Equilibrium Constant The relationship between the concentrations of

Equilibrium Constant The relationship between the concentrations of the reactants and products at equilibrium for the following reaction: a. A+b. B⇌c. C+d. D can be expressed as such: Kc = [C]c[D]d [A]a[B]b This is the equilibrium-constant expression The [X] refers to the concentration (in molarity) of the reactants and products once at equilibrium The exponents refer to the coefficients
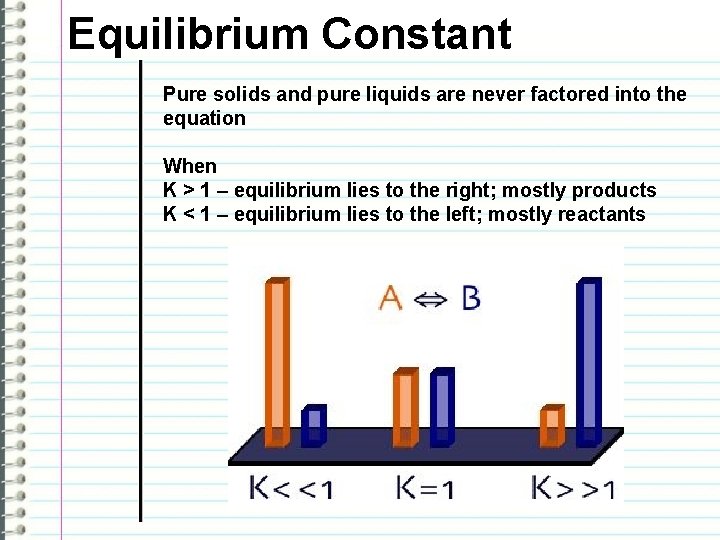
Equilibrium Constant Pure solids and pure liquids are never factored into the equation When K > 1 – equilibrium lies to the right; mostly products K < 1 – equilibrium lies to the left; mostly reactants

Reaction Quotient Once the equilibrium constant has been established, hypothetical concentrations can be introduced into the equilibrium-constant expression to obtain a reaction quotient which can then be compared to the equilibrium constant to determine how the reaction will proceed. For a given reaction: a. A+b. B⇌c. C+d. D The reaction quotient is the same as the equilibrium constant: Q = [C]c [D]d [A]a [B]b

Reaction Quotient When Q = K – the system is at equilibrium Q > K – concentration of products is larger than reactants, the reaction moves to the left to reach equilibrium Q < K – concentration of products is smaller than reactants, the reaction moves the right to reach equilibrium

Reaction Quotient
- Slides: 5