Equilibrium Calculations Lesson 7 How can we describe

Equilibrium Calculations Lesson 7
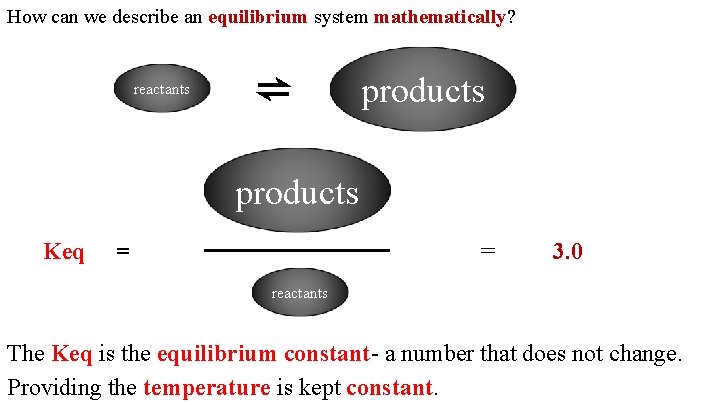
How can we describe an equilibrium system mathematically? reactants ⇌ products Keq = = 3. 0 reactants The Keq is the equilibrium constant- a number that does not change. Providing the temperature is kept constant.

Equilibrium Calculations An equilibrium system, at any given temperature, can be described by an equilibrium expression and equilibrium constant. a. A + b. B Keq ⇌ = = c. C + d. D Products Reactants [C]c[D]d [A]a[B]b Equilibrium Constant- a number Expression- mathematical equation (aq) and (g) are included! (l) and (s) are not-constant concentration!
![1. At equilibrium at 25 o. C, [SO 3] = 0. 200 M. [H 1. At equilibrium at 25 o. C, [SO 3] = 0. 200 M. [H](http://slidetodoc.com/presentation_image_h2/9bd5c0919b0018051d68ddc149b315c3/image-4.jpg)
1. At equilibrium at 25 o. C, [SO 3] = 0. 200 M. [H 2 O] = 0. 480 M, and [H 2 SO 4] = 24 M. Calculate the Keq. No ICE SO 3(g) Keq + = = H 2 O(g) ⇌ 1 [SO 3] [H 2 O] H 2 SO 4(l) don’t count (l)! Use 1 1 (0. 200)(0. 480) = 10. 4 The Keq has no units but concentration units that go in the expression must be M!

2. 0. 500 mole PCl 5, 0. 40 mole H 2 O, 0. 200 mole HCl, and 0. 400 mole POCl 3 are found in a 2. 0 L container at equilibrium at 125 o. C. Calculate the Keq. No ICE PCl 5(s) + H 2 O(g) ⇌ 2 HCl(g) + POCl 3(g) = 0. 200 moles 2. 0 L [HCl] [POCl 3] = 0. 400 moles = 0. 10 M = 0. 20 M Keq = 2. 0 L [H 2 O] = 0. 40 moles 2. 0 L [HCl]2[POCl 3] [H 2 O] = 0. 20 M Keq = [0. 10]2[0. 20] Keq = 0. 010

3. If 0. 600 mole of SO 3 and 0. 0200 mole of SO 2 are found in a 2. 00 L container at equilibrium at 25 o. C. Calculate the [O 2]. 2 SO 2(g) + O 2(g) ⇌ 2 SO 3(g) [SO 3] = 0. 600 mole/2. 00 L = 0. 300 M [SO 2] = 0. 0200 mole/2. 00 L = 0. 0100 M Keq = 798 Keq = [SO 3]2 [SO 2]2[O 2] 798 1 = (0. 3)2 = [O 2] = (0. 300)2 (0. 0100)2[O 2] 798(0. 01)2[O 2] (0. 3)2 798(0. 01)2 = 1. 13 M

4. When 0. 800 moles of SO 2 and 0. 800 moles of O 2 are placed into a 2. 00 L container and allowed to reach equilibrium, the equilibrium [SO 3] is to be 0. 300 M. Calculate the Keq value. Implies initial and not equilibrium concentrations 2 SO 2 I (g) 0. 400 M + O 2 ⇋ (g) 0. 400 M C -0. 300 M -0. 150 M E 0. 100 M 0. 250 M - ICE x 2/ 2 2 SO 3 (g) 0 x 1/2 +0. 300 M Equilibrium concentrations go in the equilibrium equation! Keq = [SO 3]2 [SO 2]2[O 2] = (0. 3)2 (0. 1)2(0. 25) = 36. 0
- Slides: 7