Equilibrium Asst Prof Miss Vidya K Patil Department

Equilibrium Asst. Prof. Miss. Vidya K. Patil. Department of Chemistry KCES’s Post Graduate College of Science Technology and Research, Jalgaon

Equilibrium • Equilibrium Constant, K (or Keq) describes conditions AT equilibrium Ca. CO 3(calcite) + H+ Ca 2+ + HCO 3 -

Activity • Sometimes called ‘effective concentration’, which is misleading and reflects a poor understanding of the property… • Think of more of the effect the rest of a solution has on how easily two ions come together. .

Activity • For solids or liquid solutions: ai=Xigi • For gases: ai=Pigi = fi Xi=mole fraction of component i Pi = partial pressure of component i mi = molal concentration of component i • For aqueous solutions: ai=migi

Activity Coefficients • Where do they come from? ? • The standard state for dissolved ions is actually an infinitely dilute solution… • Activity of phases - gases, minerals, and bulk liquids (H 2 O) are usually pretty close to 1 in waters • Dissolved molecules/ ions have activity coefficients that change with concentration (ions are curved lines relating concentration and activity coefficients, molecules usually more linear relation)

Application to ions in solution • Ions in solutions are obviously nonideal mixtures! a i = g i mi • The activity coefficient, gi, is found via some empirical foundations • Dependent on the other ions in water…

Dissolved species gi • First must define the ionic strength (I) of the solution the ion is in: Where mi is the molar concentration of species i and zi is the charge of species I

Activity Coefficients • Debye-Huckel approximation (valid for I: • Where A and B are constants (depending on T, see table 10. 3 in your book), and a is a measure of the effective diameter of the ion (table 10. 4)
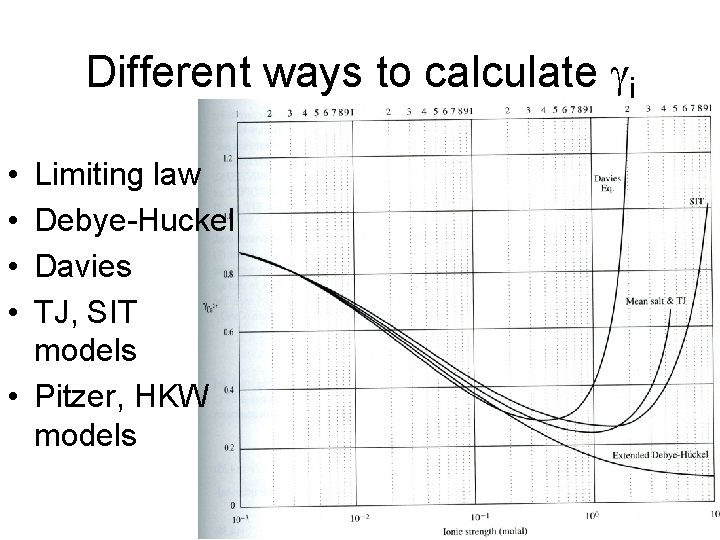
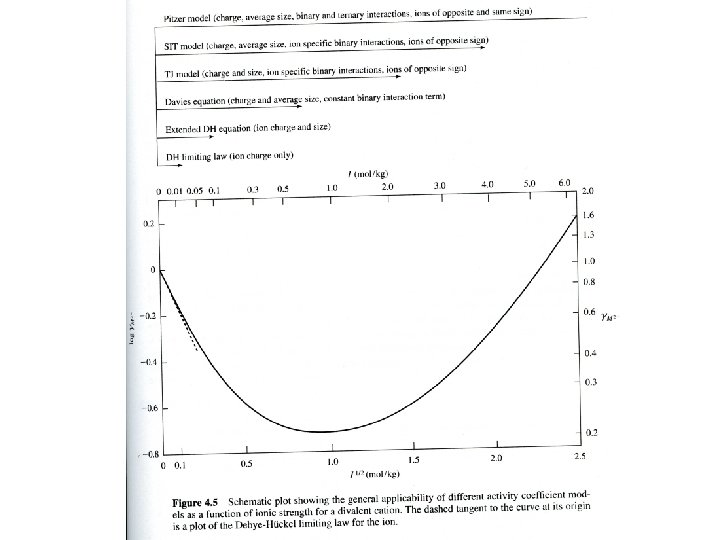
Different ways to calculate gi • • Limiting law Debye-Huckel Davies TJ, SIT models • Pitzer, HKW models

Neutral species • Setchnow equation: • Logan=ks. I For activity coefficient (see table 4 -2 for selected coefficients)
- Slides: 11