Equilibrium AP Chemistry What is equilibrium n Position

Equilibrium AP Chemistry

What is equilibrium? n Position of equilibrium depends upon n n Initial concentrations Relative energy of reactants & products
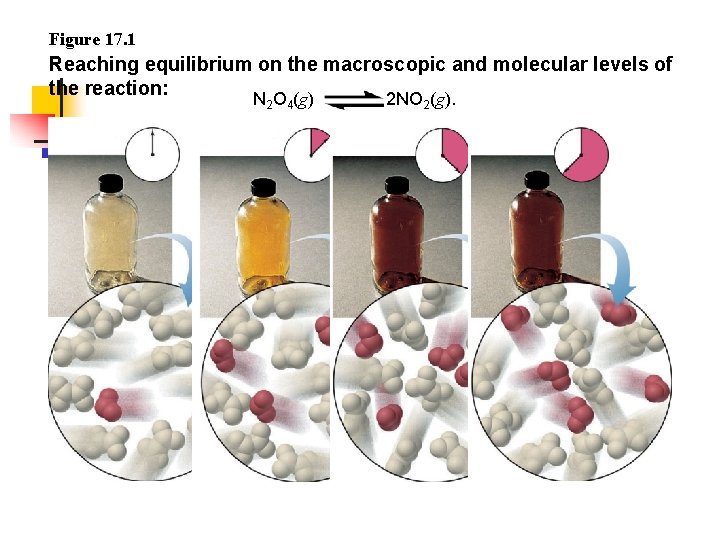
Figure 17. 1 Reaching equilibrium on the macroscopic and molecular levels of the reaction: N O (g) 2 NO (g). 2 4 2
![If rateforward = ratereverse then kforward[reactants]m = kreverse[products]n kforward kreverse = [products]n [reactants]m = If rateforward = ratereverse then kforward[reactants]m = kreverse[products]n kforward kreverse = [products]n [reactants]m =](http://slidetodoc.com/presentation_image/4569106f16b5542e8935cbcce95410d3/image-4.jpg)
If rateforward = ratereverse then kforward[reactants]m = kreverse[products]n kforward kreverse = [products]n [reactants]m = K the equilibrium constant This is also known as the LAW OF MASS ACTION.

Equilibrium expression n Reaction quotient, law of mass action 4 NH 3(g) +7 O 2(g) 4 NO(g) + 6 H 2 O(g) K for a system is constant at a particular temperature.

Homogeneous equilibria n All reagents are in same phase

Heterogeneous equilibria n Different states of matter Ca. CO 3(s) Ca. O(s) + CO 2(g) n K= n Reverse reaction? n n Data shows K does not depend on amounts of pure solids or liquids present in heterogeneous equilibria systems.

Equilibrium Constant n n n K very large= reaction proceeds far towards completion, products favored K ≈1; reactants almost equal to products K very small; hardly any products are formed; reactants are favored
![Type 1 Problems Given: 1] initial concentrations of reactants or products 2] one equilibrium Type 1 Problems Given: 1] initial concentrations of reactants or products 2] one equilibrium](http://slidetodoc.com/presentation_image/4569106f16b5542e8935cbcce95410d3/image-9.jpg)
Type 1 Problems Given: 1] initial concentrations of reactants or products 2] one equilibrium concentration Solve for K.
![For the following reaction, reactants = [0. 100 M] initial concentration. At equilibrium [I For the following reaction, reactants = [0. 100 M] initial concentration. At equilibrium [I](http://slidetodoc.com/presentation_image/4569106f16b5542e8935cbcce95410d3/image-10.jpg)
For the following reaction, reactants = [0. 100 M] initial concentration. At equilibrium [I 2] = 0. 020 M Find K n H 2(g) + I 2(g) 2 HI(g)

Sample Problem 17. 7 PROBLEM: Calculating Kc from Concentration Data In order to study hydrogen halide decomposition, a researcher fills an evacuated 2. 00 -L flask with 0. 200 mol of HI gas and allows the reaction to proceed at 453 o. C. 2 HI(g) H 2(g) + I 2(g) At equilibrium, [HI] = 0. 078 M. Calculate Kc?

Sample Problem 17. 8 PROBLEM: Determining Equilibrium Concentrations from Kc In a study concerning the conversion of methane to other fuels, a chemical engineer mixes gaseous CH 4 and H 2 O in a 0. 32 -L flask at 1200 K. At equilibrium the flask contains 0. 26 mol of CO, 0. 091 mol of H 2, and 0. 041 mol of CH 4. What is the [H 2 O] at equilibrium? Kc = 0. 26 for the equation CH 4(g) + H 2 O(g) CO(g) + 3 H 2(g) Qc = [CO][H 2]3 [CH 4][H 2 O]

Sample Problem 17. 9 PROBLEM: Determining Equilibrium Concentrations from Initial Concentrations and Kc Fuel engineers use the extent of the change from CO and H 2 O to CO 2 and H 2 to regulate the proportions of synthetic fuel mixtures. If 0. 250 mol of CO and 0. 250 mol of H 2 O are placed in a 125 -m. L flask at 900 K, what is the composition of the equilibrium mixture? At this temperature, Kc is 1. 56 for the equation CO(g) + H 2 O(g) concentration initial change equilibrium CO(g) + H 2 O(g) CO 2(g) + H 2(g)
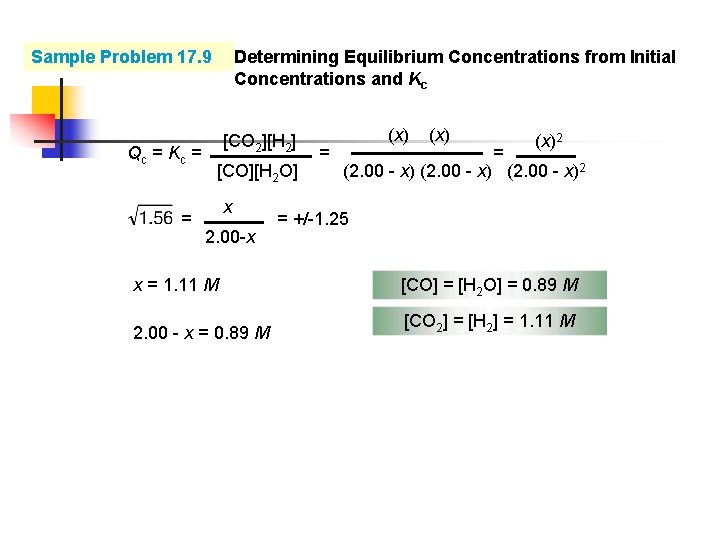
Sample Problem 17. 9 Qc = Kc = = Determining Equilibrium Concentrations from Initial Concentrations and Kc [CO 2][H 2] [CO][H 2 O] x 2. 00 -x = (x)2 (2. 00 - x)2 = +/-1. 25 x = 1. 11 M [CO] = [H 2 O] = 0. 89 M 2. 00 - x = 0. 89 M [CO 2] = [H 2] = 1. 11 M

Sample Problem 17. 10 Calculating Equilibrium Concentrations with a Simplifying Assumption PROBLEM: Phosgene is a potent chemical warfare agent that is now outlawed by international agreement. It decomposes by the reaction COCl 2(g) CO(g) + Cl 2(g) Kc = 8. 3 x 10 -4 (at 360 o. C) Calculate [CO], [Cl 2], and [COCl 2] when the following amounts of phosgene decompose and reach equilibrium in a 10. 0 -L flask. (a) 5. 00 mol COCl 2 (b) 0. 100 mol COCl 2
![Sample Problem 17. 10 Calculating Equilibrium Concentrations with a Simplifying Assumption Kc = [CO][Cl Sample Problem 17. 10 Calculating Equilibrium Concentrations with a Simplifying Assumption Kc = [CO][Cl](http://slidetodoc.com/presentation_image/4569106f16b5542e8935cbcce95410d3/image-16.jpg)
Sample Problem 17. 10 Calculating Equilibrium Concentrations with a Simplifying Assumption Kc = [CO][Cl 2] [COCl 2]

Type 2 Problems Given: 1. initial concentrations 2. K value Since most problems have initial concentrations of both reactants and products, we need to use Q to find direction of shift.
![Reaction Quotient Qc = [products initial]x n [reactants initial]y n n Q = K; Reaction Quotient Qc = [products initial]x n [reactants initial]y n n Q = K;](http://slidetodoc.com/presentation_image/4569106f16b5542e8935cbcce95410d3/image-18.jpg)
Reaction Quotient Qc = [products initial]x n [reactants initial]y n n Q = K; system at equilibrium Q>K; system shifts to left Q<K; system shifts to right
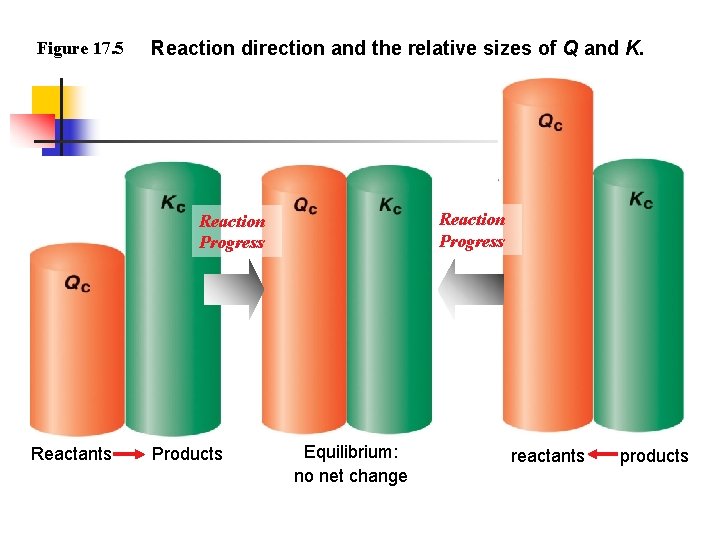
Figure 17. 5 Reaction direction and the relative sizes of Q and K. Reaction Progress Reactants Products Equilibrium: no net change reactants products

Sample Problem 17. 6 PROBLEM: Comparing Q and K to Determine Reaction Direction For the reaction N 2 O 4(g) 2 NO 2(g), Kc = 0. 21 at 100 o. C. At a point during the reaction, [N 2 O 4] = 0. 12 M and [NO 2] = 0. 55 M. Is the reaction at equilibrium? If not, in which direction is it progressing?

Sample Problem 17. 11 Predicting Reaction Direction and Calculating Equilibrium Concentrations PROBLEM: The research and development unit of a chemical company is studying the reaction of CH 4 and H 2 S, two components of natural gas. CH 4(g) + 2 H 2 S(g) CS 2(g) + 4 H 2(g) In one experiment, 1. 00 mol of CH 4, 1. 00 mol of CS 2, 2. 00 mol of H 2 S, and 2. 00 mol of H 2 are mixed in a 250 -m. L vessel at 960 o. C. At this temperature, Kc = 0. 036. (a) In which direction will the reaction proceed to reach equilibrium? (b) If [CH 4] = 5. 56 M at equilibrium, what are the equilibrium concentrations of the other substances?

Sample Problem 17. 11 Predicting Reaction Direction and Calculating Equilibrium Concentrations Concentration (M) initial change equilibrium CH 4(g) + 2 H 2 S(g) CS 2(g) + 4 H 2(g)
- Slides: 22