Equilibria Polyprotic weak acids For example phosphoric acid

Equilibria: Polyprotic weak acids For example, phosphoric acid, H 3 PO 4 H 2 PO 4 -1 + H+1 KA 1= 7. 5*10 -3 H 2 PO 4 -1 HPO 4 -2 + H+1 KA 2= 6. 2*10 -8 HPO 4 -2 PO 4 -3 + H+1 KA 3= 4. 8*10 -13 H+1 T = H+11 + H+12 + H+13 ~ H+11

Equilibria: Salts Illustrated using a simple =1/-1 salt There are 4 reactions to consider. 1. 2. 3. 4. Cat. An Cat+1 + An-1 Cat+1 + H 2 O Cat. OH + H+1 KA = KW/KB An-1 + H 2 O Han + OH-1 KB = KW/KA H 2 O H+1 + OH-1 KW
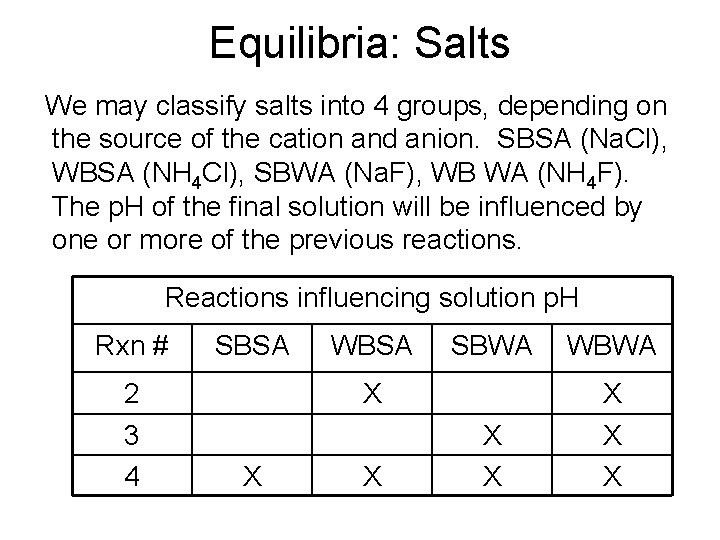
Equilibria: Salts We may classify salts into 4 groups, depending on the source of the cation and anion. SBSA (Na. Cl), WBSA (NH 4 Cl), SBWA (Na. F), WB WA (NH 4 F). The p. H of the final solution will be influenced by one or more of the previous reactions. Reactions influencing solution p. H Rxn # SBSA 2 3 4 WBSA SBWA X X X WBWA X X X

Equilibria: Salts WAWB salts are especially interesting. The p. H is influenced by both the cation and anion hydrolysis reactions. 1. Cat+1 + H 2 O Cat. OH + H+1 KA = KW/KB 2. An-1 + H 2 O Han + OH-1 KB = KW/KA The p. H of the final solution will depend on the size of KA & KB. KA > KB, acidic, etc.

Equilibria: Titrations 1. 2. 3. 3. If one titrates an acetic acid solution with a sodium hydroxide solution. There are 4 stages in the titration. Only acetic acid, p. H = -log(CAKA)/2 Buffer, p. H = p. KA + log [A-] [HA] WASB salt, p. OH = -log(CAKB)/2 note, must find new “acid” concentration Strong base p. H = 14 - p. OH

Equilibria: Solubility & Solubility Products If a salt is “insoluble” or sparingly soluble, one may write an equilibrium expression for the reaction with water. MXAY(S) XM+Y(aq) + YA-X(aq) KSP = [M+Y]X[A-X]Y e. g. Ca. F 2(S) Ca+2(aq) + 2 F-1(aq) KSP = [Ca+2]1[F-1]2 KSP = 4. 2*10 -11

Equilibria: Solubility & Solubility Products We prepare a solution which is 1. 0*10 -4 M in copper(I) and 2. 0*10 -3 M in lead(II). To this solution we slowly add sodium iodide. Which will precipitate first, Cu. I or Pb. I 2? The KSP’s are respectively, 5. 3*10 -12 and 1. 4*10 -8.
- Slides: 7