Equilibrium Dynamic Equilibrium Rate of Forward Reaction Rate
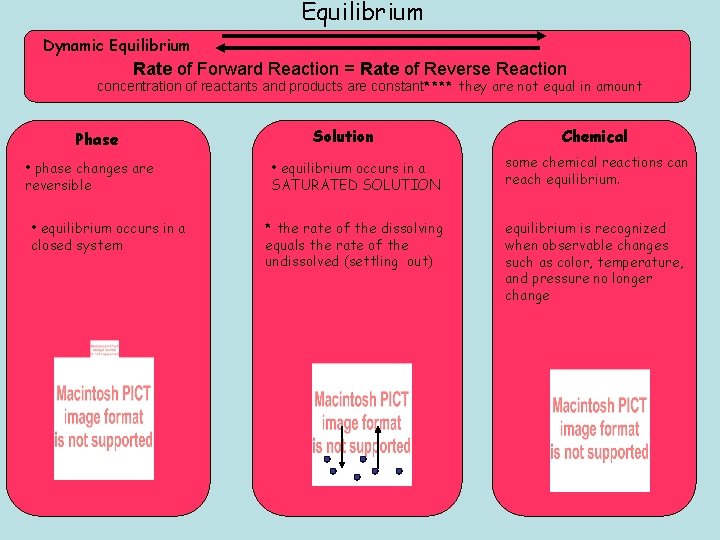
Equilibrium Dynamic Equilibrium Rate of Forward Reaction = Rate of Reverse Reaction concentration of reactants and products are constant**** they are not equal in amount Phase • phase changes are reversible • equilibrium occurs in a closed system Solution Chemical • equilibrium occurs in a SATURATED SOLUTION some chemical reactions can reach equilibrium. * the rate of the dissolving equals the rate of the undissolved (settling out) equilibrium is recognized when observable changes such as color, temperature, and pressure no longer change
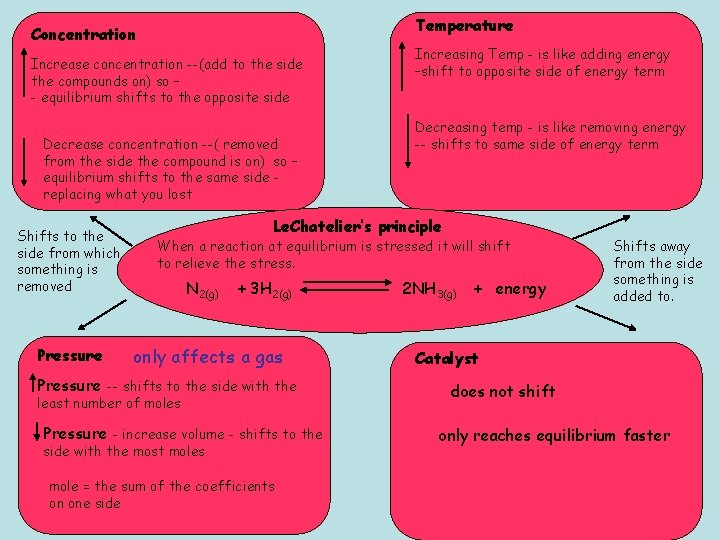
Temperature Concentration Increase concentration --(add to the side the compounds on) so – - equilibrium shifts to the opposite side Decrease concentration --( removed from the side the compound is on) so – equilibrium shifts to the same side replacing what you lost Shifts to the side from which something is removed Pressure Increasing Temp - is like adding energy –shift to opposite side of energy term Decreasing temp - is like removing energy -- shifts to same side of energy term Le. Chatelier’s principle When a reaction at equilibrium is stressed it will shift to relieve the stress. N 2(g) + 3 H 2(g) only affects a gas Pressure -- shifts to the side with the least number of moles Pressure - increase volume - shifts to the side with the most moles mole = the sum of the coefficients on one side 2 NH 3(g) + energy Shifts away from the side something is added to. Catalyst does not shift only reaches equilibrium faster

Common Ion Effect Add ion to decrease the solubility of a salt Li 2 SO 4(s) = 2 Li(aq) + + SO 42 -(aq) What would happen if I add H 2 SO 4 = 2 H+ + SO 42 Added SO 42 - so increasing concentration on the right of ion Shift the equation to left Causes Li 2 SO 4 to precipitate (become less soluble)
- Slides: 3