Equation of State Real Gas Relationships Relates PVT













- Slides: 15

Equation of State Real Gas Relationships

• Relates PVT properties of a pure substance/mixture via Theoratical and empirical relations • Source of information: computer databases • Effective Eq. of state: experimental data with good precision


Van-der Waals Equation • • Simple, explains computational problems. Also illustrates Theoretical developments • • Fortan computer Programs for non linear equations One Code: Newton Method, Other Minimization


Critical Temperature and Pressure • The highest temperature at which a species can coexist in two phases (liquid vapor) is the critical temperature of that species, Tc, and the corresponding pressure is the critical pressure, Pc. • A substance at Tc and Pc is said to be at its critical state. • A vapor is a gaseous species below its critical temperature, and a gas is a species above its critical temperature at a pressure low enough for the species to be more like a vapor than a liquid (i. e. , a density closer to 1 g/L than 1000 g/L). • Substances at temperatures above Tc and pressures above Pc are referred to as supercritical fluids.

Cubic Equation of State • Fit the experimental data with as few constants in equation as possible • Concise summary of a large of experimental data • Accurate interpolation between experimental data points • Provide a continuous function to facilitate calculation of physical properties involving differentiation and integration. • Point of departure for the treatment of thermodynamic properties of mixtures.

Virial Equation of state • A virial equation of state expresses the quantity PV/ RT as a power series in the inverse of specific volume: • B, C, and D are functions of temperature …known as the second, third, and fourth virial coefficients, • Theoretical basis in statistical mechanics • procedures for estimating the coefficients are not well developed, those beyond B. • The ideal gas equation of state is obtained if B=C=D…

• For polar compounds (asymmetrical compounds with a nonzero dipole moment, such as water). • Can estimate V or P for a given T for a nonpolar species (one with a dipole moment close to zero, such as hydrogen and oxygen and all other molecularly symmetrical compounds). • Solution for P is straightforward. • For V, the equation can be rearranged into a quadratic and solved using the quadratic formula. • One of the two solutions is reasonable and the other is not and should be discarded; • if there is any doubt, estimate V from the ideal gas equation of state and accept the virial equation solution that comes closest to V ideal'

Steps Involved • Look up the critical temperature and pressure (Tc and Pc) for the species of interest • Pitzer acentric factor, w. a parameter that reflects the geometry and polarity of a molecule. • Calculate the reduced temperature, Tr = TITc. • Estimate B using the following equations • Substitute into Equation the values of B and whichever of the variables P and V is known and solve for the other variable

Cubic equations of state (SRK) • Empirical but have proved remarkably robust in describing a 'Wide variety of systems. • SRK equation of state is • Empirical functions of critical temperature and Pressure

Correlations

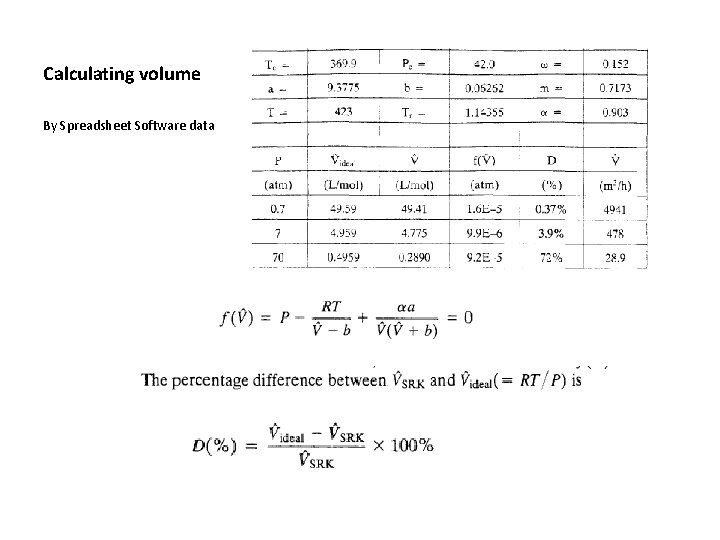
Calculating volume By Spreadsheet Software data


Considerations • SRK equation of state (and every other equation of state) is itself an • approximation. • All equations of state have parameters obtained by fitting empirical expressions to experimental PVT data. • The fit may be excellent in the temperature and pressure ranges where the data were obtained but may be terrible elsewhere. • Always try to ascertain the region of validity of any equation of state intend to be used. • No assurance of the accuracy of the equation for condition far from acceptable region.