Epidemiology of Poliomyelitis Dr Ali Jafar Abedi MD

Epidemiology of Poliomyelitis Dr. Ali Jafar Abedi MD, DCH 1

What is polio disease? Polio (also called Poliomyelitis) is a highly infectious disease caused by a virus The virus invades the nervous system and can cause permanent paralysis Polio is spread through person-to-person contact and can spread rapidly through a community Most infected people (90%) have no symptoms or very mild symptoms However, one in 200 infections leads to permanent paralysis (can’t move parts of the body) and even death www. immune. org. nz 2

Poliomyelitis The words polio (grey) and myelon (marrow, indicating the spinal cord) are derived from the Greek. It is the effect of poliomyelitis virus on the spinal cord that leads to the classic manifestation of paralysis. 3

The disease of poliomyelitis has a long history. The first example may even have been more than 3000 years ago. An Egyptian stele dating from the 18 th Egyptian dynasty (1580 - 1350 BCE) shows a priest with a deformity of his leg characteristic of the flaccid paralysis typical of poliomyelitis. . First described by Michael Underwood in 1789 4

Introduction A viral infection most often recognized by acute onset of flaccid paralysis. Infection with poliovirus results in a spectrum of clinical manifestations from inapparent infection to non-specific febrile illness, aseptic meningitis, paralytic disease, and death. Poliomyelitis is a highly infectious disease caused by three serotypes of poliovirus. 5

Two phases of acute poliomyelitis can be distinguished: a non-specific febrile illness (minor illness) followed, in a small proportion of patients, by aseptic meningitis and/or paralytic disease (major illness). The ratio of cases of inapparent infection to paralytic disease ranges from 100: 1 to 1000: 1. 6
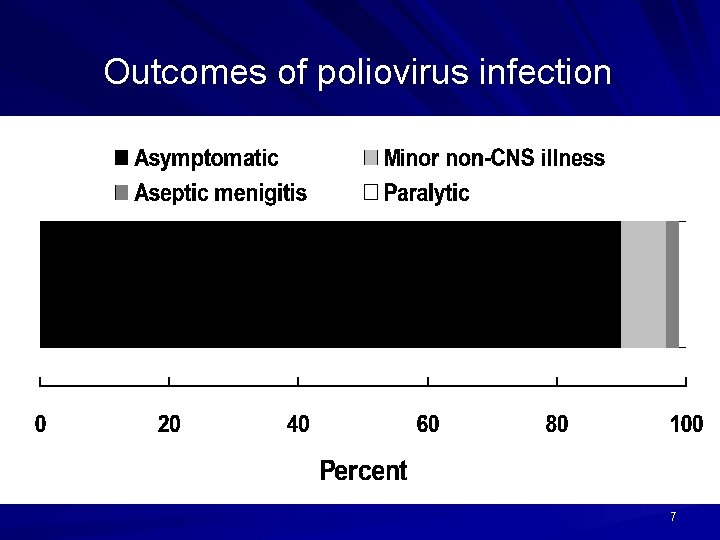
Outcomes of poliovirus infection 7

Epidemiological pattern The epidemiological pattern of polio depends upon the degree of the socioeconomic development and health care services of a country. The pattern of the disease has been considerably modified by widespread immunization. 8

Causative organism Poliovirus: belongs to “Picorna” viruses which are small RNA-containing viruses. Polioviruses have three antigenically distinct types, giving no cross immunity: – Type I: “Leon”; the commonest in epidemics – Type II: “Berlinhide”; the prevailing type in endemic areas. – Type III: “Lansing”; occasionally causes epidemics. Polioviruses are relatively resistant and survive for a long time under suitable environmental conditions, but are readily destroyed by heat (e. g. pasteurization of milk, and chlorination of water). 9

Types of polioviruses OPV related Wild VAPP** VDPVs* • • 99% reduction in cases of wild poliovirus since 1988 Type 1 (341 cases as of 20 November 2013†) Type 2 (eliminated worldwide in 1999) Type 3 (none detected since November 2012) • Vaccine-associated paralytic poliomyelitis (VAPP)** • Estimated ~250 -500 globally per year • Type 2 accounts for about 40% of VAPP • • Vaccine derived polioviruses (VDPV) Most are circulating VDPVs (c. VDPVs)* ~49 -184 per year since 2008 (through 20 Nov 2013) Type 2 c. VDPVs account for 97% of c. VDPVs † More up-to-date numbers can be found at http: //www. polioeradication. org/Dataandmonitoring/Poliothisweek. aspx *Other extremely rare VDPVs include primary immunodeficiency VDPVs (i. VDPVs) and ambiguous VDPVs (a. VDPVs) **Refers to spontaneous reversion to neurovirulence of one of the attenuated viruses in OPV. VAPP occurs in OPV recipients or their close contacts in contrast to c. VDPVs which are widely transmitted in a community and are not likely to be related to contact with a recent vaccine recipient. 10

Reservoir of infection Man is the only reservoir of infection of poliomyelitis. Man: cases and carriers Cases: all clinical forms of disease Carriers: all types of carriers (e. g. incubatory, convalescent, contact and healthy) except chronic type. In endemic areas, healthy carriers are the most frequent type encountered. 11

Foci of infection Pharynx: the virus is found in the oropharyngeal secretions. Small intestine: the virus finds exit in stools. 12

Modes of transmission Since foci of infection are throat and small intestines, poliomyelitis spreads by two routes: Oral-oral infection: direct droplet infection Faeco-oral infection: – Food-borne (ingestion) infection through the ingestion of contaminated foods. Vehicles include milk, water, or any others that may be contaminated by handling, flies, dust…. – Hand to mouth infection. (polio virus has the ability to survive in cold environments. Overcrowding and poor sanitation provide opportunities for exposure to infection. ) 13
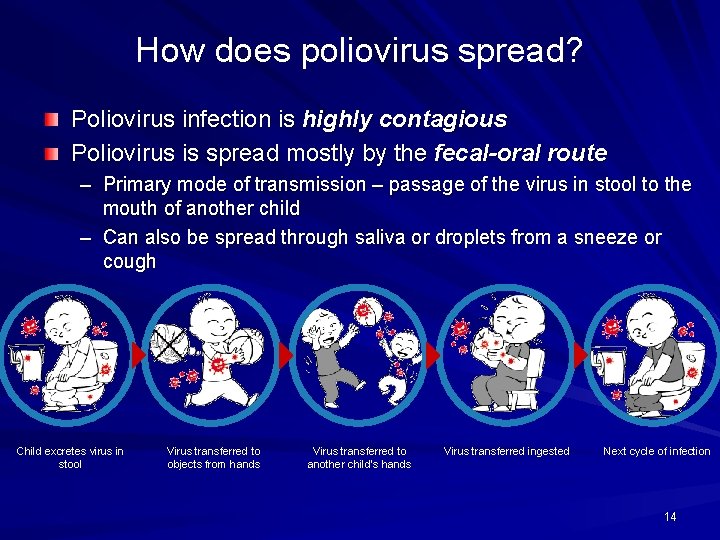
How does poliovirus spread? Poliovirus infection is highly contagious Poliovirus is spread mostly by the fecal-oral route – Primary mode of transmission – passage of the virus in stool to the mouth of another child – Can also be spread through saliva or droplets from a sneeze or cough Child excretes virus in stool Virus transferred to objects from hands Virus transferred to another child’s hands Virus transferred ingested Next cycle of infection 14

Period of infectivity Contact and healthy carriers: about 2 weeks Cases: the cases are most infectious 7 to 10 days before and after the onset of symptoms. In the feaces, the virus is excreted commonly for 2 to 3 weeks, sometimes as long as 3 to 4 months. In polio cases, infectivity in the pharyngeal foci is around one week, and in the intestinal foci 6 -8 weeks. Incubation Period: 7 -14 days 15

Susceptibility Age: more than 95% reported in infancy and childhood with over 50% of them in infancy. Sex: no sex ratio differences, but in some countries, males are infected more frequently than females in a ratio 3: 1. Risk factors: (provocative factors of paralytic polio in individuals infected with polio virus): fatigue, trauma, intramuscular injections, operative procedures, pregnancy, excessive muscular exercise… Immunity: The maternal antibodies gradually disappear during the first 6 months of life. Immunity following infection is fairly solid, although infection with other types of polio virus can still occur. 16

Sequelae of polio infection Polio infection Inapparent infection Clinical poliomyelitis Abortive polio (minor illness) Involvement of CNS (major illness) Paralytic polio Non-paralytic polio Spinal polio Bulbar polio Bulbospinal polio 17

Inapparent infection Incidence is more than 100 to 1000 times the clinical cases. No clinical manifestations, but infection is associated with acquired immunity, and carrier state. 18

Clinical poliomyelitis Abortive polio (minor illness): I. The majority of clinical cases are abortive, with mild systemic manifestations for one or two days only, then clears up giving immunity. Some abortive cases may be so mild to pass unnoticed. Manifestations: – – – Moderate fever Upper respiratory manifestations: pharyngitis and sore throat Gastrointestinal manifestations: vomiting, abdominal pain, and diarrhea. 19

Clinical poliomyelitis (cont. ) II. Involvement of the CNS (major illness): Affects a small proportion of the clinical cases, and appears few days after subsidence of the abortive stage. It takes two forms: nonparalytic and paralytic polio. Nonparalytic polio is manifested by fever, headache, nausea, vomiting, and abdominal pain. Signs of meningeal irritation (meningism), and aseptic meningitis (pain and stiffness in the neck back and limbs) may also occur. The case either recovers or passes to the paralytic stage, and here the nonpralytic form is considered as a “preparalytic stage”. 20
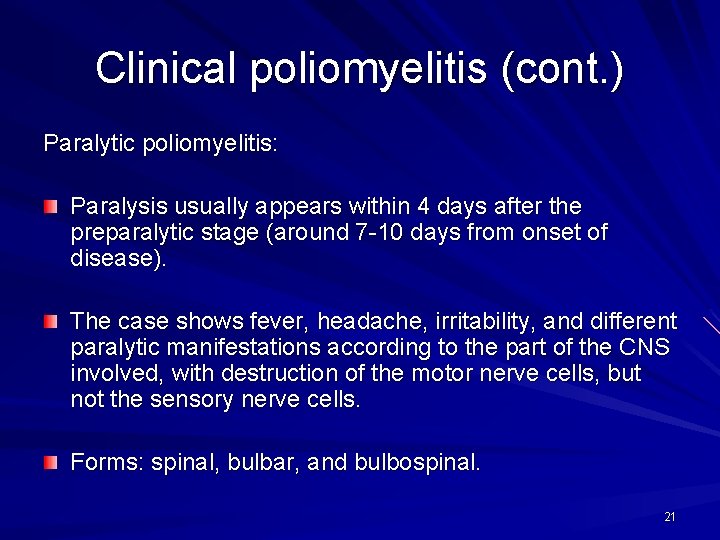
Clinical poliomyelitis (cont. ) Paralytic poliomyelitis: Paralysis usually appears within 4 days after the preparalytic stage (around 7 -10 days from onset of disease). The case shows fever, headache, irritability, and different paralytic manifestations according to the part of the CNS involved, with destruction of the motor nerve cells, but not the sensory nerve cells. Forms: spinal, bulbar, and bulbospinal. 21

Spinal polio Different spinal nerves are involved, due to injury of the anterior horn cells of the spinal cord, causing tenderness, weakness, and flaccid paralysis of the corresponding striated muscles. The lower limbs are the most commonly affected. 22

Bulbar polio Nuclei of the cranial nerves are involved, causing weakness of the supplied muscles, and maybe encephalitis. Bulbar manifestations include dysphagia, nasal voice, fluid regurgitation from the nose, difficult chewing, facial weakness and diplopia Paralysis of the muscles of respiration is the most serious life-threatening manifestation. 23

Bulbospinal polio Combination of both spinal and bulbar forms 24

Complications and case fatality Respiratory complications: pneumonia, pulmonary edema Cardiovascular complications: myocarditis, cor pulmonale. Late complications: soft tissue and bone deformities, osteoporosis, and chronic distension of the colon. Case fatality: varies from 1% to 10% according to the form of disease (higher in bulbar), complications and age ( fatality increases with age). 25

Case definition The following case definition for paralytic poliomyelitis has been approved by CDC (1997) Clinical case definition Acute onset of a flaccid paralysis of one or more limbs with decreased or absent tendon reflexes in the affected limbs, without other apparent cause, and without sensory or cognitive loss. 26

Case classification Probable: A case that meets the clinical case definition. Confirmed: A case that meets the clinical case definition and in which the patient has a neurologic deficit 60 days after onset of initial symptoms, has died, or has unknown follow-up status. Confirmed cases are then further classified based on epidemiologic and laboratory criteria. Only confirmed cases are included in the Morbidity and Mortality Weekly Report (MMWR). Indigenous case: Any case which cannot be proved to be imported. Imported case: A case which has its source outside the country. A person with poliomyelitis who has entered the country and had onset of illness within 30 days before or after entry 27

HOT CASE A case of AFP with any of the following set of conditions Ø Age < 5 year plus H/O fever at onset plus asymmetrical proximal paralysis. Ø Age < 5 year with rapidly progressive paralysis leading to bulbar involvement (cranial nerves affected) & death. Ø Any case which in the opinion of SMO/DIO looks like polio. 28

Diagnosis The diagnosis of paralytic poliomyelitis is supported by: (i) clinical course, (ii) virological testing, and (iii) residual neurologic deficit 60 days after onset of symptoms. Laboratory testing, such as the measurement of antibodies (especially pre- and post-onset of paralysis), and other studies, such as magnetic resonance imaging, electromyography, and/or nerve conduction tests, can help strengthen or exclude the diagnosis of poliomyelitis. 29

WHO uses a sensitive screening case definition for the identification of AFP cases and for investigation of any case of AFP in a person younger than 15 years or in a person of any age in whom poliomyelitis is suspected. However, virological examination is essential for confirmation of the diagnosis of poliomyelitis; this involves detection of poliovirus from the stools of patients with AFP and further characterization of the isolated poliovirus to determine whether it is vaccine-associated, vaccine-derived or wild virus. 30

Molecular diagnostics such as polymerase chain reaction are used to differentiate WPV, VDPV, and Sabin-like poliovirus. In addition all discordant poliovirus isolates are partially sequenced to determine their origin and relatedness to other isolates. According to the laboratory results and review by national polio expert committees, cases are further classified as confirmed, polio-compatible, or polio-negative. The AFP surveillance is supplemented by environmental surveillance which involves testing sewage or other environmental samples for the presence of poliovirus 31

Diagnosis and laboratory testing (cont. ) Serologic testing A four-fold titer rise between the acute and convalescent specimens suggests poliovirus infection. Cerebrospinal fluid (CSF) analysis The cerebrospinal fluid usually contains an increased number of leukocytes—from 10 to 200 cells/mm 3 (primarily lymphocytes) and a mildly elevated protein, from 40 to 50 mg/100 ml. 32

Prevention General prevention: Health promotion through environmental sanitation. Health education (modes of spread, protective value of vaccination). 33

Prevention Seroprophylaxis by immunoglobulins: Not a practical way of giving protection because it must be given either or before or very shortly after exposure to infection. (0. 3 ml/kg of body weight). 34

an armoury of five different vaccines to stop polio transmission: Oral polio vaccine (OPV) Monovalent oral polio vaccines (m. OPV 1 and m. OPV 3) Bivalent oral polio vaccine (b. OPV) Inactivated polio vaccine (IPV) 35

prevention Active immunization: – Salk vaccine (intramuscular polio trivalent killed vaccine). – Sabin vaccine (oral polio trivalent live attenuated vaccine). 36

Inactivated Polio Vaccine Contains 3 serotypes of vaccine virus Grown on monkey kidney (Vero) cells Inactivated with formaldehyde Contains 2 -phenoxyethanol, neomycin, streptomycin, polymyxin B 37

Oral Polio Vaccine Contains 3 serotypes of vaccine virus Grown on monkey kidney (Vero) cells Contains neomycin and streptomycin Shed in stool for up to 6 weeks following vaccination 38

Salk versus Sabin vaccine IPV (Salk) killed formolised virus Given SC or IM Induces circulating antibodies, but not local (intestinal immunity) Prevents paralysis but does not prevent reinfection Not useful in controlling epidemics More difficult to manufacture and is relatively costly Does not require stringent conditions during storage and transportation. Has a longer shelf life. OPV (Sabin) live attenuated virus given orally immunity is both humoral and intestinal. induces antibody quickly Prevents paralysis and prevents reinfection Can be effectively used in controlling epidemics. Easy to manufacture and is cheaper Requires to be stored and transported at subzero temperatures, and is damaged easily. 39

Polio Vaccination of Unvaccinated Adults IPV Use standard IPV schedule if possible (0, 1 -2 months, 6 -12 months) May separate doses by 4 weeks if accelerated schedule needed 40

Polio Vaccine Adverse Reactions Rare local reactions (IPV) No serious reactions to IPV have been documented Paralytic poliomyelitis (OPV) 41

What is vaccine-derived polio? Vaccine-derived polioviruses (VDPVs) are rare strains of poliovirus that have genetically mutated from the strain contained in the oral polio vaccine. 42

VDPV can Cause Sustained Person-to-Person Spread and Reintroduction of Poliovirus VDPVs are classified into 3 categories: – Circulating VDPVs (c. VDPVs) Emerge in areas with inadequate OPV coverage May lead to sustained person-to-person spread – Immunodeficient-associated VDPVs (i. VDPVs) Isolated from persons with primary immunodeficiencies who exhibit prolonged VDPV infection after OPV exposure Potential source of PV reintroduction in the future; they may excrete virus for up to 20 years – Ambiguous VDPVs (a. VDPVs) Clinical isolates from persons with no known immunodeficiency or Environmental isolates with unidentified source WHO. WER, 2006. Mc. Lennan, et al. Lancet, 2000

VAPP: Rare But Serious Vaccine-Associated Paralytic Polio: – Defined as those cases of AFP from whose stool samples, vaccine-related poliovirus but no wild polio virus is isolated – Caused by mutation of vaccine virus during replication in the gut of vaccinee (reversion to neurovirulence) VAPP is indistinguishable from naturally occurring polio Same incubation period, range of severity and Case Fatality Rate May affect both vaccinees & close contacts. 1. Sutter et al. Vaccines. 2008. 2. Paul. Vaccine, 2004. 3. John. Bull of the WHO, 2004. 44

B. Control of patient, contacts and the immediate environment: 1) Report to local health authority: Obligatory case report of paralytic cases as a Disease under surveillance by WHO, Class 1. 2) Isolation: Enteric precautions in the hospital for wild virus disease; of little value under home conditions because many household contacts are infected before poliomyelitis has been diagnosed. 3) Concurrent disinfection: Throat discharges, feces and articles soiled therewith. Terminal cleaning. 4) Quarantine: Of no community value. 45

5) Protection of contacts: Immunization of familial and other close contacts is recommended but may not contribute to immediate control; the virus has often infected susceptible close contacts by the time the initial case is recognized. 6) Investigation of contacts and source of infection: Occurrence of a single case of poliomyelitis due to wild poliovirus must be recognized as a public health emergency prompting immediate investigation and planning for a large-scale response. A thorough search for additional cases of AFP in the area around the case assures early detection, facilitates control and permits appropriate treatment of unrecognized and unreported cases. 7) Specific treatment: None; however, Physical therapy is used to attain maximum function after paralytic poliomyelitis. 46

C. Epidemic measures: In any country, a single case of poliomyelitis must now be considered a public health emergency, requiring an extensive supplementary immunization response over a large geographic area. 47

D. Disaster implications: Overcrowding of non-immune groups and collapse of the sanitary infrastructure pose an epidemic threat. 48

E. International measures: Poliomyelitis is a Disease under surveillance by WHO and is targeted for eradication by 2005. National health administrations are expected to inform WHO immediately of individual cases and to supplement these reports as soon as possible with details of the nature and extent of virus transmission. Planning a large-scale immunization response must begin immediately and, if epidemiologically appropriate, in coordination with bordering countries. 49

E. International measures (cont. ): Once a wild poliovirus is isolated, molecular epidemiology can often help trace the source. Countries should submit monthly reports on case of poliomyelitis AFP cases and AFP surveillance performance to their respective WHO offices. International travelers visiting areas of high prevalence must be adequately immunized. 50

51

Polio Eradication Last case in United States in 1979 Western Hemisphere certified polio free in 1994 Last isolate of type 2 poliovirus in India in October 1999 Global eradication goal 52

WHY POLIO IS A CANDIDATE FOR ERADICATION ? MAN IS THE ONLY RESERVIOR NO LONG TERM CARRIER STATE ROUTE OF TRANSMISSION IS FAECOORAL HALF LIFE OF EXCRETED VIRUS IN SEWAGE SAMPLE IN TROPICAL CLIMATE LIKE INDIA IS 48 HOURS. POTENT AND EFFECTIVE VACCINE. 53
![FOUR KEY STRATEGIES FOR POLIO ERADICATION RI-PROGRAMME [ UIP ] MASS IMMUNIZATION(PPI) CAMPAIGNS APF FOUR KEY STRATEGIES FOR POLIO ERADICATION RI-PROGRAMME [ UIP ] MASS IMMUNIZATION(PPI) CAMPAIGNS APF](http://slidetodoc.com/presentation_image_h2/66958bab8f4775628bb7d719032818d9/image-54.jpg)
FOUR KEY STRATEGIES FOR POLIO ERADICATION RI-PROGRAMME [ UIP ] MASS IMMUNIZATION(PPI) CAMPAIGNS APF SURVEILLANCE MOPING UP IN FOCAL AREAS 54
55
56

Surveillance Acute Flaccid Paralysis (AFP) surveillance Nationwide AFP (acute flaccid paralysis) surveillance is the gold standard for detecting cases of poliomyelitis. 57

The four steps of surveillance are: finding and reporting children with acute flaccid paralysis (AFP) transporting stool samples for analysis isolating and identifying poliovirus in the laboratory mapping the virus to determine the origin of the virus strain. 58

WHY OPV ? ALSO KNOWN AS SABIN VACCINE POTENT LIVE VACCINE GIVES GUT IMMUNITY GIVES HERD IMMUNITY- INTERRUPT’s TRANSMISSION CYCLE EASY TO ADMINISTER COST EFFECTIVE 59

WHAT IS PULSE POLIO ? TO IMMUNIZE ALL THE KIDS< 5 YRS NATION WIDE ON A SINGLE DAY IN THE SHORTEST POSSIBLE TIME WITH OPV & THAT THE ENVIRONMENT WILL GET SATURATED WITH THE VACCINE VIRUS SO THAT IT WILL REPLACE THE WILD VIRUS AND THUS INTERUPT THE TRANSMISSION OF WILD VIRUS. 60

WHY AFP SURVEILLANCE INSTEAD OF POLIO SURVEILLANCE ? SURVEILLANCE OF A POLIO CASE ALONE IS NOT SUFFICIENT BECAUSE IT IS IMPOSSIBLEE TO PRECISELY IDENTIFY ALL CASES OF POLIO CLINICALLY DUE TO CONFUSING AND AMBIGUOUS CLINICAL SIGNS AND VARIABLE CLINICAL KNOWLEDGE & SKILLS OF DOCTOR. CLINICALLY POLIO IN ACUTE STAGE, IS DIFFICULT TO DISTINGUISH FROM OTHER CAUSES OF ACUTE ONSET OF FLACCID PARALYSIS. ----61

The principle of AFP surveillance is to identify children below 15 years with the syndrome of Acute Flaccid Paralysis Acute-Rapid progression or short, brief duration Flaccid-Floppy or soft and yielding to passive stretching at any time during illness Paralysis–Loss of motor strength Severe loss of motor strength is called paralysis or plegia Paresis-indicates slight loss of motor strength 62

All cases of acute flaccid paralysis should be reported, irrespective of diagnosis, within 6 months of onset All cases with flaccid paralysis should be reported and their stools must be collected within 14 days of onset. If it is not possible to collect stool specimens within 14 days, the specimens should still be collected up to 60 days after onset of paralysis. 63

Include every case with: Current flaccid paralysis History of flaccid paralysis in the current illness Borderline or ambiguous clinical signs. 64

A case should not be included as AFP if there is no evidence of acute flaccid paralysis at the time of examination or anytime during the course of illness or if the onset of paralysis is more than 6 months prior to notification. 65

Causes of AFP Poliomyelitis Gullain Barre Syndrome Traumatic neuritis Transverse Myelitis Any other flaccid/lower motor presentation 66

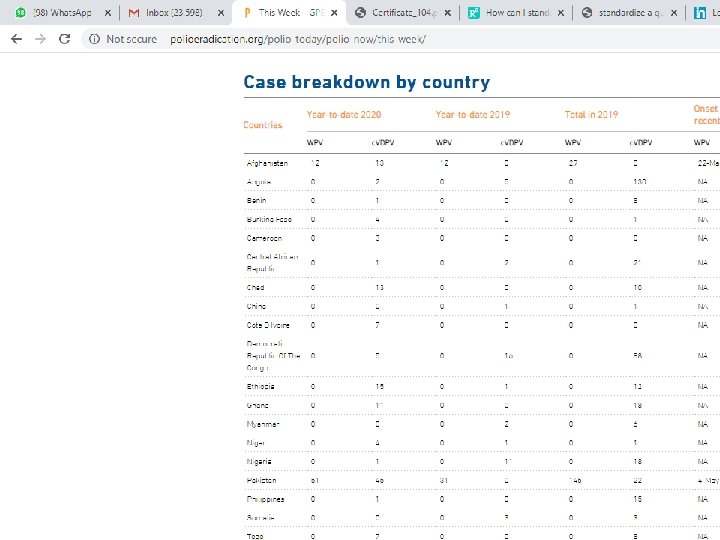
AFP SURVEILLANCE 1. 2. 3. 4. 5. STEPS FOR EACH AFP CASE INVESTIGATION 2 STOOL SPECIMENS, COLLECTED 24 HOURS APART, AND WITHIN 14 DAYS OF ONSET OF PARALYSIS SENT FOR CULTURES TO LAB TO ISOLATE POLIO VIRUS ORI ACTIVITY & SEARCH FOR MORE AFP CASES IN THE AREA 60 DAYS FOLLOW-UP EXAMINATION AFTER ONSET. 67
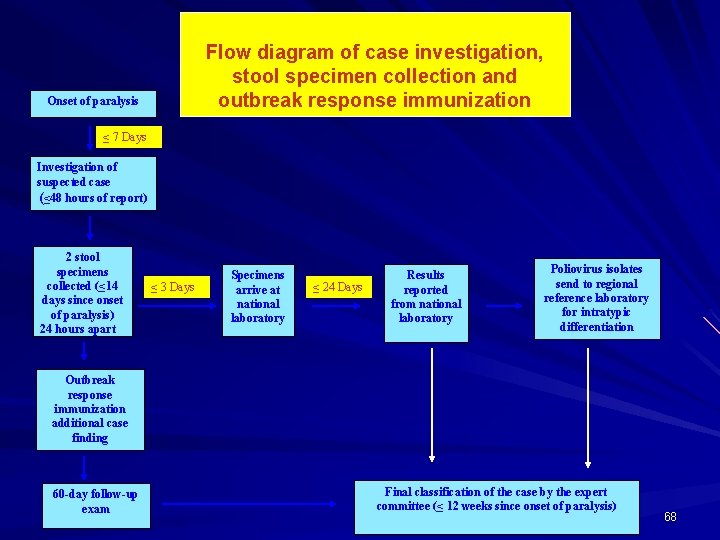
Flow diagram of case investigation, stool specimen collection and outbreak response immunization Onset of paralysis ≤ 7 Days Investigation of suspected case (≤ 48 hours of report) 2 stool specimens collected (≤ 14 days since onset of paralysis) 24 hours apart ≤ 3 Days Specimens arrive at national laboratory ≤ 24 Days Results reported from national laboratory Poliovirus isolates send to regional reference laboratory for intratypic differentiation Outbreak response immunization additional case finding 60 -day follow-up exam Final classification of the case by the expert committee (≤ 12 weeks since onset of paralysis) 68
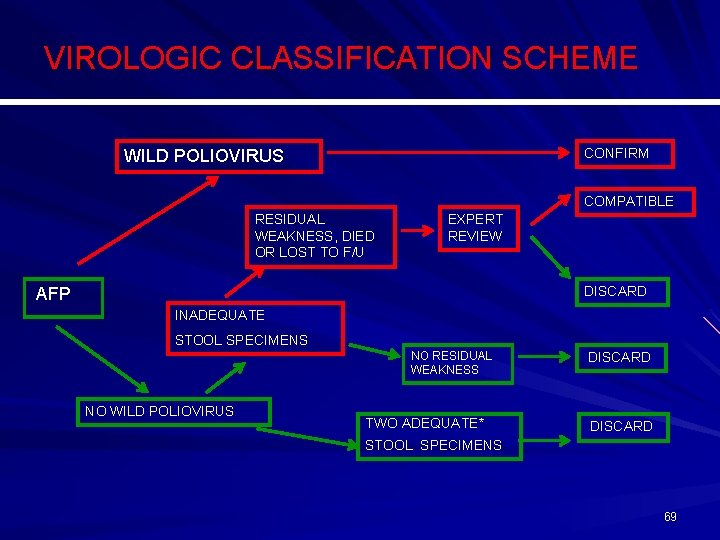
VIROLOGIC CLASSIFICATION SCHEME CONFIRM WILD POLIOVIRUS COMPATIBLE RESIDUAL WEAKNESS, DIED OR LOST TO F/U EXPERT REVIEW DISCARD AFP INADEQUATE STOOL SPECIMENS NO RESIDUAL WEAKNESS NO WILD POLIOVIRUS TWO ADEQUATE* DISCARD STOOL SPECIMENS 69

Surveillance indicators Indicator Completeness of reporting Minimum levels for certification standard surveillance At least 80% of expected routine reports should be received on time (inc. zero reports) The distribution of reporting sites should be representative of the geography and demography of the country At least one case of non-polio AFP should be detected Sensitivity of surveillance annually per 100 000 population aged less than 15 years. In endemic regions this rate should be two per 100 000. Completeness of case investigation All cases should have a full clinical and virological investigation with at least 80% of AFP cases having ‘adequate’ stool specimens collected. Completeness of followup At least 80% of AFP cases should have a follow-up examination for residual paralysis at 60 days after the 70 onset of paralysis

Environmental surveillance involves testing sewage or other environmental samples for the presence of poliovirus. Environmental surveillance often confirms wild poliovirus infections in the absence of cases of paralysis. 71

Systematic environmental sampling (e. g. in Egypt and Mumbai, India) provides important supplementary surveillance data. Ad-hoc environmental surveillance elsewhere (especially in polio-free regions) provides insights into the international spread of poliovirus. 72

Certification of polio eradication Certification is done for WHO Regions and not for individual countries WHO Regions that have been certified polio free: – Americas: 20 August 1994 – Western Pacific: 29 October 2000 – Europe: 21 June 2002 73

Certification of a region is considered only when – All countries in the area demonstrate the absence of wild poliovirus transmission for at least three consecutive years presence of certification standard surveillance global action plan for laboratory containment of wild poliovirus 74

Certification standard surveillance Non-polio AFP rate: ≥ 2 per 100, 000 population aged less than 15 years annually Adequate stool specimens : ≥ 80% All stool specimens tested for poliovirus at a WHOaccredited laboratory Additional Criteria – Investigation of AFP cases within 48 hours of initial notification: ≥ 80% – Timeliness of weekly AFP surveillance reports: ≥ 80% 75

Laboratory containment of WPV To minimize the risk of reintroduction of WPV into the community from a laboratory WHO action plan comprises three phases: – Phase 1: laboratory survey and inventory – Phase 2: global certification: implement appropriate biosafety measures – Phase 3: post global certification: more stringent, will be prepared when there is global strategy to stop OPV – For regional certification evidence that phase 1 has been implemented 76

Rukhsar Khatoon last case of WPV detected in India (Jan 2011), her mother Shabida Bibi in Shahapar village, WB 77

Tommorows lecture End game polio 78

Thank You 79
- Slides: 79