Equilibrium Equilibrium The reactions are reversible Both reactions

Equilibrium

Equilibrium • The reactions are reversible • Both reactions are occurring at the same time • The forward and reverse reaction occur at the same rate.

Conditions of Equilibrium 1) Closed system 2) Opposite reactions at the same rate 3) Started reaction with either the reactant or product 4) Temperature, pressure, concentration are kept constant. Dynamic System

Le Chatelier’s Principle • Stress – Any change in • Concentration • Temperature • Pressure • Volume • Le Chatelier’s Principle – If a stress is added to a system in equilibrium, the system counteracts that change and reaches a new state of equilibrium.

Terminology • • Shift right Products favored Forward reaction speeds up • • Shifts left Reactants favored Reverse reaction speeds up Favored = greater concentration

Changes in Concentration and Temperature • An increase in concentration or temperature cause the reaction to shift away from the change • A decrease causes the reaction to shift towards the change • Note: whichever way the arrow is pointing, the molecules increase and the others decrease.

2 SO 3(g) + heat 2 SO 2(g) + O 2(g) • • • Which way will the reaction shift if? Increase SO 3 Increase the temperature Increase SO 2 Decrease SO 3 Decrease O 2 – Do the reactants and products decrease or increase?

Changes in Pressure • Only affects gases • Increase in pressure causes a shift towards the side with less gas moles • Decrease in pressure causes a shift towards the side with more gas moles • Increase in volume causes a decrease in pressure • Decrease in volume causes an increase in pressure

N 2(g) + 3 H 2(g) 2 NH 3 (g) • Which way would the reaction shift if pressure was increased? • Would N 2, H 2, NH 3 increase or decrease? • What would happen if the volume is increased?
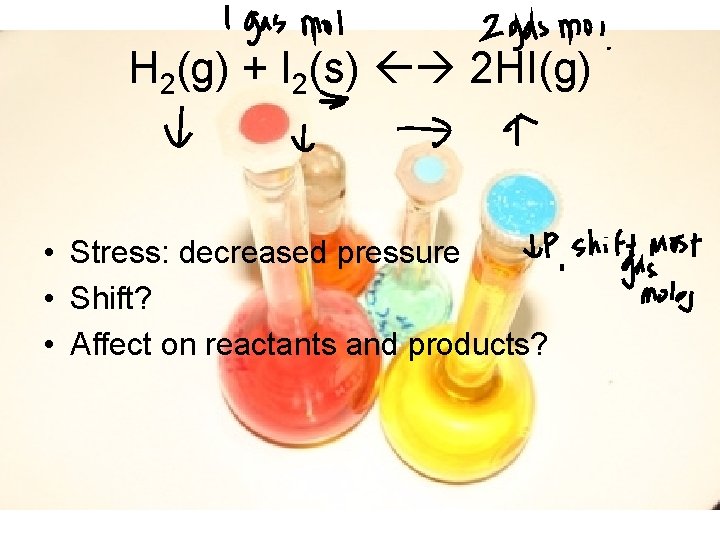
H 2(g) + I 2(s) 2 HI(g) • Stress: decreased pressure • Shift? • Affect on reactants and products?

Adding a Catalyst • Increases both the forward and reverse rates • Favors neither reaction • DOES NOT create a stress
- Slides: 11