Equilibrium Acid Base Short Review Equilibrium n n

Equilibrium & Acid Base Short Review

Equilibrium n n Some reactions are reversible n Conversion of reactants into products & conversion of products into reactants occur simultaneously When forward reactions = back reactions (at the same rate) we say equilibrium has been reached Determine the equilibrium law expression this reaction…

Rules for Equilibrium Expressions What is included in the K expression? n Always included: Gases (g) and solutions (aq) n Never Included: Solids (s) n Sometimes: liquids (l). More than one liquid – include them all. Only 1 liquid – not included. K (constant) remains the same as long as temp does not change!

Classes of Chemical Equilibria

Le Chatelier’s Principle n n If a stress is applied to a system at equilibrium, the system shifts to relieve the stress We study the effects of the following stresses on equilibrium n addition or deletion of reactants or products n addition or deletion of energy (changing temp) n change in pressure/volume n addition of catalysts n addition of inert gases

Le Chatelier Examples Determine the shift direction if… • this equilibrium system is put in ice: ________ • the concentration of sulfur dioxide is increased : ____ • the pressure of the container that contains this equilibrium system is increased : ________ • argon gas is added to this equilibrium system: ______ • hydrosulfuric acid is removed from the system : _____

One more… Determine the direction of equilibrium shift if sodium hydroxide is added to the above system.
![Equilibrium Graphs Equilibrium Concentration [CO] or [H 2 O] [CO 2] or [H 2] Equilibrium Graphs Equilibrium Concentration [CO] or [H 2 O] [CO 2] or [H 2]](http://slidetodoc.com/presentation_image_h2/9ae1f359a461a35c553fb6e20e8ed799/image-8.jpg)
Equilibrium Graphs Equilibrium Concentration [CO] or [H 2 O] [CO 2] or [H 2] Time Draw on this graph where equilibrium has occurred. Which is favoured? Products or Reactants?
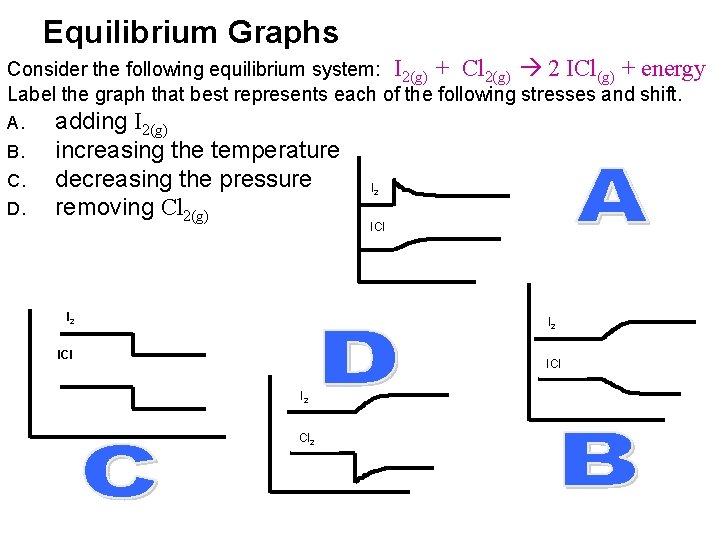
Equilibrium Graphs Consider the following equilibrium system: I 2(g) + Cl 2(g) 2 ICl(g) + energy Label the graph that best represents each of the following stresses and shift. A. B. C. D. adding I 2(g) increasing the temperature decreasing the pressure removing Cl 2(g) I 2 ICl I 2 Cl 2
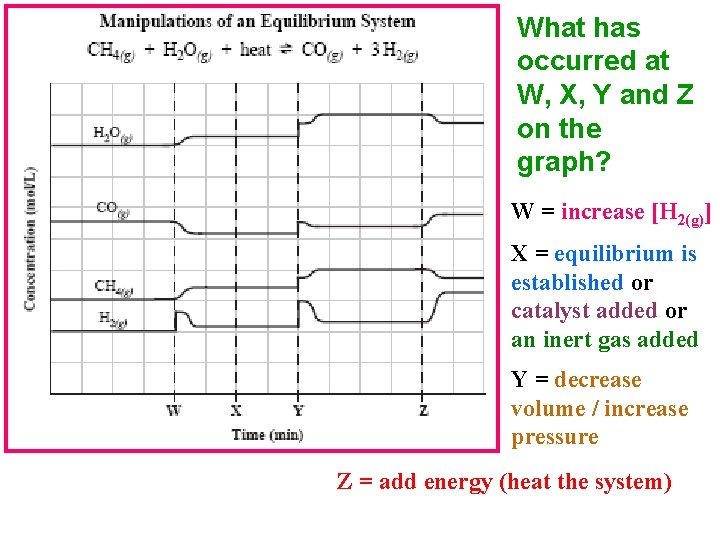
What has occurred at W, X, Y and Z on the graph? W = increase [H 2(g)] X = equilibrium is established or catalyst added or an inert gas added Y = decrease volume / increase pressure Z = add energy (heat the system)

Determining the Equilibrium Constant

Equilibrium Constants reveal info about reverse reaction too • Given that K = 0. 46 for this • What would the K be for: K for the reverse reaction is …
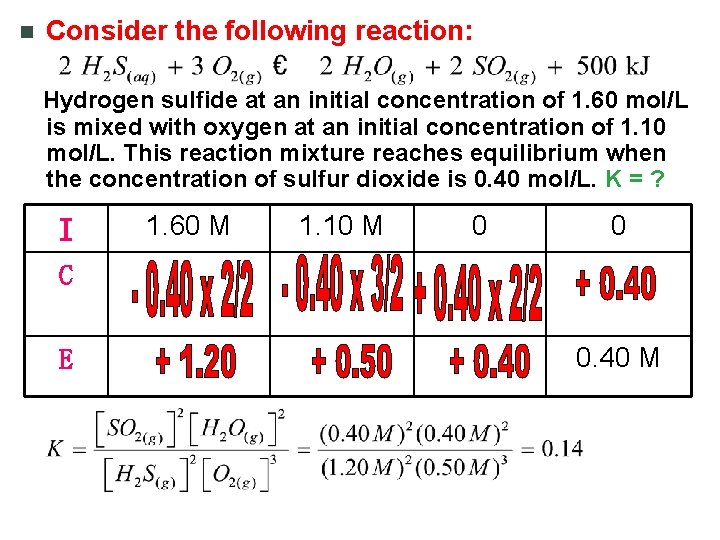
n Consider the following reaction: Hydrogen sulfide at an initial concentration of 1. 60 mol/L is mixed with oxygen at an initial concentration of 1. 10 mol/L. This reaction mixture reaches equilibrium when the concentration of sulfur dioxide is 0. 40 mol/L. K = ? I 1. 60 M 1. 10 M 0 0 C E 0. 40 M

Acids and Bases n Acid – Base Definitions Arrhenius (Chem 20) Acids – ionize into H+ and the negative ion Bases – dissociate into OH- and the positive ion Arrhenius modified (chem 20) Bronsted. Lowry Acids – react with water to form H 3 O+ Acids – proton donors Bases – react with water to form OH- Bases – proton acceptors
![Acid-Base Calculations n n n n p. H = -log[H 3 O+] = 2 Acid-Base Calculations n n n n p. H = -log[H 3 O+] = 2](http://slidetodoc.com/presentation_image_h2/9ae1f359a461a35c553fb6e20e8ed799/image-15.jpg)
Acid-Base Calculations n n n n p. H = -log[H 3 O+] = 2 nd log (-p. H) p. OH = -log[OH-] = 2 nd log (-p. OH) p. H + p. OH = 14 Kw = [H 3 O+] • [OH-] Kw = Ka • Kb n n Ka = [H 3 O+]2 [acid] Kb = [OH-]2 [base] % = _[H 3 O+]_ x 100 [initial acid] % = _[OH-]_ x 100 [initial base]

Calculate the p. H of a barium hydroxide solution with a concentration of 0. 25 mol/L. p. H = -log 2. 0 x 10 -14 mol/L = 13. 70

Calculate the p. H of a 0. 25 mol/L solution of lactic acid. I 0. 25 M 0 0 C -X +X +X E 0. 25 M X X

Calculate the p. H of a 0. 25 mol/L solution of sodium carbonate. I 0. 25 M 0 0 C -X +X +X E 0. 25 M X X How do we get Kb?

Calculate the percent dissociation for a 0. 45 mol/L solution of potassium nitrite with a p. H of 12. 74.

Write all the reactions (including the net) for the complete neutralization of sodium carbonate with hydrobromic acid, using methyl orange indicator. Let’s draw a p. H curve sketch of the titration information above…label endpoints and buffering areas
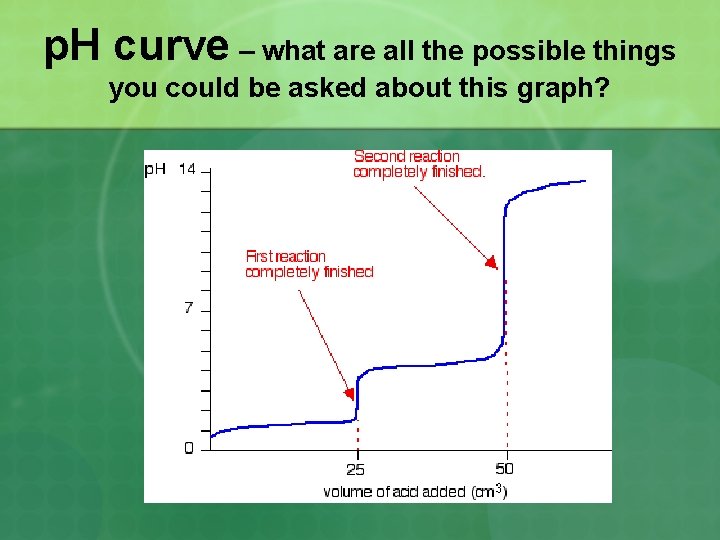
p. H curve – what are all the possible things you could be asked about this graph?
- Slides: 21