction is exothermic What conditions do we need to increase the yield, i. e. , produce more ammonia? Volume Decreased Volume Increased (Pressure Increased) (Pressure Decreased) V reactants > V products Equilibrium shift right (toward products) Temperature Raised Exothermic Reaction (liberate heat) Equilibrium Shifts left (toward reactants) Temperature Lowered Equilibrium Shifts left Equilibrium shift right (toward reactants) (toward products)

Problem l Hello Kitty has concocted a vile gas, βψ2(g), to combat the forces of Kerokeropi the Green. This gas is harmless, but when in contact with sunlight, it decomposes into the toxic nerve gas β 2(g) and the harmless liquid ψ(l). 2βψ2(g) β 2(g) + 4ψ(l) l At equilibrium, the partial pressures of βψ2(g) and β 2(g) are 0. 02 and 0. 87 atm at 32°C, the usual temperature in Isengard. ¡ Determine the gas equilibrium constant of the reaction. ¡ Determine the standard reaction Gibbs energy of the reaction. l In Rohan, the temperature is a cooler 27°C and when Saruman’s Uruk-Hai warriors deployed the gas there, the partial pressures ofβψ2(g) and β 2(g) are 0. 057 and 0. 76 atm at equilibrium. ¡ Determine the standard reaction Gibbs energy of the reaction when it takes place in Rohan. 93 ¡ What is the standard reaction enthalpy change of the reaction?

Thank you for listening! 3/5/2021 94
- Slides: 94