EQUILIBRIUM ACIDS BASES CHEMICAL EQUILIBRIUM 100 Not all

EQUILIBRIUM, ACIDS & BASES

CHEMICAL EQUILIBRIUM 100% • Not all reactions are quantitative (reactants products) • Evidence: For many reactions reactants are present even after the reaction appears to have stopped • Recall the conditions necessary for a chemical reaction: • Particles must collide with the correct orientation and have sufficient energy • If product particles can collide effectively also, a reaction is said to be reversible • Rate of reaction depends on temperature, surface area and concentration

CHEMICAL EQUILIBRIUM • Consider the following reversible reaction: • The final state of this chemical system can be explained as a competition between: The collisions of reactants to form products The collisions of products to re-form reactants • We assume this system is closed (so the reactants and products cannot escape) and will eventually reach a: DYNAMIC EQUILBRIUM - Opposing changes occur simultaneously at the same rate

CHEMICAL EQUILIBRIUM • Consider the following hypothetical system: AB + CD AD + BC forward reaction, therefore AD + BC AB + CD reverse reaction • Initially, only AB and CD are present. The forward reaction is occurring exclusively at its highest rate. • As AB and CD react, their concentration decreases. This causes the reaction rate to decrease as well. • As AD and BC form, the reverse reaction begins to occur slowly. • As AD and BC’s concentration increases, the reverse reaction speeds up. • Eventually, both the forward and reverse reaction occur at the same rate = DYNAMIC EQUILIBRIUM

4 CONDITIONS OF DYNAMIC EQUILIBRIUM* 1. Can be achieved in all reversible reactions when the rates of the forward and reverse reaction become equal Represented by rather than by 2. All observable properties appear constant (color, p. H, etc. ) 3. Can only be achieved in a closed system (no exchange of matter and must have a constant temperature) 4. Equilibrium can be approached from either direction. This means that the equilibrium concentrations will be the same regardless if you started with all reactants, all products, or a mixture of the two

TWO TYPES OF EQUILIBRIUM: 6 • Static Equilibrium; • opposing forces are balanced and transfer stops • Dynamic Equilibrium: • opposing forces proceed at the same rate • system displays constant macroscopic properties (temp, color, p. H etc. )

VARIOUS TYPES OF DYNAMIC EQUILIBRIUM SYSTEMS CAN OCCUR: 1. Phase Equilibrium: a single substance existing in more than 1 phase • Example: Liquid water in a sealed container with water vapour in the space above it • Water evaporates until the concentration of water vapour rises to a maximum and then remains constant 2. Solubility Equilibrium: a saturated solution • Rate of dissolving = rate of recrystallization
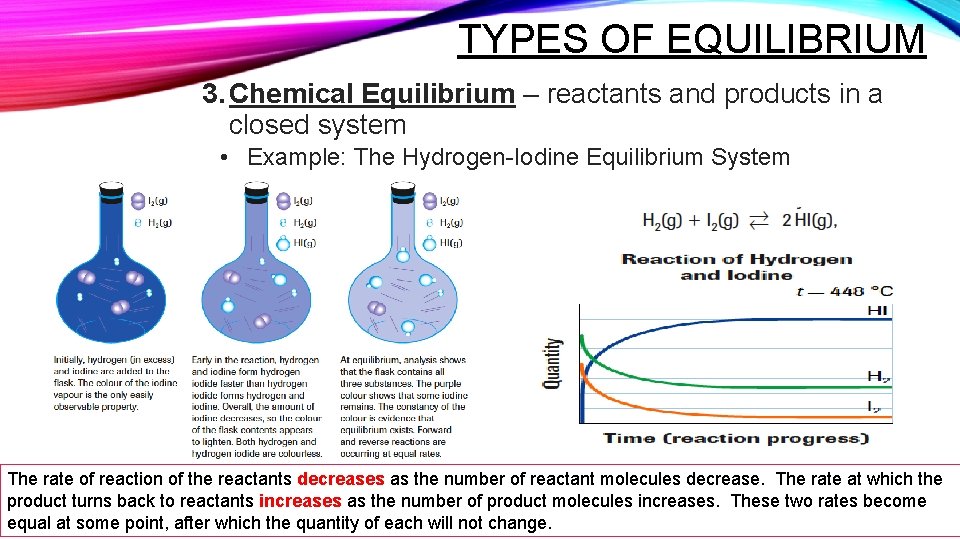
TYPES OF EQUILIBRIUM 3. Chemical Equilibrium – reactants and products in a closed system • Example: The Hydrogen-Iodine Equilibrium System The rate of reaction of the reactants decreases as the number of reactant molecules decrease. The rate at which the product turns back to reactants increases as the number of product molecules increases. These two rates become equal at some point, after which the quantity of each will not change.


DESCRIBING THE POSITION OF EQUILIBRIUM 1. Percent Yield- the yield of product measured at equilibrium compared with the maximum possible yield of product. • • % yield = product eq’m x 100 % product max The equilibrium concentration is determined experimentally, the maximum concentration is determined with stoichiometry

Describing the Position of Equilibrium 1. Percent Yield – Example • If 2. 50 mol of hydrogen gas reacts with 3. 0 mol of iodine gas in a 1. 00 L vessel, what is the percent yield if 3. 90 mol of hydrogen iodide is present at equilibrium

Describing the Position of Equilibrium 2. Using an Equilibrium Constant, (Kc) • This relationship only works if all concentrations are at equilibrium at a constant temperature in a closed system • Think “products over reactants” • If the Kc > 1, the equilibrium favors products • If the Kc < 1, the equilibrium favors reactants


Describing the Position of Equilibrium 2. Using an Equilibrium Constant, (Kc) • Example #1: Write the equilibrium law expression for the reaction of nitrogen monoxide gas with oxygen gas to form nitrogen dioxide gas.

Describing the Position of Equilibrium 2. Using an Equilibrium Constant, (Kc) • Note: The Kc value describes the extent of the forward reaction. Kc reverse = 1 . = The reciprocal value • Kc forward Example #2: The value of Kc for the formation of HI(g) from H 2(g) and I 2(g) is 40, at a given temperature. What is the value of Kc for the decomposition of HI(g) at the same temperature.

Describing the Position of Equilibrium 2. Using an Equilibrium Constant, (Kc) • Note: For heterogeneous equilibrium systems, DO NOT include liquids and solids in the expression. (They are assumed to have fixed concentrations) • Example #3: Write the equilibrium law expression for the decomposition of solid ammonium chloride to gaseous ammonia and gaseous hydrogen chloride The solid is omitted from the expression • Example #4: Write the equilibrium law expression for the reaction of zinc in copper(II) chloride solution. The solids, as well as spectator ions (Cl-) are omitted from the expression

GENERAL RULES FOR THE EQUILIBRIUM EXPRESSION • Solids are omitted from the equilibrium expression because their concentrations are unchanging as a result of density (they cannot be significantly compressed). • Liquids are omitted from the equilibrium expression if there is only one (as in aqueous solutions). If there is more than one liquid present, the liquid can have its concentration changed by dilution. Liquid water is not included, since the dissolved substances do not change the concentration of the water. • If there is more than one liquid, include all the liquids in the equilibrium expression. Liquids must be miscible for their proportions to change

• All other states are included • A complete description of equilibrium state includes the temperature, composition and concentration of all entities present. The concentration for gases is often expressed as pressure

Describing the Position of Equilibrium

CALCULATIONS IN EQUILIBRIUM SYSTEMS • Using the equilibrium law expression to determine whether a system is at equilibrium: • Substitute in the given concentrations to the equilibrium expression. If the value is the equilibrium constant, the system is at equilibrium • If the value is larger, this means there are more products that reactants. To reach equilibrium, the reaction must proceed to the left (towards the reactants) • If the value is smaller, this means there are more reactants than products. To reach equilibrium, the reaction must proceed to the right (towards the products)

CALCULATIONS IN EQUILIBRIUM SYSTEMS • Example #1: In the following system: N 2(g) + 3 H 2(g) ↔ 2 NH 3(g) • 0. 249 mol N 2(g), 3. 21 X 10 -2 mol H 2(g) and 6. 42 X 10 -4 mol NH 3(g) are combined in a 1. 00 L vessel at 375 o. C, Kc = 1. 2 • Is the system at equilibrium?

CALCULATIONS IN EQUILIBRIUM SYSTEMS • Example #2: Find the equilibrium concentration of the ions that are formed when solid silver chloride is dissolved in water. The equilibrium constant for this reaction is Kc = 5. 4 X 10 -4. Ag. Cl(s) Ag+(aq) + Cl-(aq)

ICE CHARTS AND EQUILIBRIUM CALCULATIONS STEPS: • Always write out the equilibrium reaction and equilibrium law expression if not given. • Draw an ICE Chart (Initial, Change in and Equilibrium concentrations) (I + C = E) • Substitute values where appropriate • Solve for x • Solve for equilibrium concentrations





ICE CHARTS AND EQUILIBRIUM CALCULATIONS • Example #1: Consider the following equilibrium at 100 o. C: N 2 O 4(g) ↔ 2 NO 2(g) • 2. 0 mol of N 2 O 4(g) was introduced into an empty 2. 0 L bulb. After equilibrium was established, only 1. 6 mol of N 2 O 4(g) remained. What is the value of Kc?

ICE CHARTS AND EQUILIBRIUM CALCULATIONS Example #2 • A 10 L bulb is filled with 4. 0 mol of SO 2(g), 2. 2 mol of O 2(g) and 5. 6 mol of SO 3(g). The gases then reach equilibrium according to the following equation: 2 SO 2(g) + O 2(g) ↔ 2 SO 3(g) • At equilibrium, the bulb was found to contain 2. 6 mol of SO 2(g). Calculate Kc for this reaction.

YOUR TASK

ICE CHARTS AND EQUILIBRIUM CALCULATIONS • Example #4: Using the Approximation Rule Calculate the concentration of gases produced when 0. 100 mol/L COCl 2(g) decomposes into carbon monoxide and chlorine gas. The Kc for this reaction is 2. 2 X 10 -10.

LE CHATELIER’S PRINCIPLE • If a stress is applied to a dynamic equilibrium system, the system changes to relieve the stress. • Equilibrium shifts can result from: • changes in concentration (pressure – gas only); • temperature change; • Nature of reactants (the chemical properties) • Catalyst Changes in the temperature, will change the Kc value. No other changes will affect this value.

LE CHATELIER’S PRINCIPLE • Effect of Changes in Concentration • If a system at equilibrium is disturbed by the addition of a reactant (or the removal of a product), Le Chatelier’s principle predicts that the equilibrium will shift right. 2 N 2 O(g) + 3 O 2(g) 4 NO 2(g) • If the disturbance is the removal of a reactant (or the addition of a product), Le Chatelier’s principle predicts that the equilibrium will shift left. 2 N 2 O(g) + 3 O 2(g) 4 NO 2(g) • Since concentrations of solids are constants and do not appear in expressions for K, removing or adding some solid does not cause shifts.
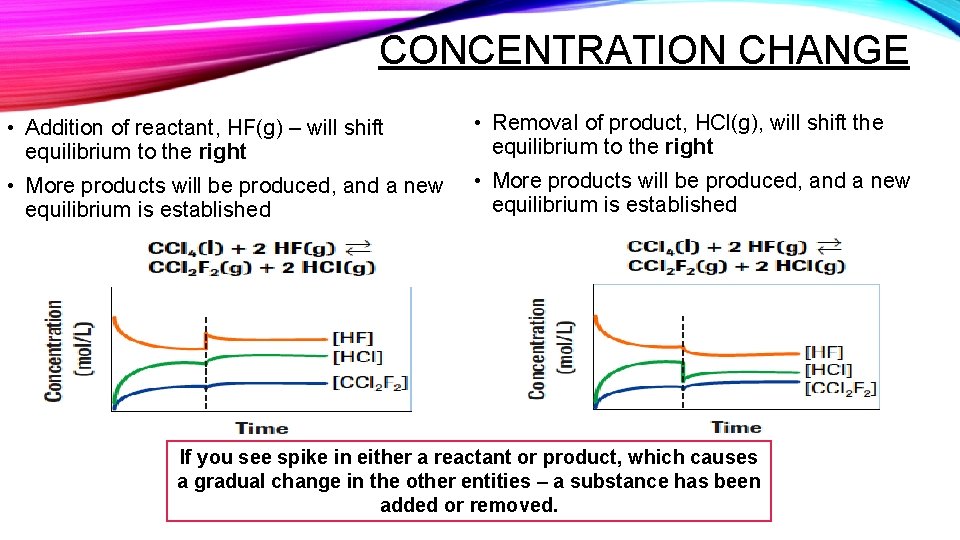
CONCENTRATION CHANGE • Addition of reactant, HF(g) – will shift equilibrium to the right • Removal of product, HCl(g), will shift the equilibrium to the right • More products will be produced, and a new equilibrium is established If you see spike in either a reactant or product, which causes a gradual change in the other entities – a substance has been added or removed.

LE CHATELIER’S PRINCIPLE • Effect of Changes in Temperature • In endothermic reaction, heat acts like a reactant. Increasing the temperature shifts the reaction right. Decreasing the temperature, shifts the reaction left Heat + 2 N 2 O(g) + 3 O 2(g) 4 NO 2(g) • In exothermic reactions, heat acts like a product. Increasing the temperature shifts the reaction left. Decreasing the temperature, shifts the reaction right. 4 NO 2(g) 2 N 2 O(g) + 3 O 2(g) + Heat • The equilibrium constant, Kc is temperature dependent Remember K gets bigger if there are more products being created (i. e. shifts to the right)
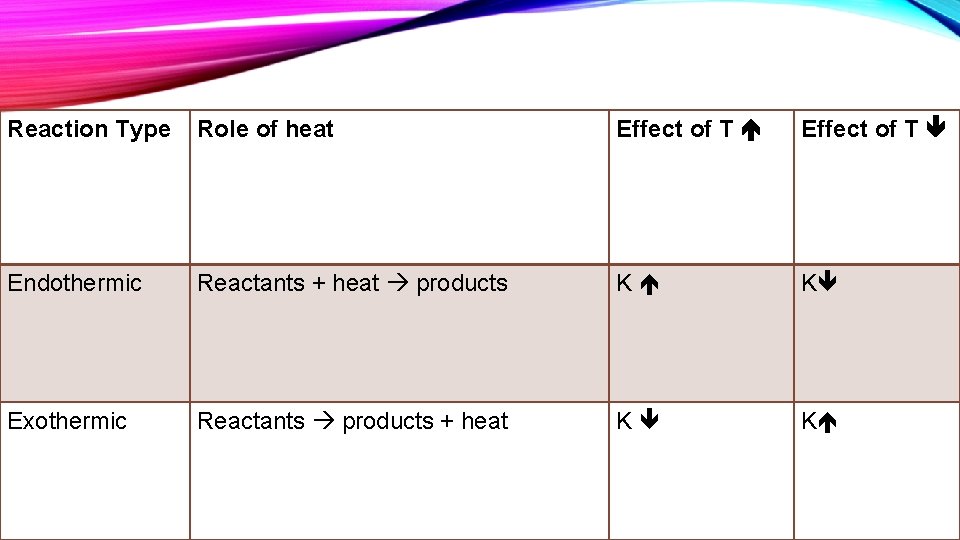
Reaction Type Role of heat Effect of T Endothermic Reactants + heat products K K Exothermic Reactants products + heat K K
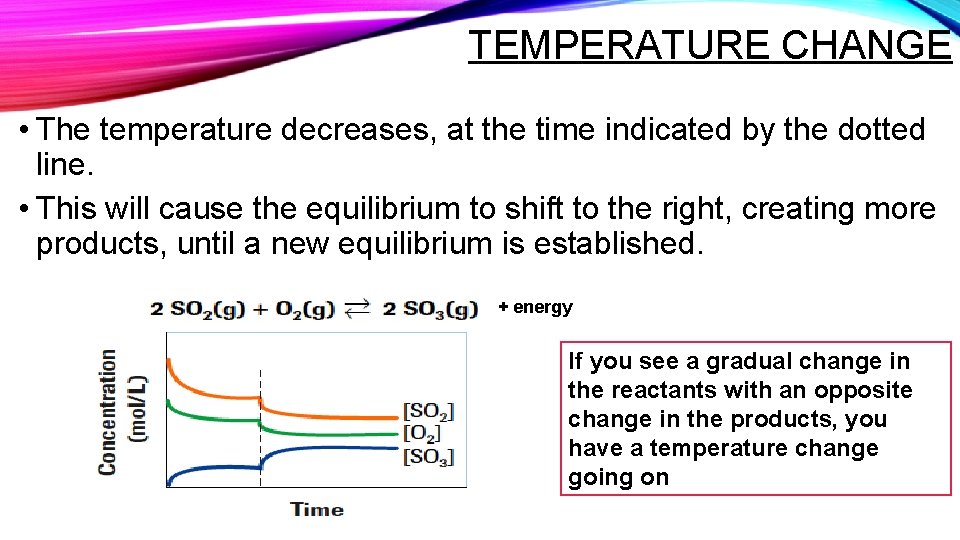
TEMPERATURE CHANGE • The temperature decreases, at the time indicated by the dotted line. • This will cause the equilibrium to shift to the right, creating more products, until a new equilibrium is established. + energy If you see a gradual change in the reactants with an opposite change in the products, you have a temperature change going on

PRESSURE CHANGE • Pressure affects only gases. • When volume decreases, the pressure increases. • When volume increases, the pressure decreases. • Le Chatelier’s principle states that when you change the pressure of a closed system at equilibrium, the reaction will shift to produce more or fewer molecules of gas to oppose the shift. • Example: If you decrease the pressure, the reaction will counteract the shift and increase the pressure because the opposing reaction will create more molecules. The reaction will shift to the side where there are molecules of gas.

LE CHATELIER’S PRINCIPLE • Effect of Changes in the Volume of the Container • If volume is decreased, pressure increases (Boyle’s Law – Chem 20) So the reaction will shift in the direction which contains the fewest moles of gas Pressure 2 N 2 O(g) + 3 O 2(g) 4 NO 2(g) 4 moles are fewer than 5 • If volume is increased, pressure decreases (Boyle’s Law – Chem 20) So the reaction will shift in the direction which contains the most moles of gas Pressure 2 N 2 O(g) + 3 O 2(g) 4 NO 2(g) 5 moles are more than 5 • If both sides of the equation have the same number of moles of gas, the change in volume of the container has no effect on the equilibrium.
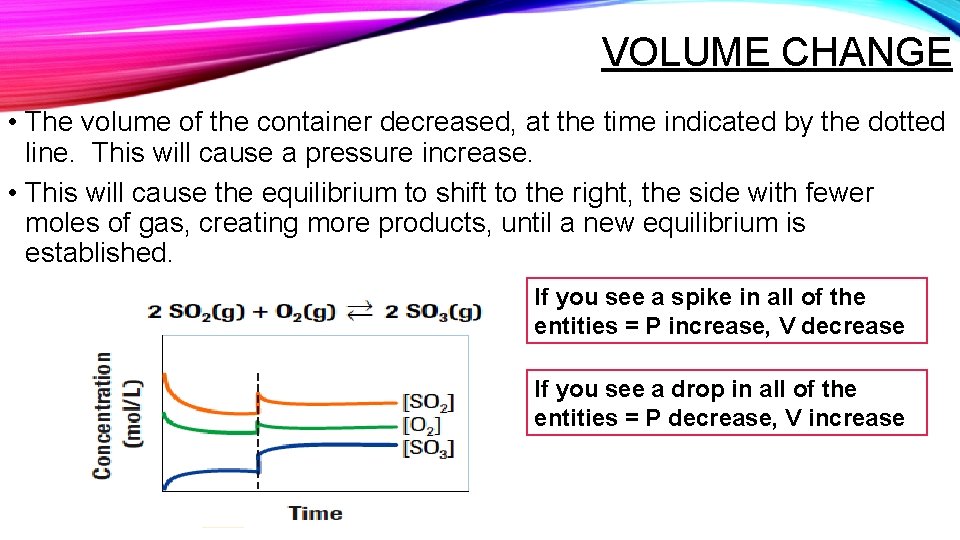
VOLUME CHANGE • The volume of the container decreased, at the time indicated by the dotted line. This will cause a pressure increase. • This will cause the equilibrium to shift to the right, the side with fewer moles of gas, creating more products, until a new equilibrium is established. If you see a spike in all of the entities = P increase, V decrease If you see a drop in all of the entities = P decrease, V increase

LE CHATELIER’S PRINCIPLE • Effect of the Addition of a Catalyst • Catalysts speed up the rate at which equilibrium is obtained, but have no effect on the magnitude of K. They increase both the forward and backward rate of reaction.

HABER-BOSCH PROCESS • Is an important process for adding nitrates to fertilizers and was used in the manufacture of explosive during the Second World War.
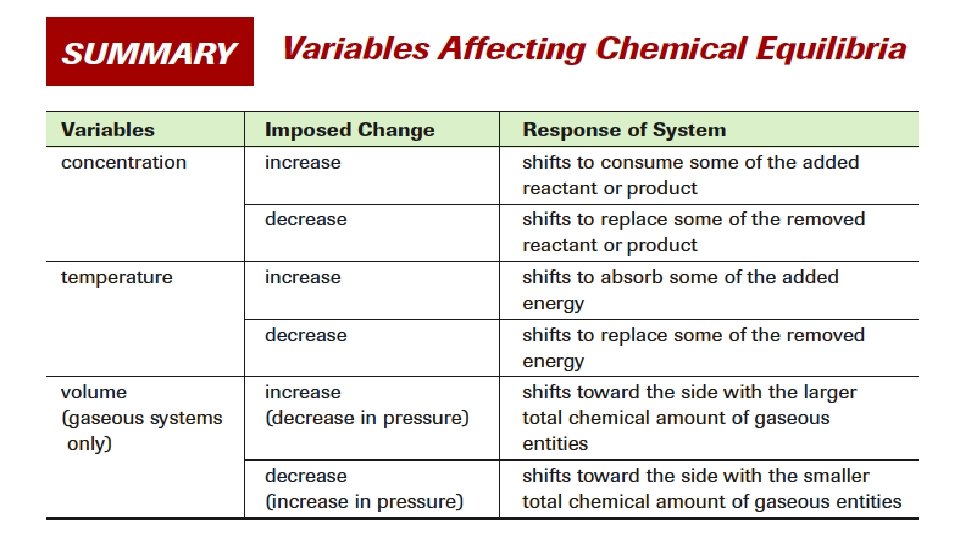
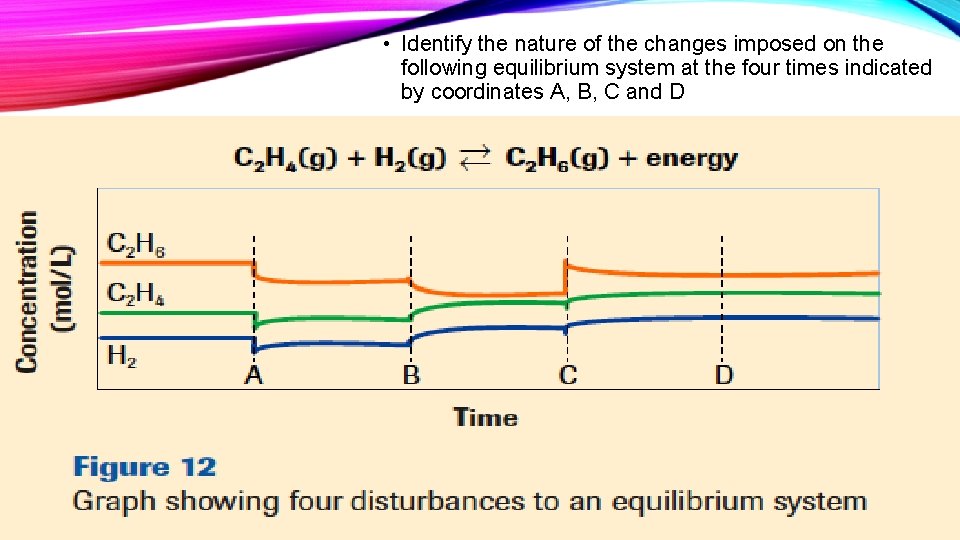
• Identify the nature of the changes imposed on the following equilibrium system at the four times indicated by coordinates A, B, C and D
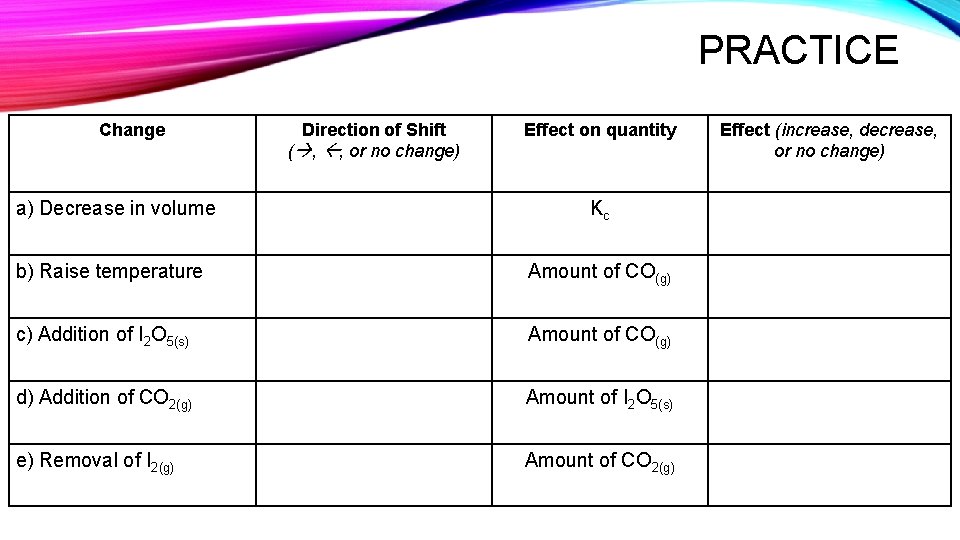
PRACTICE Change a) Decrease in volume Direction of Shift ( , , or no change) Effect on quantity Kc b) Raise temperature Amount of CO(g) c) Addition of I 2 O 5(s) Amount of CO(g) d) Addition of CO 2(g) Amount of I 2 O 5(s) e) Removal of I 2(g) Amount of CO 2(g) Effect (increase, decrease, or no change)
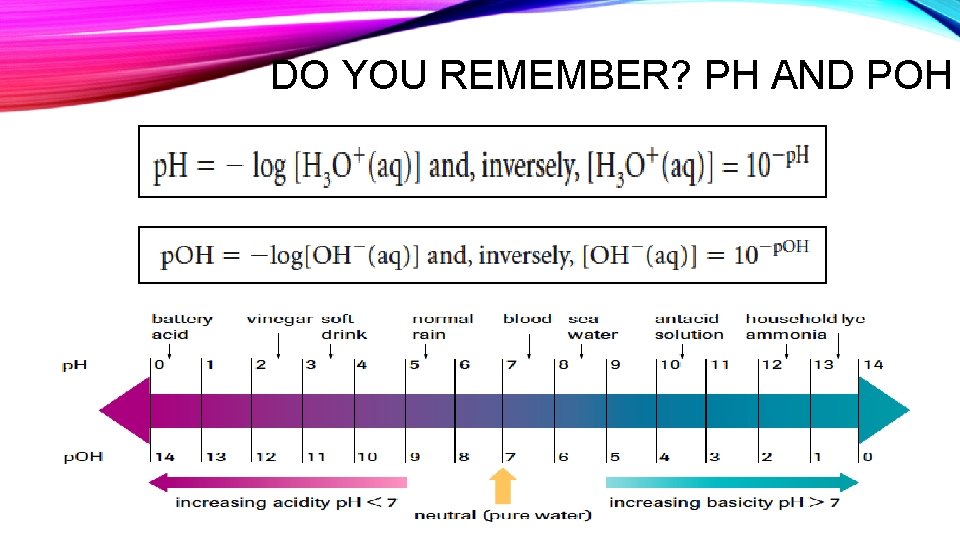
DO YOU REMEMBER? PH AND POH
![PH MEASUREMENTS & CALCULATIONS • p. H = - log [H 3 O+ (aq)] PH MEASUREMENTS & CALCULATIONS • p. H = - log [H 3 O+ (aq)]](http://slidetodoc.com/presentation_image/1a07de3de01fb6d0e826846efe2bc75c/image-47.jpg)
PH MEASUREMENTS & CALCULATIONS • p. H = - log [H 3 O+ (aq)] • p. H + p. OH = 14. 0

SIGNIFICANT DIGITS AND PH • Significant Digits: The number of digits FOLLOWING the decimal point in the p. H value is equal to the number of significant digits in the hydronium ion concentration.

PRACTICE

ARRHENIUS ACIDS & BASES • An Arrhenius: • Acid reacts with water to form hydronium ions (H 3 O+) • Base reacts with water to form hydroxide ions (OH-)

CONCENTRATION VS. STRENGTH • Concentration refers to the number of moles dissolved per liter of solution • Dilute vs. Concentrated • Strength is the extent to which an acid ionizes

ACID STRENGTH AS AN EQUILIBRIUM POSITION • Do you remember the difference between strong and weak acids? • Strong acids – ionize completely (quantitatively), even though this could be written with a double arrow, it is simpler to use a single arrow to show the reaction is >99. 9% • Strong Acids: HCl. O 4(aq), HI(aq), HBr(aq) , HCl(aq) , HSO 4(aq), HNO 3(aq) • Weak acids – ionize (react with water) only partially (<50%)

ACID IONIZATION • To calculate the equilibrium constant you must compare the concentration of the products with the concentration of the reactants

% IONIZATION • In a 0. 10 mol/L solution of acetic acid, only 1. 3% of the CH 3 COOH molecules have reacted at equilibrium to form hydronium ions. Calculate the hydronium ion amount concentration.

% IONIZATION • The p. H of 0. 10 mol/L methanoic acid solution is 2. 38. Calculate the percent reaction for ionization of methanoic acid.
![ACID/BASE TITRATIONS • Standard solution – [known] • Equivalence point – amount of base ACID/BASE TITRATIONS • Standard solution – [known] • Equivalence point – amount of base](http://slidetodoc.com/presentation_image/1a07de3de01fb6d0e826846efe2bc75c/image-56.jpg)
ACID/BASE TITRATIONS • Standard solution – [known] • Equivalence point – amount of base added = amount of acid in solution • End point – indicator color change at p. H change
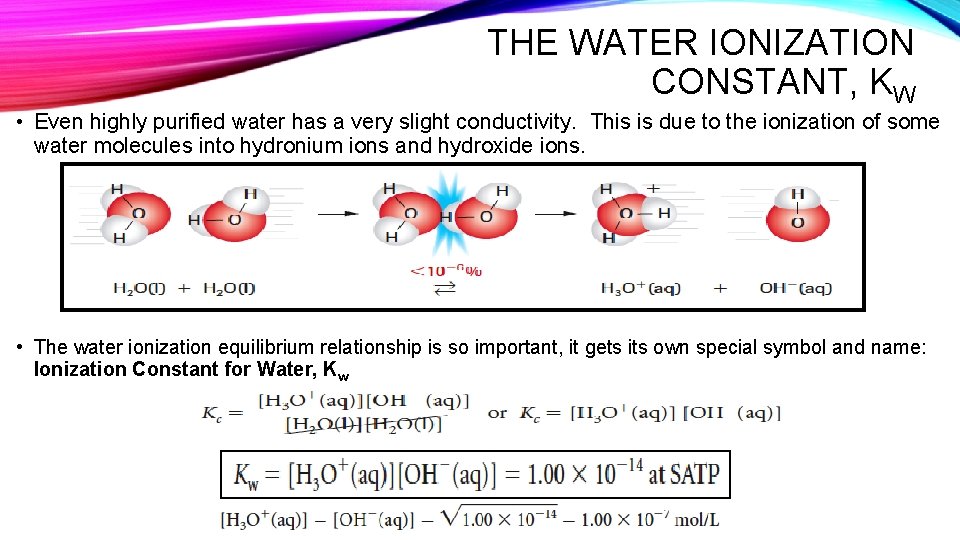
THE WATER IONIZATION CONSTANT, KW • Even highly purified water has a very slight conductivity. This is due to the ionization of some water molecules into hydronium ions and hydroxide ions. • The water ionization equilibrium relationship is so important, it gets its own special symbol and name: Ionization Constant for Water, Kw

THE WATER IONIZATION CONSTANT, KW • Since the mathematical relationship is simple, we can easily use Kw to calculate either the hydronium or hydroxide ion concentration, if the other concentration is know. The presence of substances other than water decreases the certainty of the Kw value to two significant digits; 1. 0 x 10 -14

KW CALCULATIONS • Example: A 0. 15 mol/L solution of hydrochloric acid at 25°C is found to have a hydronium ion concentration of 0. 15 mol/L. Calculate the amount concentration of the hydroxide ions.

KW CALCULATIONS • Example #2: Calculate the amount concentration of the hydronium ion in a 0. 25 mol/L solution of barium hydroxide. • Ba 2+ = 0. 25 mol/L x 2 mol = 0. 50 mol/L 1 mol

KW CALCULATIONS • Example #3: Determine the hydronium ion and hydroxide ion amount concentration in 500 m. L of an aqueous solution for home soap-making containing 2. 6 g of dissolved sodium hydroxide.

YOUR TASK

BRONSTED-LOWRY ACID-BASE CONCEPT • Focuses on the role of the chemical species in a reaction rather than on the acidic or basic properties of their aqueous solutions. • Bronsted Lowry Definition for an Acid: proton donor • Bronsted Lowry Definition for an Base: proton acceptor


BRONSTED-LOWRY ACID-BASE CONCEPT • Bronsted-Lowry Reaction Equation: is an equation written to show an acidbase reaction involving the transfer of a proton from one entity (an acid) to another (a base) • Bronsted-Lowry Neutralization: is a competition for protons that results in a proton transfer from the strongest acid present to the strongest base present. • The Bronsted-Lowry concept does away with defining a substance as being an acid or base. Only an entity that is involved in a proton transfer in a reaction can be defined as an acid or base – and only for a particular reaction Remember this is just a theoretical definition, not a theory, because it does not explain why a proton is donated or accepted, and cannot predict which reaction will occur for a given entity in any new situation.

BRONSTED-LOWRY ACID-BASE CONCEPT • Protons may be gained in a reaction with one entity, but lost in a reaction with another entity. • The empirical term, amphoteric, refers to a chemical substance with the ability to react as either an acid or base. • The theoretical term, amphiprotic, describes an entity (ion or molecule) having the ability to either accept or donate a proton. • Example: When bicarbonate ions are in aqueous solution, some react with the water molecules by acting as an acid, and some react by acting as a base. Kc values given for these reactions, show that one predominates. The number of ions acting as a base is over 2000 x more than the number reacting as an acid. = BASIC Solution

• Polyprotic acid is an acid with more than one proton to donate. • Polybasic Base is a base that can accept more than one proton.

BRONSTED-LOWRY ACID BASE CONCEPT • The Amphoteric Nature of Baking Soda • It can partially neutralize a strong acid, but also a strong base

CONJUGATE ACIDS AND BASES • In an acid-base reaction, there will always be two acids and two bases. • The original acid on the left and the acid on the right created by adding a proton. • The original base on the left and the base on the right created by removing a proton • A pair of substances with formulas that differ only by a proton is called a conjugate acid-base pair This analogy shows why acetic acid is a weak acid. The proton is more strongly attracted to the acetic acid molecule than it is to the water.

ACIDS/BASES • What about strong acids: • When HCl reacts with water, the water wins the competition against the Cl- for the proton. This is why at equilibrium, essentially all of the HCl molecules have lost protons to water. >99% • NOTE: The stronger the base, the more it attracts a proton (proton acceptor). The stronger the acid, the less it attracts its own proton (proton donor)

CONJUGATE ACIDS AND BASES • RULE: The stronger the base, the more it attracts a proton (proton acceptor). The stronger the acid, the less it attracts its own proton (proton donor) • What does this mean about their conjugate pair? ? • The stronger an acid, the weaker is its conjugate base. • If you are good at donating a proton, this means the conjugate base is not good at competing for it (weak attraction for protons) • The stronger a base, the weaker is its conjugate acid. • If you are good at accepting a proton, this means the conjugate acid is not good at giving it up (strong attraction for protons).


CONJUGATE ACID-BASE PAIRS • Write formulae for the conjugate bases of the following acids; HCO 3 - (aq) HCl (aq) H 2 SO 4 (aq) NH 4+ (aq) H 2 O (l) H 2 CO 3 (aq) HOOCCOOH (aq) H 2 PO 4 - (aq)

CONJUGATE ACID-BASE PAIRS • Write formulae for the conjugate acids of the following bases; Br - (aq) NO 2 - (aq) PO 4 3 - (aq) HSO 4 - (aq) HO 2 - (aq) HS- (aq) HCO 3 - (aq) CO 3 2 - (aq)
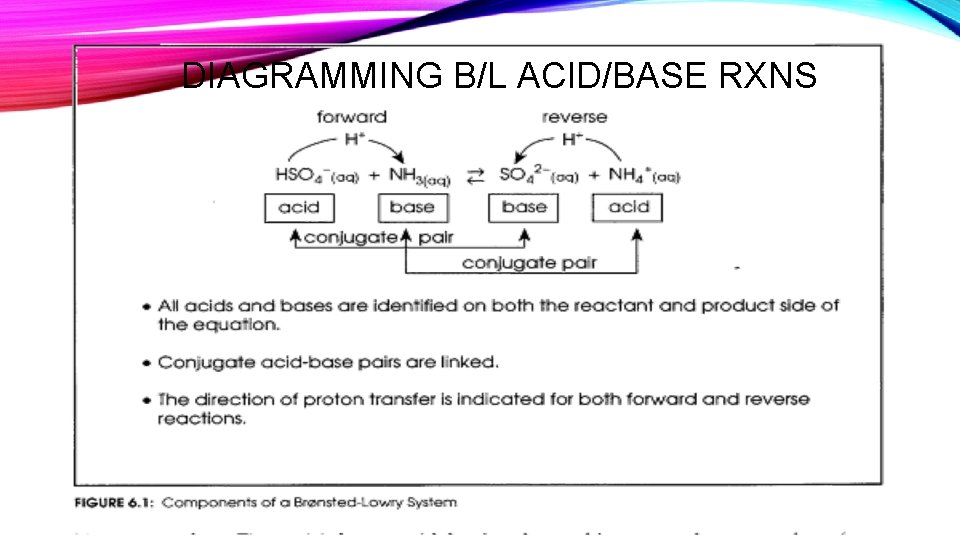
DIAGRAMMING B/L ACID/BASE RXNS

AMPHIPROTIC SUBSTANCES • An amphiprotic substance will act as a(n): • Acid if it is a better proton donor than the substance with which it is reacting • Base if it is a better proton acceptor than the substance with which it is reacting.

AMPHIPROTIC SUBSTANCES • Note: You have seen amphiprotic substances show up on both sides of the Acids-Bases Table. When identifying substances as possible acids or bases it is important for you to remember to check BOTH sides of the table.

AMPHIPROTIC SUBSTANCES • Examine the Acid-Bases Table. List 10 substances that are amphiprotic. • hydrogen sulfate ion • hydrogen phosphate ion • hydrogen oxalate ion • dihydrogen borate ion • hydrogen sulfide ion • dihydrogen phosphate ion • hydrogen sulfite ion • hydrogen borate ion • hydrogen carbonate ion • water

ACID-BASE REACTIONS • What is happening? • Collisions are constantly occurring. Each time, a proton is transferred to a stronger proton attractor. • Theoretically, it could transfer several times (each time to a stronger proton attractor. ) But once, it is transferred to the strongest base present, the proton will remain there as nothing outcompetes it. • Likewise, once the strongest acid has given up its proton, its conjugate base cannot gain one back (as it is the weakest proton attractor in the whole system) • So what does this mean? • Proton transfer occurs between the strongest acid and strongest base. All other transfers are negligible so are ignored.

PREDICTING ACID-BASE REACTIONS

PREDICTING ACID-BASE REACTIONS • 1) List all entities present initially (ions, atoms, molecules, H 2 O(l)) as they exist in aqueous solution. No entity can react as a base if it is weaker than water. For this reason, the conjugate bases of the strong acids are not considered bases in aqueous solutions • H 3 O+(aq) is the SA that can exist. If a stronger acid is dissolved, it reacts instantly and completely with water to form H 3 O+(aq). So all strong acids are written as H 3 O+(aq) • OH-(aq) is the SB that can exist. If a stronger base is dissolved, it reacts instantly and completely with water to form OH-(aq). The only example of this: soluble ionic oxides, write the cation and the oxide ion is written as OH-(aq)

PREDICTING ACID-BASE REACTIONS • 1) List all entities present initially (ions, atoms, molecules, H 2 O(l)) as they exist in aqueous solution. • Example: What will be the predominant reaction if spilled drain cleaner (sodium hydroxide) solution is neutralized by vinegar? • List entities present:

PREDICTING ACID-BASE REACTIONS 2) Identify and list all possible aqueous acids and bases, using the Bronsted-Lowry definitions. • Use the entity lists of the Relative Strengths of Acids and Bases (in your data booklet). Amphiprotic entities are labeled fro both possibilities. Conjugate bases on SA’s are not included. Metal ions are treated as spectators. • Example: What will be the predominant reaction if spilled drain cleaner (sodium hydroxide) solution is neutralized by vinegar? • List entities present:

PREDICTING ACID-BASE REACTIONS 3) Identify the strongest acid and the strongest base present, using the table of Relative Strengths of Acids and Bases. • Use the order of the entities in the Relative Strengths of Acids and Bases table to identify the SA (the highest one on the table) and the SB (the lowest one on the table) • Example: What will be the predominant reaction if spilled drain cleaner (sodium hydroxide) solution is neutralized by vinegar? • List entities present:

PREDICTING ACID-BASE REACTIONS 4) Write an equation showing a transfer of one proton from the SA to the SB, and predict the conjugate base and the conjugate acid to be the products. • Example: What will be the predominant reaction if spilled drain cleaner (sodium hydroxide) solution is neutralized by vinegar? • List entities present:

PREDICTING ACID-BASE REACTIONS • 5) Predict the approximate position of equilibrium, using the following Learning Tip • Example: What will be the predominant reaction if spilled drain cleaner (sodium hydroxide) solution is neutralized by vinegar?

TABLE BUILDING • If a reaction is >50%, it favours products and the SA is above the SB. • If a reaction is <50%, it favours reactants and the SA is below the SB.
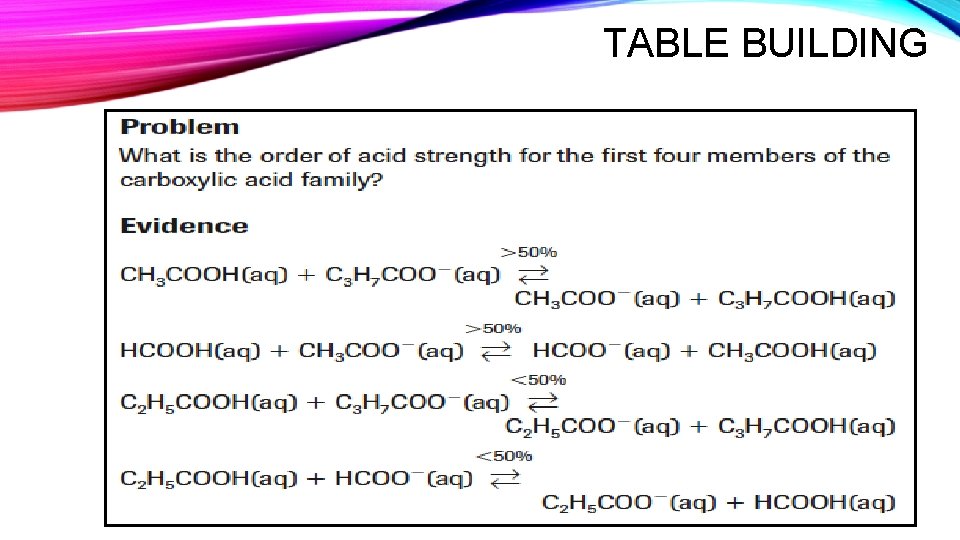
TABLE BUILDING
Acid Base

THE RELATIVE ACID-BASE STRENGTH TABLE • Acid-base strength tables are built upon the principle of comparing acids and bases under identical conditions. • Use the Acid-Base Strength Data Table to arrange the following acids according to strength in a Table with the following headings. Acid Name Formula % Reaction Eq. Constant (Ka)

• ammonium ion • ethanoic acid • benzoic acid • hydroiodic acid CREATING AN ACID STRENGTH TABLE • hydrosulfuric acid • hydrogen sulfate ion • hydrogen carbonate ion • hydrogen borate ion • Explain the trends you notice comparing the percent reaction to the equilibrium constants.

TRENDS ON ACID BASE TABLE • Weaker acids - % reaction smaller & Ka smaller because decreasing ability to donate protons • Example: reactants favored over products so denominator gets larger

THE ACID IONIZATION CONSTANT, KA • One important way that chemists communicate the strength of any weak acid is by using the equilibrium constant expression for the ionization of weak acids. (Ka) • Look at your Relative Strengths of Acids and Bases Table: • All of the strong acids have a Ka value listed as very large; remember they ionize quantitatively, so the actual acid species present is H 3 O+(aq) • All other acids are weak and vary greatly in the extent of reaction at equilibrium. • We use the equilibrium law to write the formula for Ka We omit liquid water because we assume its value is essentially constant for dilute solutions Ka Values have only two sig digs because they are somewhat inaccurate because we calculate them based on assumptions.


KA CALCULATIONS • Two common Ka calculations: • Calculating Ka from empirical data • Using Ka to predict hydronium ion concentration from an initial concentration of weak acid.



KA CALCULATIONS • Example #1: The p. H of a 1. 00 mol/L solution of acetic acid is carefully measured to be 2. 38 at SATP. What is the value of Ka for acetic acid?



KA CALCULATIONS • Example #2: A student measures the p. H of a 0. 25 mol/L solution of carbonic acid to be 3. 48. Calculate the Ka for carbonic acid from this evidence.

KA CALCULATIONS • Example #3: The p. H of a 0. 400 mol/L solution of sulfurous acid is measured to be 1. 17. Calculate the Ka for sulfurous acid from this evidence.



KA CALCULATIONS • Example #4: Predict the hydronium ion concentration and p. H for a 0. 200 mol/L aqueous solution of methanoic acid.



KA CALCULATIONS • Example #5: Predict the hydronium ion concentration and p. H for a 0. 500 mol/L aqueous solution of hydrocyanic acid.





BASE STRENGTH & IONIZATION CONSTANT, KB • Ionic hydroxides, such as Na. OH(aq) or Ca(OH)2(aq), are assumed to dissociate completely upon dissolving. • Finding the hydroxide ion concentration does not involve any reaction with water • Example: Find the hydroxide ion concentration of a 0. 064 mol/L solution of barium hydroxide

BASE STRENGTH & IONIZATION CONSTANT, KB • We can communicate the strength of weak bases, with the equilibrium constant for its reaction with water. • The equilibrium constant is called the base ionization constant, Kb • Two common Kb calculations: • Calculating Kb from empirical data Using Kb to predict the concentration of hydroxide ions when the initial concentration is known

KB CALCULATIONS • Example #1: A student measures the p. H of a 0. 250 mol/L solution of aqueous ammonia and finds it to 11. 32. Calculate the Kb for ammonia

THE RELATIONSHIP BETWEEN KA AND KB

EXAMPLE • Using the values for the weak acid NH 4+, determine the Kb for its conjugate base

YOUR TASK

CALCULATING OH- FROM KB • First problem, the data booklet has Ka values not Kb values. What do we do? • Kw = Ka. Kb So Kb = Kw/Ka remember Kw = 1. 00 x 10 -14 • Example #1: Solid sodium benzoate forms a basic solution. Determine the Kb for the weak base present.

CALCULATING OH- FROM KB • Example #2: Find the hydroxide ion amount concentration, p. OH, p. H and the percent reaction (ionization) of a 1. 20 mol/L solution of baking soda. • Baking soda = Na. HCO 3(s) Na+(aq) + HCO 3 -(aq) • For HCO 3 -(aq), the conjugate acid is H 2 CO 3(aq) whose Ka is = 4. 5 x 10 -7

CALCULATING OH- FROM KB • Example #2: Find the hydroxide ion amount concentration, p. OH, p. H and the percent reaction (ionization) of a 1. 20 mol/L solution of baking soda.

SOME THINGS TO CONSIDER ABOUT ACIDS AND BASES

AMPHOTERIC SPECIES • If an entity can react as either a Bronsted-Lowry acid or base, how do you know which will be the predominant reaction? • Find the Ka value, calculate the Kb value, which ever is larger wins! • Example: Which reaction predominates when Na. HSO 3(s) is dissolved in water to produce HSO 3 -(aq) solution? Will the solution be acidic or basic? The Ka value far exceeds the Kb value, so an aqueous solution of this substance will be acidic because the hydrogen sulfite ion will react predominately as a Bronsted-Lowry acid.

TITRATION TERMINOLOGY REVIEW

• A graph showing the continuous change of p. H during an acidbase titration, which continues until the titrant is in great excess, is called a p. H curve • Endpoint refers to the point in a titration analysis where the addition of titrant is stopped. The endpoint is defined (empirically) by the observed colour change of an indicator. • Equivalence Point refers to the point in any chemical reaction where chemically equivalent amounts of the reactants have combined. This point is determined by stoichiometry.
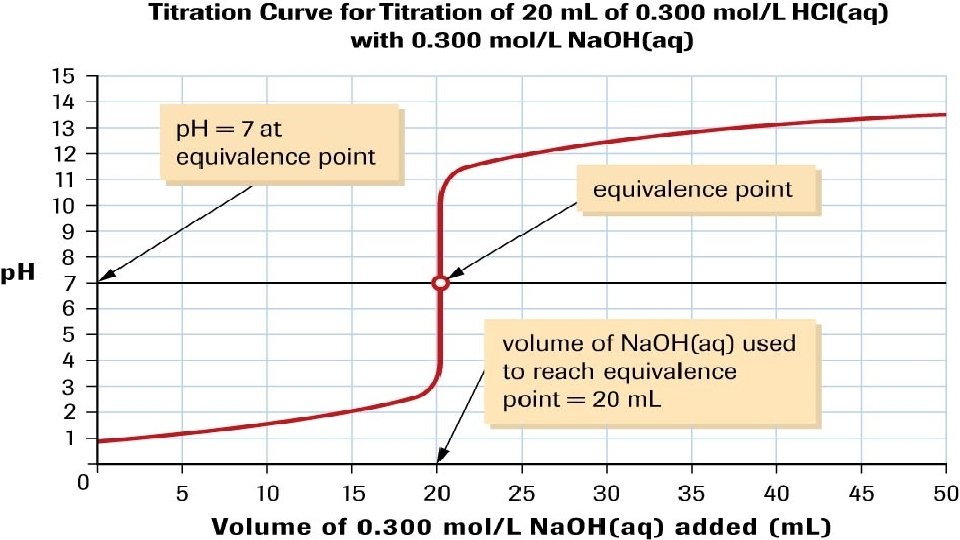
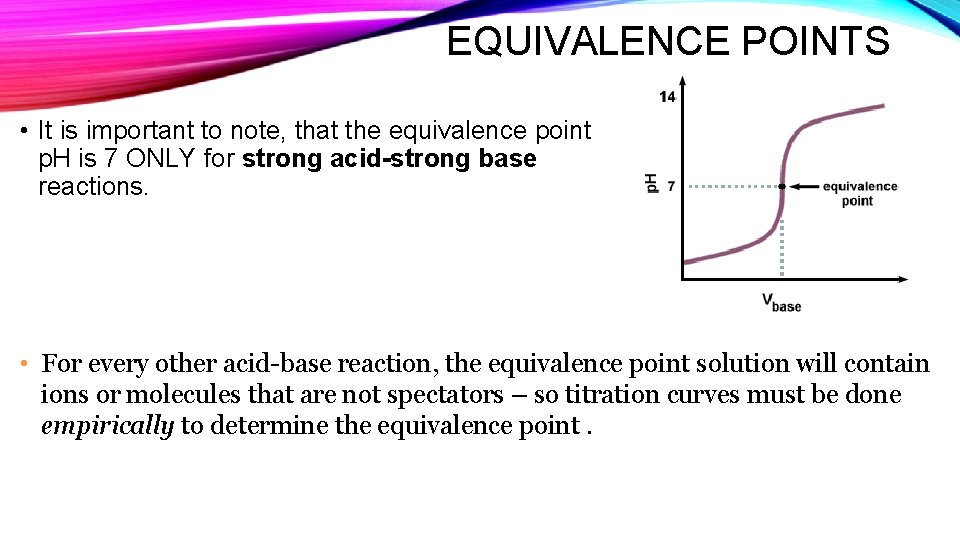
EQUIVALENCE POINTS • It is important to note, that the equivalence point p. H is 7 ONLY for strong acid-strong base reactions. • For every other acid-base reaction, the equivalence point solution will contain ions or molecules that are not spectators – so titration curves must be done empirically to determine the equivalence point.

INTERPRETING PH CURVES • Buffering: is the property of some solutions to resist any significant change in p. H when an acid or base is added. • Buffering region (nearly level portions of the graph) Why is it buffering? Initially, the solution is mainly all water and OH- ions. Any additional acid added (H 3 O+) immediately reacts with OH- to become water – which does not change the p. H significantly. This “leveling effect” finally fails near the equivalence point, when the OH- is almost completely consumed. Once excess acid has been added, the solution consists of water and H 3 O+ ions, so the p. H has dropped to the acid range. Then any additional acid that is added, simple increase the H 3 O+ concentration slightly, but does not change the p. H much.
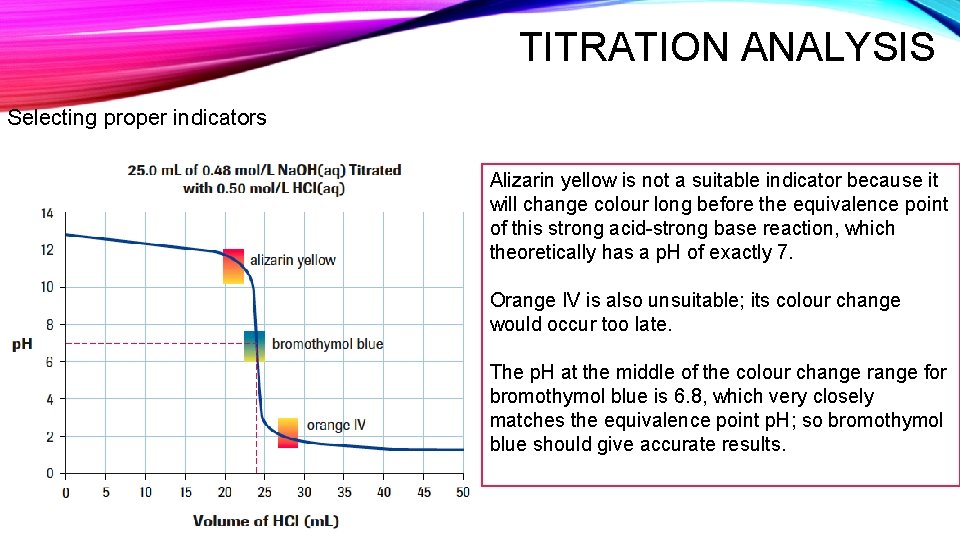
TITRATION ANALYSIS Selecting proper indicators Alizarin yellow is not a suitable indicator because it will change colour long before the equivalence point of this strong acid-strong base reaction, which theoretically has a p. H of exactly 7. Orange IV is also unsuitable; its colour change would occur too late. The p. H at the middle of the colour change range for bromothymol blue is 6. 8, which very closely matches the equivalence point p. H; so bromothymol blue should give accurate results.

ACID BASE INDICATORS • Any acid base indicator is really two entities for which we use the same name: a Bronsted-Lowry conjugate acid-base pair. • At lease one of the entities is visibly colored, so you can tell simply by looking when it forms or is consumed. Examples include: • Phenolphthalein: conjugate acid is colorless, conjugate base is bright pink • Bromothymol Blue: conjugate acid is yellow, conjugate base is blue, and when they are in equal quantities – (appear green to the human eye) • Litmus Paper – red (HIn) to blue (In-) • We will use the designation HIn for the conjugate acid and In- for the conjugate base as their actual formulas can be very complex. • Summary: An indicator is a conjugate weak acid-weak base pair formed when an indicator dye dissolves in water.


SELECTING AN INDICATOR FROM TITRATION CURVE DATA • To select an indicator from titration curve data use the following steps: 1) Decide where the equivalence point is 2) From the indicator list in the data tables, select an indicator that changes color at a p. H close to the actual equivalence point.

INDICATOR EXAMPLE • A solution that is colorless in phenolphthalein, yellow in methyl orange and yellow in cholorphenol red could have a p. H of what?

INDICATOR EXAMPLE • A solution with a p. H of 3. 5 would have the following colors in cresol red, methyl orange and bromocresol green respectively
INDICATOR CHALLENGE QUESTION • When a 0. 10 mol/L HF(aq) is titrated with a 0. 10 mol/L KOH(aq) the best indicator to use would be what?
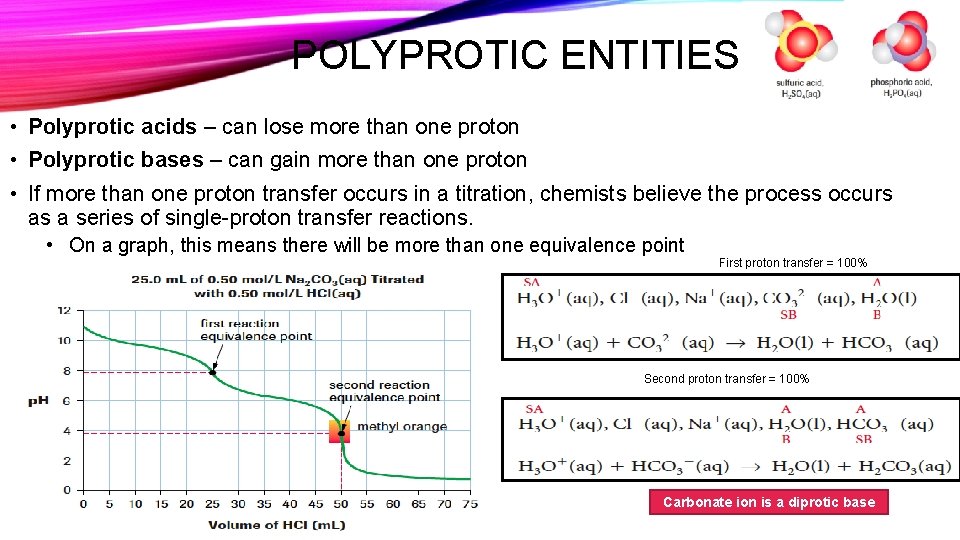
POLYPROTIC ENTITIES • Polyprotic acids – can lose more than one proton • Polyprotic bases – can gain more than one proton • If more than one proton transfer occurs in a titration, chemists believe the process occurs as a series of single-proton transfer reactions. • On a graph, this means there will be more than one equivalence point First proton transfer = 100% Second proton transfer = 100% Carbonate ion is a diprotic base
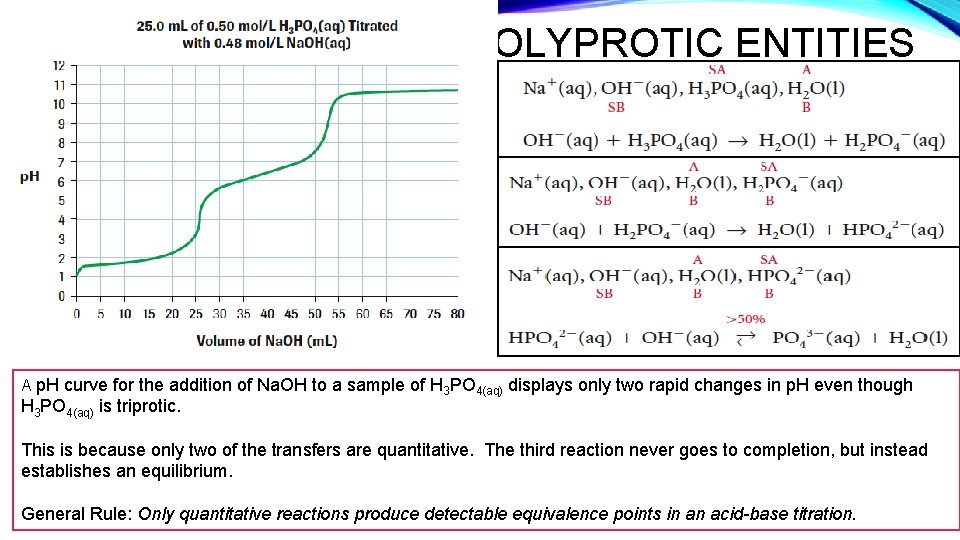
POLYPROTIC ENTITIES A p. H curve for the addition of Na. OH to a sample of H 3 PO 4(aq) displays only two rapid changes in p. H even though H 3 PO 4(aq) is triprotic. This is because only two of the transfers are quantitative. The third reaction never goes to completion, but instead establishes an equilibrium. General Rule: Only quantitative reactions produce detectable equivalence points in an acid-base titration.

EXAMPLE • Sketch the titration curve of a polyprotic base Na 2 CO 3 titrated with a strong acid, hydrochloric acid. Use 25 m. L of 0. 20 mol/L Na 2 CO 3 titrated with 0. 20 mol/L hydrochloric acid for your data

EXAMPLE • Sketch the titration curve of the titration of a polyprotic acid, H 3 PO 4, titrated with a strong base, Na. OH. In terms of experimental data, use 25 m. L of 0. 12 mol/L H 3 PO 4 titrated with 0. 12 mol/L Na. OH.


PH CURVES
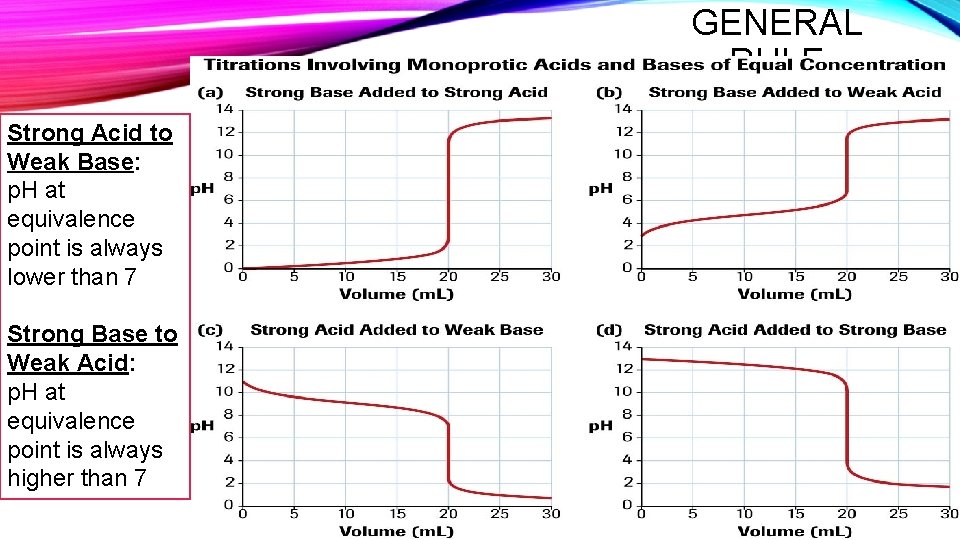
GENERAL RULE Strong Acid to Weak Base: p. H at equivalence point is always lower than 7 Strong Base to Weak Acid: p. H at equivalence point is always higher than 7



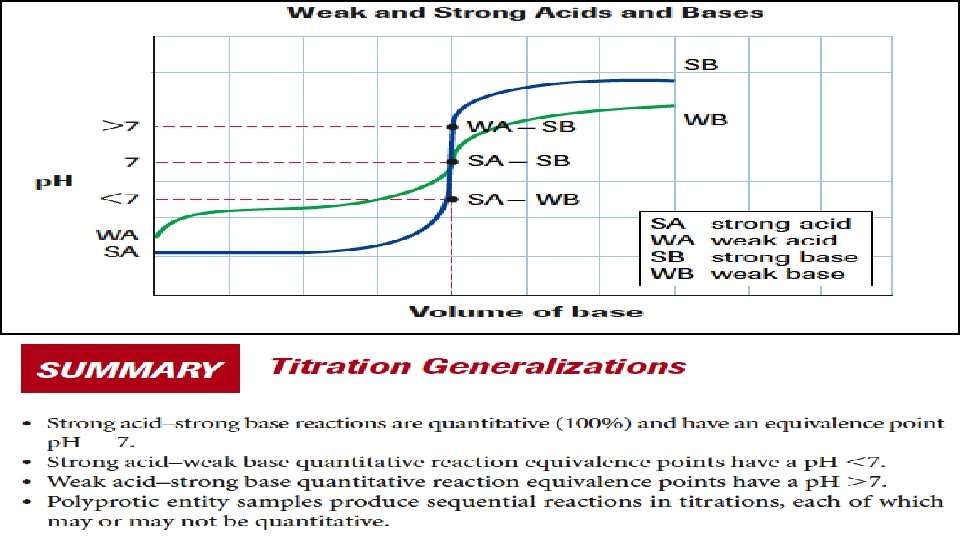

PH CURVE SHAPE • SA-SB: water is the only acid or base present = neutral solution • SA-WB: a weak acid (NH 4+)is present along with water, at the equivalence point, so the solution is acidic (p. H < 7) • WA-SB: a weak base (CH 3 COO-) is present along with water, at the equivalence point, so the solution is basic (p. H > 7) • WA-WB: do not have detectable equivalence points, because the reactions are usually not quantitative.
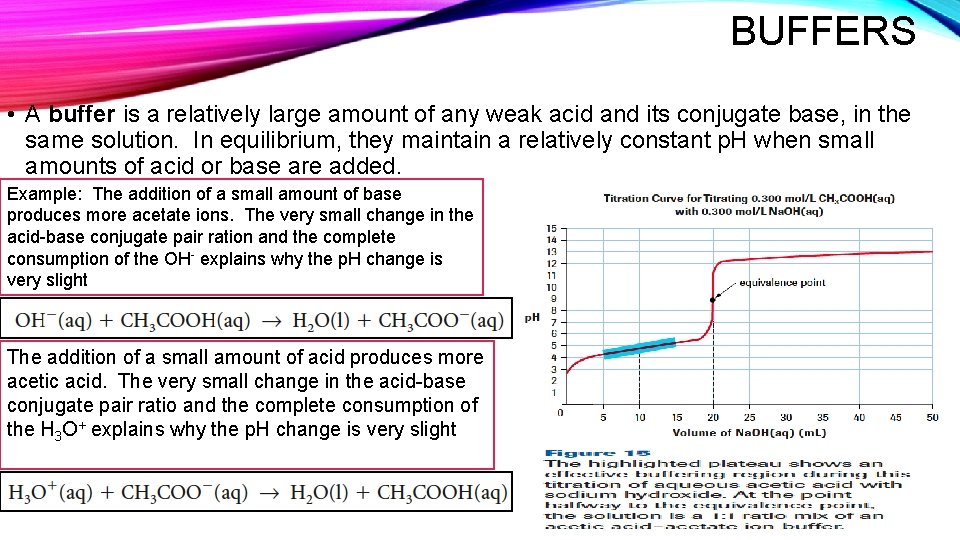
BUFFERS • A buffer is a relatively large amount of any weak acid and its conjugate base, in the same solution. In equilibrium, they maintain a relatively constant p. H when small amounts of acid or base are added. Example: The addition of a small amount of base produces more acetate ions. The very small change in the acid-base conjugate pair ration and the complete consumption of the OH- explains why the p. H change is very slight The addition of a small amount of acid produces more acetic acid. The very small change in the acid-base conjugate pair ratio and the complete consumption of the H 3 O+ explains why the p. H change is very slight



EQUILIBRIUMS IN ACIDIC BUFFER SOLUTIONS • An acidic buffer solution consists of a weak acid and its conjugate base, which are both in reasonably high concentrations. To write the equilibrium equation for an acidic buffer, simply write the equation for the ionization of the weak acid in water.

• For example, consider a buffer solution that is prepared that contains 1. 0 mol/L CH 3 COOH and 1. 0 mol/L Na. CH 3 Coo. This buffer has the following equilibrium equation:

EQUILIBRIUMS IN BASIC BUFFER SOLUTIONS • A basic buffer solution consists of a weak base and the salt of its conjugate acid. To write the equation for the equilibrium existing in a basic buffer solution, write the equation for the ionization of the weak base in water.

EXAMPLE • A buffer solution is prepared containing 1. 0 mol/L NH 3 and 1. 0 mol/L NH 4 Cl. The following equation describes the equilibrium present:

COMPOSITION AND TYPES OF BUFFER SOLUTIONS • There are two types of buffer solutions: acidic buffers and basic buffers Type Composition p. H Range of Operation Acidic Buffer A weak acid and the salt of its conjugate base p. H of 7 or below Basic Buffer A weak base and the salt of its conjugate acid p. H of 7 or above • In any buffer solution, the concentrations of both components should be moderately high. • Neither a strong acid nor a strong base can be used to prepare a buffer solution

SOME EXAMPLES OF ACIDIC BUFFERS

BUFFERS

ADDING AN ACID TO AN ACIDIC BUFFER SOLUTION • A small amount of HCl is added to a buffer solution containing 1. 0 mol/L acetic acid and 1. 0 mol/L and a 1. 0 mol/L sodium acetate. The equilibrium equation for the buffer solution has the following equilibrium equation.

• The following sequence of events takes place: 1. The addition of HCl immediately increases the hydronium ion concentration 2. In order to counteract the increase the hydronium ion concentration, the equilibrium shifts to the left 3. As the equilibrium shifts to the left, CH 3 COO- reacts with H 3 O+ 4. The hydronium ion concentration decreases close to- but not quite as low as- its original concentration 5. In the overall process the final hydronium ion concentration is slightly higher (and the p. H is slightly lower) than it was before the acid was added


EXAMPLE • Describe the effect of adding a strong acid to a solution of carbonic acid and its conjugate base, hydrogen carbonate ion.

BASIC BUFFERS

ADDING A BASE TO A BASIC BUFFER SOLUTION • A small amount of Na. OH solution is added to a buffer solution consisting of 1. 0 mol/L NH 3 and 1. 0 mol/L NH 4 Cl. • The buffer solution has the following equilibrium equation:

• The following sequence of events takes place: 1. The addition of Na. OH (strong base) immediately increases the concentration of hydroxide ions 2. In order to counteract the increase in the hydroxide ion concentration the equilibrium shifts to the left 3. As the equilibrium shifts to the left, NH 4 - reacts with OH 4. The hydroxide ion concentration decreases close to- but not quite as low as- its original concentration 5. In the overall process, the final hydroxide ion concentration is slightly higher (and the p. H slightly higher) than it was before the base was added


BUFFER EXAMPLE: BLOOD PLASMA • Blood plasma has a remarkable buffering ability, as shown by the following table. • This is very useful, as a change of more than 0. 4 p. H units, can be lethal. If the blood were not buffered, the acid absorbed from a glass of orange juice would likely be fatal.

SOME USES OF BUFFER SOLUTIONS • Buffer solutions are widely used today. Some uses include: • Stabilizing p. H in hot tubs and swimming pools • Maintaining optimum p. H for production of pharmaceuticals and other chemicals • Controlling the p. H in wines and foods • Providing solutions with accurate p. Hs for calibrating p. H meters
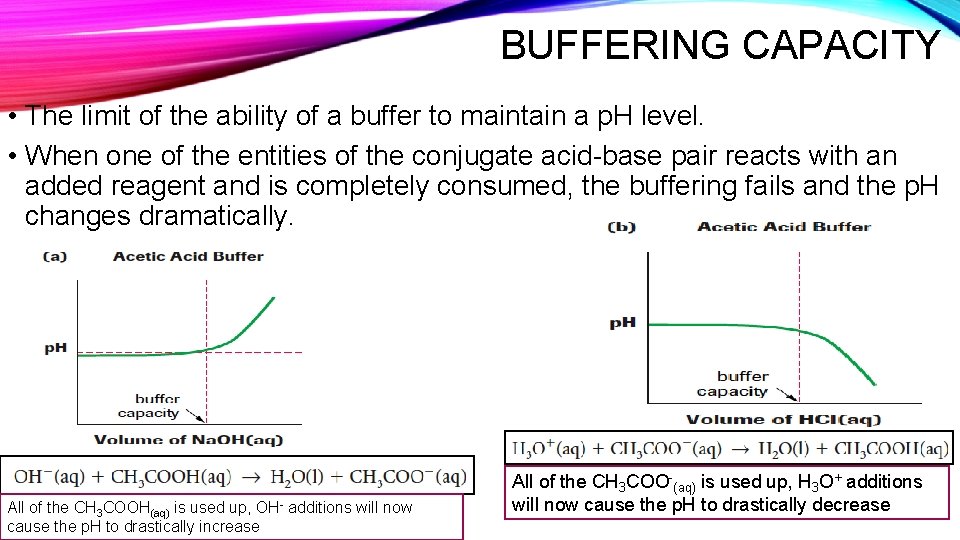
BUFFERING CAPACITY • The limit of the ability of a buffer to maintain a p. H level. • When one of the entities of the conjugate acid-base pair reacts with an added reagent and is completely consumed, the buffering fails and the p. H changes dramatically. All of the CH 3 COOH(aq) is used up, OH- additions will now cause the p. H to drastically increase All of the CH 3 COO-(aq) is used up, H 3 O+ additions will now cause the p. H to drastically decrease
- Slides: 168