Epidemiology Clinical Trials Biostatistics National Lipid Association 2009

Epidemiology, Clinical Trials & Biostatistics National Lipid Association 2009 Master’s Course in Lipidology Kevin C. Maki, Ph. D. Provident Clinical Research Glen Ellyn, IL Diplomate, Accreditation Council for Clinical Lipidology

Epidemiology § The study of the distributions and determinants of diseases in human populations § Most epidemiologic investigations are observational, therefore cannot directly demonstrate causal relationships § Results useful for generating hypotheses about possible causal relationships

Risk Factors and Risk Markers § A risk factor or risk marker is a variable associated with risk for a disease § Risk factors are correlational and not necessarily causal

Hill’s Criteria of Causation § Established by A. B. Hill as a way to assess the link between smoking and lung cancer § Criteria to establish scientifically valid causal connections between a potential causal agent and a disease

Hill’s Criteria of Causation 1. Temporal Relationship • Exposure precedes the outcome 2. Strength of Association • Stronger associations more likely to be causal 3. Dose-Response Relationship • Greater exposure produces greater risk 4. Consistency • Results are replicated in multiple studies and populations

Hill’s Criteria of Causation 5. Biologic Plausibility • There is a plausible mechanism to account for the finding 6. Alternative Explanations • Other hypotheses have been excluded 7. Experiment • Supported by data from clinical trials or, if not practical, animal studies

Hill’s Criteria of Causation 8. Specificity • A single cause produced a specific effect • Not essential, e. g. , smoking is a risk factor for both CHD and lung cancer 9. Coherence with existing evidence • Supported by evidence from other types of investigation (animal, laboratory, etc. ) • Judgments about causality must consider the totality of the evidence

Epidemiology: Historical Perspective § Epidemiological methods were originally employed in the study of infectious disease outbreaks or “epidemics” § In 2 nd half of the 20 th century, epidemiological methods were extended to the study of chronic diseases, particularly atherosclerotic cardiovascular diseases, especially CHD § Populations with high CHD mortality rates had more fatty streaks and atherosclerotic lesions in their coronary arteries at autopsy Keys. Nutrition 1970; 13: 250 -252.

CVD Epidemiology: Historical Perspective § The Seven Countries Study showed that CHD mortality varied by as much as 10 -fold between countries § Average blood cholesterol levels were associated with CHD mortality § Dietary saturated fat and cholesterol intakes were closely associated with blood cholesterol levels • Helped establish the “Diet-Heart Hypothesis” Steinberg. J Lipid Res 2005; 46: 179 -190. Robertson et al. Am J Cardiol 1977; 39: 239 -243

The Framingham Heart Study § Initiated in 1948, one of the earliest large-scale investigations in cardiovascular epidemiology • Prevailing view was that CHD was an unavoidable consequence of aging § Measured many attributes of ~5, 000 residents in Framingham, MA • Subjects were followed over decades to assess characteristics associated with the development of CHD § Provided evidence that variation in CHD rates within a population could be predicted by several “risk factors” www. epi. umn. edu/cvdepi

Study Designs in Epidemiology § Descriptive § Case-control § Cohort § Intervention

Descriptive Studies § Case reports and case series • A single patient or group of patients with a similar diagnosis § Cross sectional studies (surveys) • Exposure and disease status assessed simultaneously • Provides a “snapshot” of a population • Unit of study may be individuals or groups • Example – blood cholesterol and CHD mortality in the Seven Countries Study
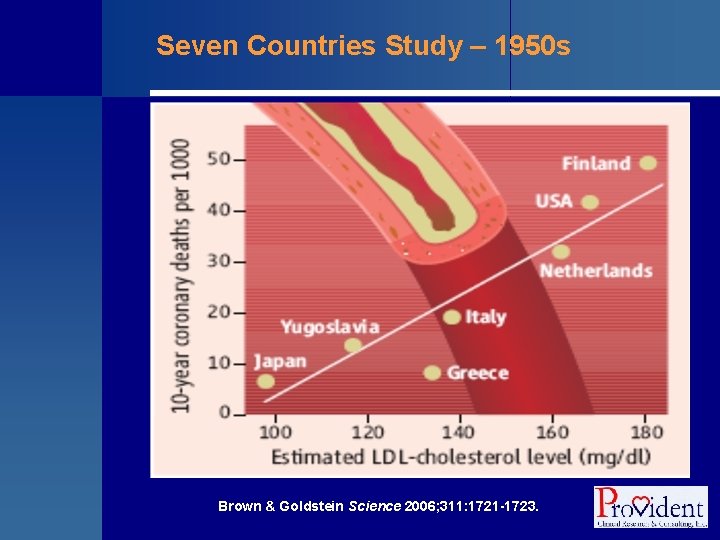
Seven Countries Study – 1950 s Brown & Goldstein Science 2006; 311: 1721 -1723.
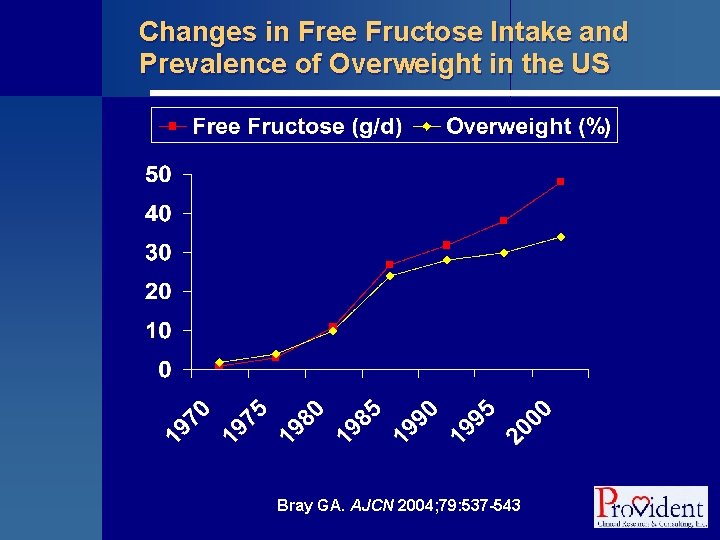
Changes in Free Fructose Intake and Prevalence of Overweight in the US Bray GA. AJCN 2004; 79: 537 -543
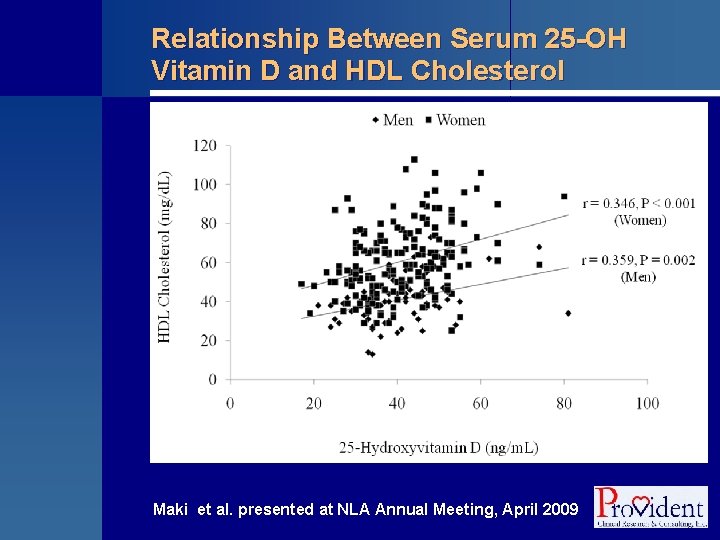
Relationship Between Serum 25 -OH Vitamin D and HDL Cholesterol Maki et al. presented at NLA Annual Meeting, April 2009

Case-Control Studies § Subjects selected based presence or absence of a particular disease • Cases versus controls § Groups compared based on a previous exposure (hypothesized risk factor) § Allows for the study of diseases with long latency and/or low incidence § Allows evaluation of multiple exposures and their relationships to the disease

INTERHEART: A Case Control Study § A case-control study of acute myocardial infarction (MI) in 52 countries • 15, 152 cases and 14, 820 controls enrolled § Evaluated the relationships between major risk factors identified in earlier epidemiologic investigations and MI § Nine potentially modifiable risk factors “accounted for” more than 90% of the variation in acute myocardial infarction among men and women Yusef et al. Lancet 2004; 364: 937 -52
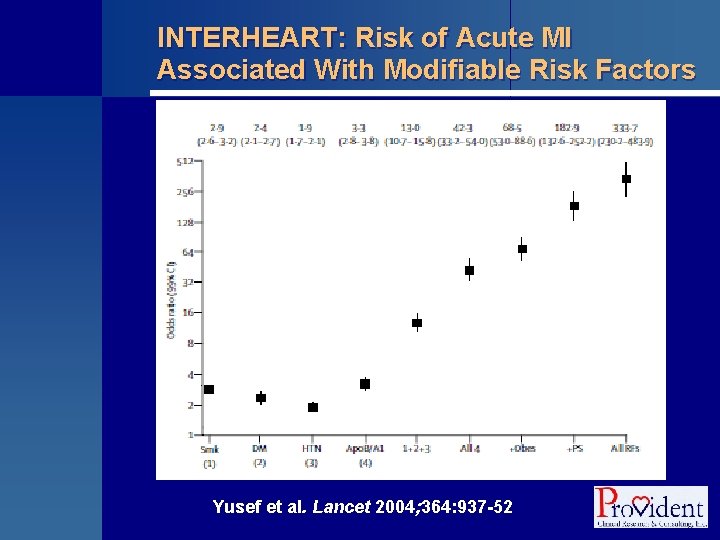
INTERHEART: Risk of Acute MI Associated With Modifiable Risk Factors Yusef et al. Lancet 2004; 364: 937 -52
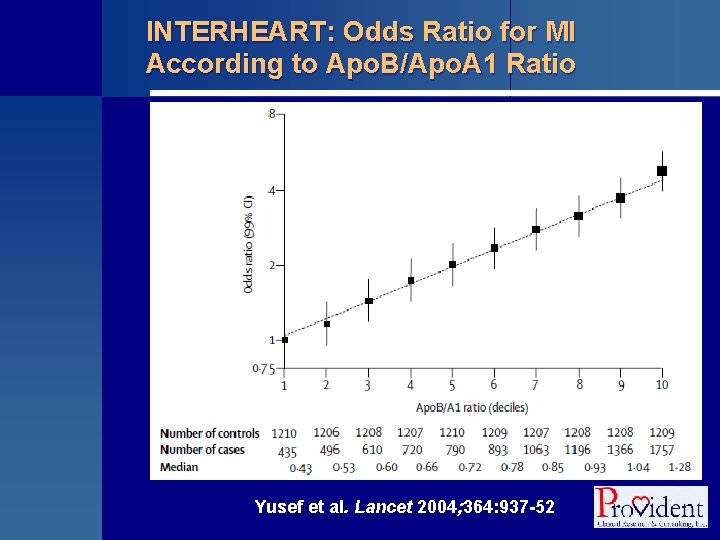
INTERHEART: Odds Ratio for MI According to Apo. B/Apo. A 1 Ratio Yusef et al. Lancet 2004; 364: 937 -52

Lifestyle-Heart Hypothesis Mozaffarian et al. Circulation 2008; 117; 3031 -3038

Cohort Studies § Subjects free of disease are followed to document disease incidence § Groups defined on the basis of exposure or lack of exposure to a suspected risk factor(s) § Allows for the examination of a single exposure for multiple outcomes • US Nurses’ Health Study § Typically requires large numbers of subjects followed over many years to accumulate events

Types of Observational Cohort Studies § Retrospective cohort studies • Exposure and outcomes of interest have already occurred before study initiation • Examples - managed care and employment physical studies (subjects followed forward in time, but to points in the past) § Prospective cohort studies • Exposures may or may not have occurred at the time of study initiation (e. g. , FHS) • Outcomes have not occurred at study initiation

PROCAM Study § Prospective Cardiovascular Münster Study § Initiated in 1979 • 4, 849 men followed over time to record incidence of CHD § Fasting triglycerides were an independent risk factor for CHD events, adding predictive information to LDL-C and LDL-C levels Assmann et al. Eur Heart J. 1998; 19(suppl M): M 8 -M 14.
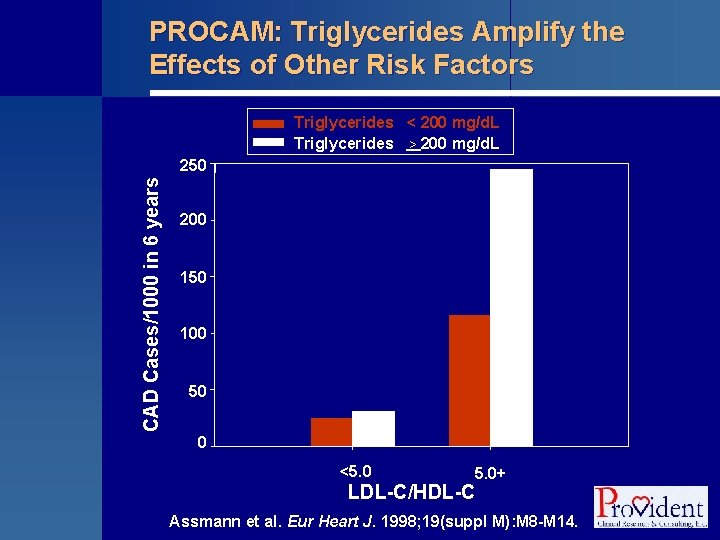
PROCAM: Triglycerides Amplify the Effects of Other Risk Factors Triglycerides < 200 mg/d. L Triglycerides > 200 mg/d. L CAD Cases/1000 in 6 years 250 200 150 100 50 0 <5. 0+ LDL-C/HDL-C Assmann et al. Eur Heart J. 1998; 19(suppl M): M 8 -M 14.

Nurses’ Health Study § Prospective cohort study § Established by Dr. Frank Speizer in 1976 to investigate the long term consequences of oral contraceptive use • Approximately 123, 000 nurses § Biannual questionnaires about diseases and health-related topics § Food frequency questionnaire initiated in 1980 § Many outcomes studied • CVD, diabetes, cancers http: //www. channing. harvard. edu/nhs/
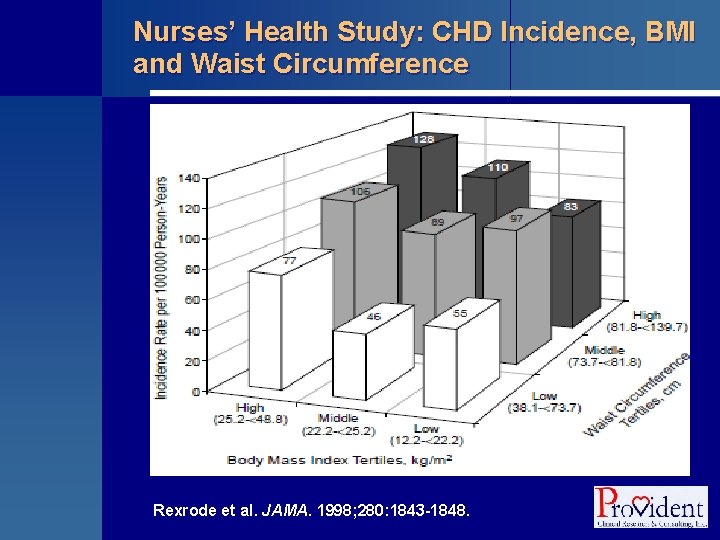
Nurses’ Health Study: CHD Incidence, BMI and Waist Circumference Rexrode et al. JAMA. 1998; 280: 1843 -1848.

Clinical Intervention Studies § The randomized, controlled trial is the “gold standard” for evaluation of medical interventions • Exposure is assigned • Least susceptible to known and unknown sources of bias and confounding § Validity and generalizability rely on: • Appropriate selection of study participants • Random subject assignment to treatments • Blinding and use of a placebo or sham intervention where practical

Issues Affecting the Validity and Generalizability of Clinical Trials § Results must be valid to be generalizable • Valid results are not always generalizable § Factors that can adversely affect validity • Poor subject compliance • Excessive or differential dropout • Incomplete blinding • Changes in treatment during the trial that confound the study results (e. g. , MR-FIT, FIELD)

Validity in Clinical Trials: Compliance § Subjects must adhere to the treatment regimen for results to be valid • Excessively complex trials may create undue subject burden and reduce compliance § Side effects may limit compliance and bias toward the null • Lipid Research Clinics Coronary Primary Prevention Trial (LRC-CPPT) • Cholestyramine vs. placebo • Protective effect of the drug was stronger in those with higher compliance

Validity in Clinical Trials: Dropout § May occur when treatment has unpleasant side effects or is difficult • Weight loss trials • Smoking cessation trials § Differential dropout • With active treatment due to side effects (e. g. , niacin) • In control treatment due to lack of benefits

Validity in Clinical Trials: Blinding § If a drug has side effects subjects will know they are on active treatment • Example: flushing from niacin treatment • Non-efficacious quantity of niacin was added to placebo so that subjects would experience mild flushing • Example: altered menstrual cycle on hormonal therapy (HERS) • Separate evaluators to assess gynecological and non-gynecological issues

Validity in Clinical Trials: Changes to Treatment § Multiple Risk Factor Intervention Trial (MR-FIT) • Standard care changed over 6 -8 yr follow-up • Narrowed the differences between the treatment arms in levels of several risk factors • Trial failed to show a significant benefit for CHD or total mortality MR-FIT Investigators. JAMA 1982; 248: 1465 -1477.

Interpretation of Study Results § Three main factors should be assessed during the interpretation of study results: 1. Chance • Was this a random chance finding? 2. Bias • Was there a non-random error that resulted in an incorrect estimate of the effect of the exposure? 3. Confounding • Is it possible that an observed association between a exposure and the response was due to the effects of differences other than the exposure under study?

Example of Possible Bias in a Clinical Trial § The Incremental Decrease in End Points through Aggressive Lipid Lowering (IDEAL) Study • Prospective, randomized, open-label, blinded end-point evaluation trial (PROBE design) • Subjects with a history of MI (n = 8888) were assigned to standard-dose simvastatin (20 mg) or high-dose atorvastatin (80 mg) and followed for an average of 4. 8 y § Compliance was higher (95% vs. 89%) and dropout was lower (7% vs. 14%) in the simvastatin group § Mean on-trial levels of LDL-C were 81 and 104 mg/d. L in the atorvastatin and simvastatin groups, respectively: a smaller difference than projected Pedersen et al. JAMA 2005; 294: 2437 -2445
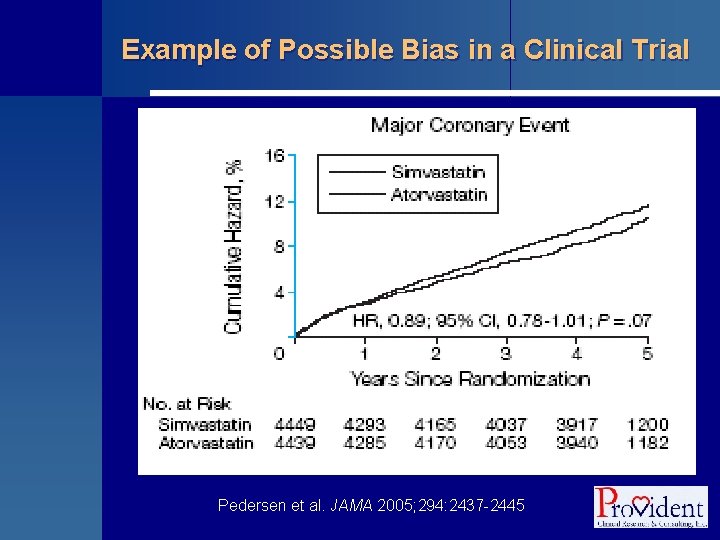
Example of Possible Bias in a Clinical Trial Pedersen et al. JAMA 2005; 294: 2437 -2445

FIELD Trial: Example of Confounding in a Clinical Trial § Subjects with Type 2 diabetes (N = 9795) § Randomly assigned to receive • Fenofibrate 200 mg/d • Placebo § Average follow-up : ~5 years § During the study more subjects in the placebo group started other lipid drug therapy (17% vs. 8%) FIELD Investigators. Lancet 2005; 366: 1849 -1861 .
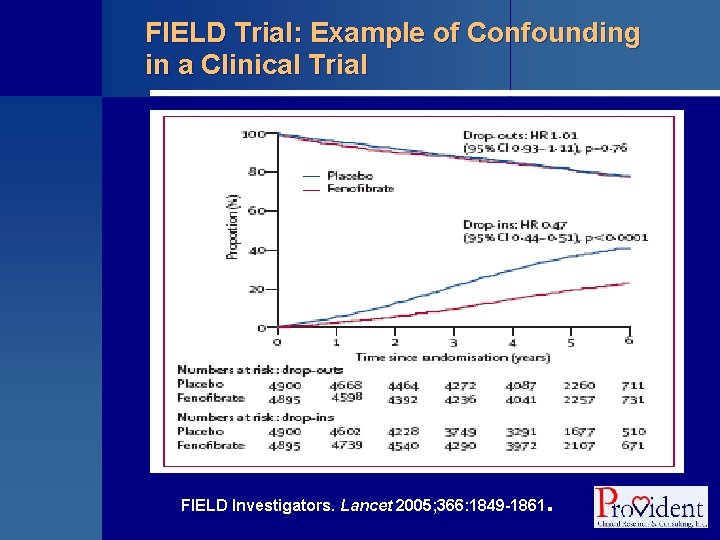
FIELD Trial: Example of Confounding in a Clinical Trial FIELD Investigators. Lancet 2005; 366: 1849 -1861 .
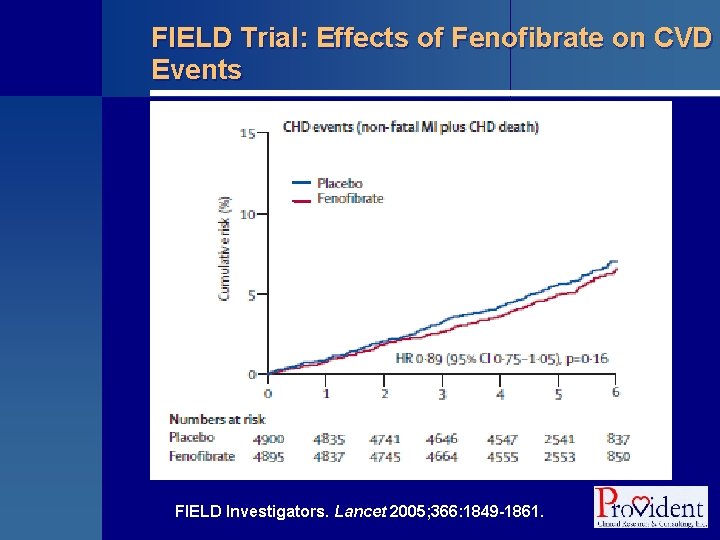
FIELD Trial: Effects of Fenofibrate on CVD Events FIELD Investigators. Lancet 2005; 366: 1849 -1861.

FIELD Trial: Effects of Fenofibrate on CVD Events § Treatment effect • HR = 0. 89, 95% CI = 0. 75 to 1. 05, P = 0. 16 § The hazard for an event in the fenofibrate group was 89% of that in the placebo group • Reduced by 11% § Because the 95% confidence interval crosses 1. 0, difference was not statistically significant at the 5% level • As confirmed by the P-value FIELD Investigators. Lancet 2005; 366: 1849 -1861.

Efficacy vs. Effectiveness in Clinical Trials § Efficacy • Refers to whether an intervention produces the desired effects when properly implemented under controlled conditions § Effectiveness • Refers to whether an intervention is typically successful in clinical practice § In order for a treatment to be useful, it must be both efficacious and effective

Evaluation of Clinical Trial Results § Intent-To-Treat Analysis • All subjects randomized are analyzed (or as close to this ideal as possible) • Subjects are part of their assigned group whether or not they actually received the treatment, adhered to the treatment regimen, or even received the opposite treatment § Per Protocol Analysis • Subjects who discontinued or who had material protocol violations, including poor adherence, are excluded from the analysis

Evaluating a Published Clinical Trial Report 1. Was the subject sample similar to patients commonly seen in clinical practice? • If not, will this limit generalizability? 2. Were important subgroups represented? • Both sexes, smokers, those with obesity and/or diabetes, various lipid phenotypes 3. Were random allocation, concurrent controls and blinding all employed?

Evaluating a Published Clinical Trial Report 4. Were subjects generally compliant with the treatment regimen? • Compliance consistent between treatments? 5. Did the trial suffer from excessive subject attrition or differential dropout between treatments? 6. Was the primary outcome variable prespecified and clearly stated? 7. Did the trial show a statistically significant difference for the primary outcome variable?

Evaluating a Published Clinical Trial Report 8. Did the results appear consistent across subgroups and secondary analyses? 9. If significant results were not achieved, could this have been due to the trial lacking sufficient statistical power to detect a clinically meaningful difference? 10. How do the results from this trial align with those from other sources? • Clinical trials, observational studies, animal experiments and mechanistic investigations

Descriptive Statistics § Central tendency • Mean (arithmetic average) • Median (50% above and below) § Dispersion • Standard deviation • Range limits (minimum and maximum) • Inter-quartile range limits (25 th, 75 th percentiles) § Precision of a point estimate for a mean • Standard error of the mean (equal to SD/√n) • 95% confidence interval (mean ± 1. 96*SEM)
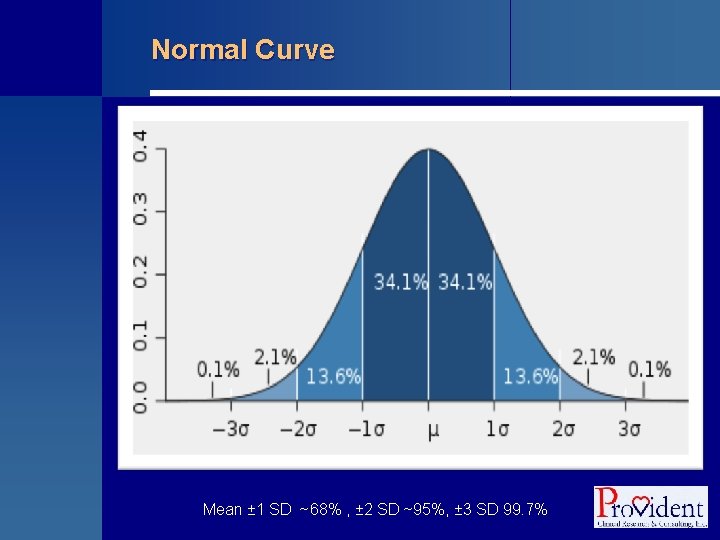
Normal Curve Mean ± 1 SD ~68% , ± 2 SD ~95%, ± 3 SD 99. 7%

Inferential Statistics for Means § Two groups • t-test § More than 2 groups • Analysis of variance (ANOVA) followed by a posthoc test such as the Tukey procedure § Tests using ranks in place of raw data are used when the sample is small or when the distribution is not normal (e. g. , skewed) • Non-parametric tests

Statistical Power and Sample Size § Type I statistical error • Finding “significance” when a relationship is due to random variation (false positive) § Type II statistical error • Failing to find significance when it is truly present • Insufficient power (false negative) § Statistical power • Likelihood of finding significance if it is truly present • 1 – power = likelihood of a type II error • Power usually set at 80% or 90% for a 2 -sided p-value of 0. 05 for a given set of assumptions when planning a study (10 -20% chance of a type II error)

Statistical Power and Sample Size § Ways to increase statistical power • Increase sample size • Increase precision of measurements • Dual x-ray absorptiometry vs. skin-folds for body fat • Average values from multiple days to minimize the influence of biological variation (e. g. , hs-CRP, TG, fibrinogen) – LDL-C example: 19%, 10%, 9% SD • Study a higher risk population • A greater number of events will increase power in clinical event studies

Multiple Comparisons, Post-hoc Analyses and Data Mining § With 100 statistical tests, it is expected to find ~5 that are “significant” due to random variation § For clinical studies • Primary and secondary hypotheses should be clearly stated before analysis • Post-hoc and sub-group analyses should be interpreted with caution • Associations uncovered during data mining should be viewed as hypotheses
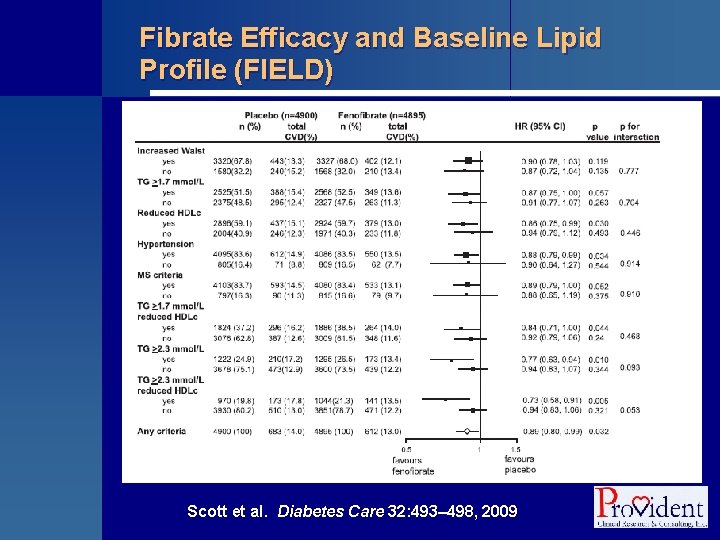
Fibrate Efficacy and Baseline Lipid Profile (FIELD) Scott et al. Diabetes Care 32: 493– 498, 2009

Methods for Assessing Associations § Correlation • Pearson or Spearman r value (from -1. 0 to +1. 0) § Regression • Linear • Y = a + b*x (univariate) • Y = a + b 1*x 1 + b 2*x 2 + … (multivariate) • Logistic • Y (0, 1) = 1/[1 + e-(a + b*x)] § Survival analysis • Kaplan Meier survival curves (log-rank test) • Cox proportional hazards regression
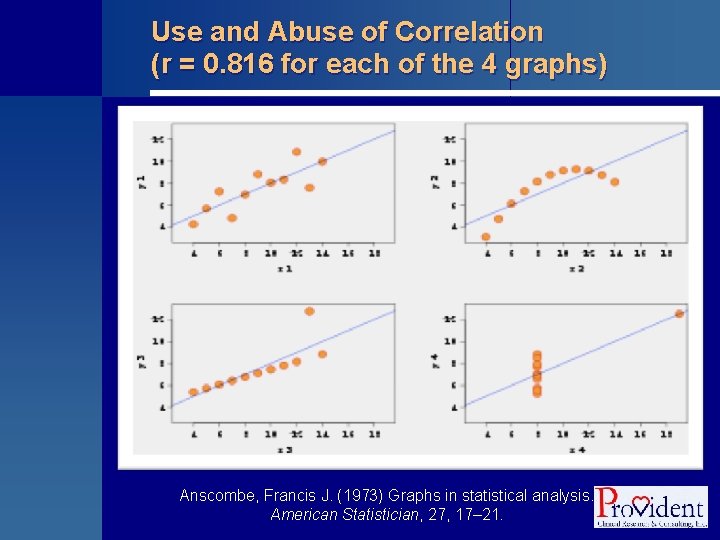
Use and Abuse of Correlation (r = 0. 816 for each of the 4 graphs) Anscombe, Francis J. (1973) Graphs in statistical analysis. American Statistician, 27, 17– 21.

Expressing Disease Frequency and Risk § Measures of disease frequency • Prevalence, incidence, risk § Measures of association • Relative risk or hazard ratio • Cohort studies • Odds ratio • Cross sectional or case-control studies § Population attributable risk

Measures of Disease Frequency § Incidence and prevalence are the two most commonly used rate statistics in epidemiology • Incidence describes the rate of development of a disease in a group over a period of time • Prevalence describes a group at a certain point in time § A snapshot of an existing situation

Incidence § There are two types of incidence measures: • Cumulative Incidence – the proportion of people who become diseased during a specific time period • Incidence Rate – the number of new cases of a disease per total person-time of observation § Person-time refers to the sum of all individuals’ time under observation § Following 1, 000 women for 5 years provides 5, 000 woman-years § Following 5, 000 women for 1 year provides 5, 000 woman-years

Incidence and Prevalence § The relationship between incidence and prevalence can be expressed mathematically: _ PαIx. D § Prevalence (P) is proportional to the product of incidence rate (I) and the average duration of the disease (D)

Number Needed to Treat § Reciprocal of absolute risk difference § Example • Symptomatic venous thromboembolism in JUPITER • • • Placebo rate = 0. 32 per 100 person-years Rosuvastatin rate = 0. 18 per 100 person-years AR difference = 0. 32 – 0. 18 = 0. 14 per 100 person years or 0. 14% annual risk = 0. 14/100 = 0. 0014 • 1/0. 0014 = 1 DVT prevented for each 714 patients treated for one year Glynn et al. N Engl J Med 2009; 360: 1 -11.

P-Values and Confidence Intervals § P-value represents the probability that an association occurred due to chance • P = 0. 05 = 5% or 5/100 chance that the association occurred due to random variation § Confidence interval • 95% CI = range within which one can be 95% confident that the true value lies

P-Values and Confidence Intervals § Relationship between CI and P-Value • If the 95% CI crosses the null value, the relationship is not significant at the 5% level • If the 95% CI does not cross the null value, the relationship is significant at the 5% level • Null value for a difference in means, medians or absolute risk = 0. 0 • Null value for RR, HR or OR = 1. 0

Measures of Association (Point Estimates) § Differences • Means, medians, absolute risk (AR) § Odds ratio (OR) § Relative risk (RR) § Hazard ratio (HR)

Relative Risk or Hazard Ratio § Relative risk (RR) or hazard ratio (HR) calculated as the ratio of the incidence of those exposed to the incidence of those not exposed § RR uses cumulative incidence § HR uses incidence rates (person-time units of observation) RR or HR = Incidence in those Exposed Incidence in those Not Exposed
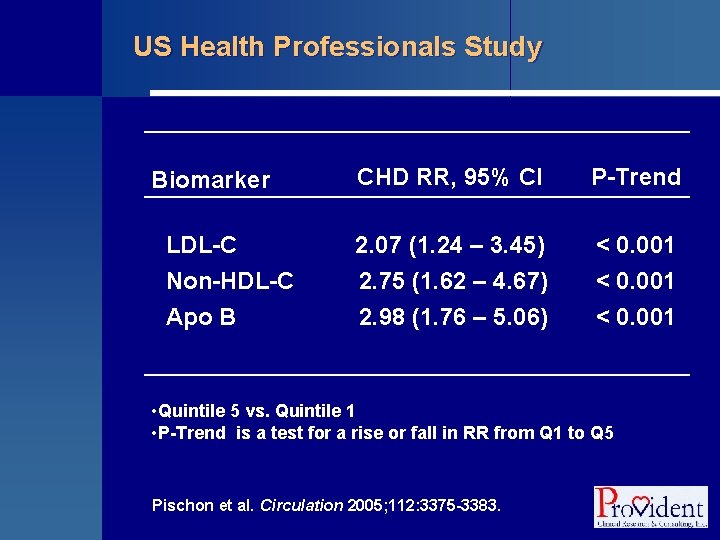
US Health Professionals Study CHD RR, 95% CI P-Trend LDL-C Non-HDL-C 2. 07 (1. 24 – 3. 45) 2. 75 (1. 62 – 4. 67) < 0. 001 Apo B 2. 98 (1. 76 – 5. 06) < 0. 001 Biomarker • Quintile 5 vs. Quintile 1 • P-Trend is a test for a rise or fall in RR from Q 1 to Q 5 Pischon et al. Circulation 2005; 112: 3375 -3383.

Framingham: VLDL-C and LDL-C Have Similar Relative Risks for CHD Liu et al. Am J Cardiol. 2006; 98: 1363 -1368.

Odds Ratios § Case control and cross sectional studies use odds ratios to reflect association § Odds of exposure in the cases divided by the odds of exposure in controls • Obesity and diabetes example (100 cases and 100 controls) • Cases with diabetes have 80% prevalence of BMI 30 kg/m 2 or higher • Controls without diabetes have 35% prevalence of BMI 30 kg/m 2 or higher

Odds Ratio Calculation § Odds of obesity = P/(1 – P) = Yes/No • Odds among cases = 80/20 = 4. 00 • Odds among controls = 35/65 = 0. 54 • Odds ratio = 4. 0/0. 54 = 7. 43 • 95% CI = 3. 92 to 14. 08, P < 0. 0001

OR vs. HR or RR § HR or RR are measures of relative incidence or “risk” § OR is a measure of relative prevalence • Will be a reasonable proxy for HR or RR when the prevalence of the condition is low • Is not equivalent to HR or RR as a measure of “risk”, particularly when the prevalence is high (as in diabetes example)

INTERHEART: Risk of Acute MI Associated With Modifiable Risk Factors § Odds ratios for acute MI based on modifiable risk factors: • Smoking • 2. 9 (95% CI = 2. 6 -3. 2) • Diabetes Mellitus • 2. 4 (95% CI = 2. 1 -2. 7) • Hypertension • 1. 9 (95% CI = 1. 7 -2. 1) • Apo. B/Apo. A 1 (extreme quintiles) • 3. 3 (95% CI = 2. 8 -3. 8) Yusef et al. Lancet 2004; 364: 937 -52

Collaborative Atorvastatin Diabetes Study (CARDS) § Effectiveness of atorvastatin 10 mg for primary prevention of CVD events in subjects with type 2 diabetes, but without high LDL-C § Randomly assigned to receive • Atorvastatin 10 mg/d • Placebo § Median duration of follow-up: 3. 9 years Colhoun et al. Lancet 2004; 364: 685 -696.
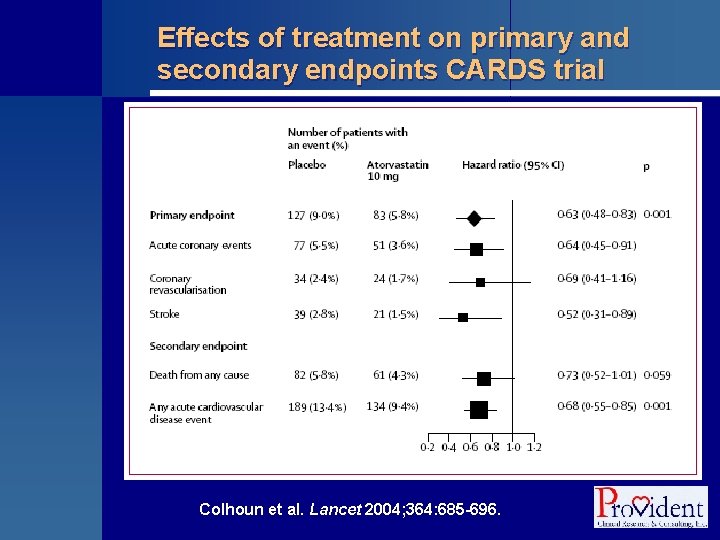
Effects of treatment on primary and secondary endpoints CARDS trial Colhoun et al. Lancet 2004; 364: 685 -696.

Population Attributable Risk § Population attributable risk provides the impact of an exposure on a disease, assuming the association is causal • Often expressed as a % of all cases • >90% of CHD incidence is attributable to potentially modifiable risk factors § The population attributable risk (PAR) for a risk factor depends on two features: 1. The strength of the relationship between the risk factor and the disease (assuming a causal relationship) 2. The prevalence of the risk factor

Population Attributable Risk § A causal factor that has a strong association with the disease outcome might have only a minor influence on the population attributable risk fraction if it has a low prevalence § Conversely, a causal factor that produces a modest increase in risk can have a high population attributable risk fraction if it is very common

Limitations of Epidemiology § B vitamins to lower homocysteine § Vitamin E § Postmenopausal hormone therapy • CEE + MPA

Postmenopausal Hormone Therapy § More than 40 epidemiological studies had shown that use of postmenopausal estrogen or estrogen + progestin was associated with lower risks for CHD morbidity and mortality with relative risk estimates in the 0. 60 to 0. 50 range (4050% lower risk)
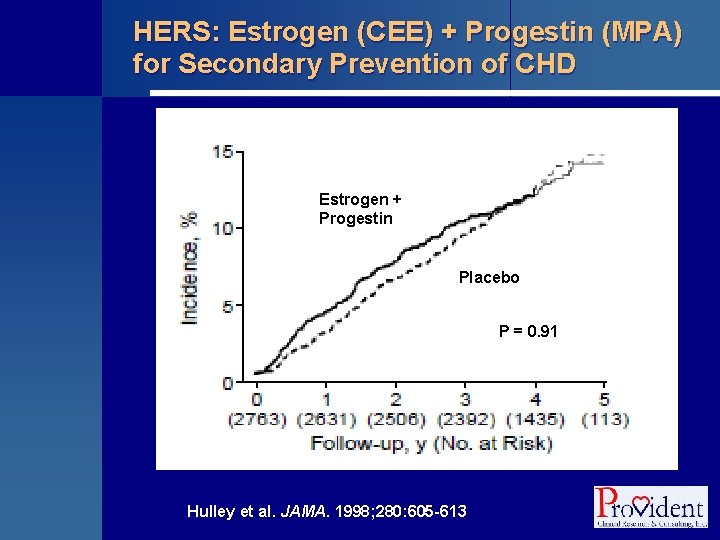
HERS: Estrogen (CEE) + Progestin (MPA) for Secondary Prevention of CHD Estrogen + Progestin Placebo P = 0. 91 Hulley et al. JAMA. 1998; 280: 605 -613

Women’s Health Initiative: Estrogen + Progestin in Postmenopausal Women § >16, 000 women with intact uteri assigned to E+P or placebo § Absolute excess risks per 10, 000 person years attributable to estrogen plus progestin were: • 7 more CHD events, 8 more strokes • 8 more pulmonary embolisms • 8 more invasive breast cancers § Reduced risks for colorectal cancer and hip fractures WHIInvestigators. JAMA. 2002; 288: 321 -333

Women’s Health Initiative: Estrogen + Progestin in Postmenopausal Women § Overall health risks exceeded benefits from use of estrogen + progestin • Results indicate estrogen + progestin should not be initiated or continued for primary prevention of CHD § Explanations for favorable epidemiology? • Chance (unlikely) • Bias and confounding • Healthy user effect, compliance effect • Age interaction (protective early, harmful later)? WHI Investigators. JAMA. 2002; 288: 321 -333

JUPITER Trial § The Justification for the Use of Statins in Prevention: An Intervention Trial Evaluating Rosuvastatin (JUPITER) • Rosuvastatin 20 mg/d • Placebo § Rate of occurrence of first major cardiovascular events in otherwise healthy individuals with elevated hs-CRP (≥ 2. 0 mg/L) and LDL-C < 130 mg/d. L Ridker et al. N Engl J Med 2008; 359: 2195 -207.
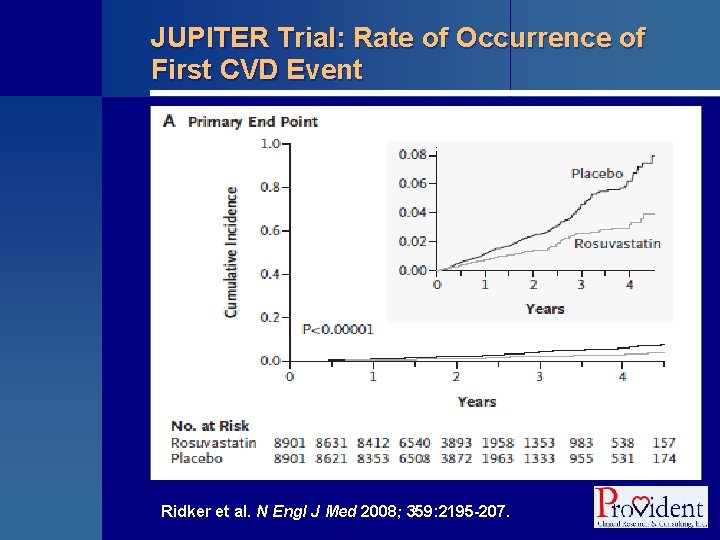
JUPITER Trial: Rate of Occurrence of First CVD Event Ridker et al. N Engl J Med 2008; 359: 2195 -207.

JUPITER Trial: Results § Rosuvastatin reduced LDL-C levels by 50% and high-sensitivity hs-CRP levels by 37% § The rates of the primary end point were 0. 77 and 1. 36 per 100 person-years of follow-up in the rosuvastatin and placebo groups, respectively • HR for rosuvastatin: 0. 56 • 95% CI: 0. 46 to 0. 69, P < 0. 00001 Ridker et al. N Engl J Med 2008; 359: 2195 -207.

JUPITER Trial Summary § Trial was stopped early because the rosuvastatin group showed reductions of ~50% in several cardiovascular endpoints relative to the placebo group § Results provide clear evidence that those with elevated hs-CRP are at increased CHD risk and that rosuvastatin therapy lowers this risk § JUPITER was not designed to answer the question of whether hs-CRP reduction per se should be a target of therapy
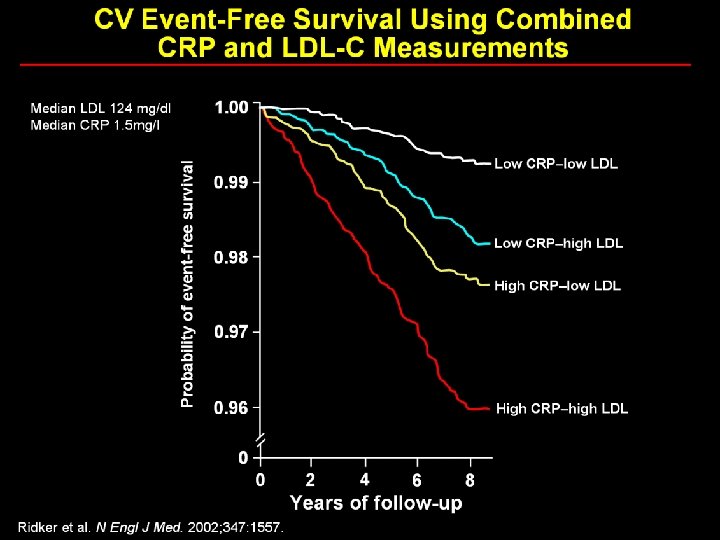
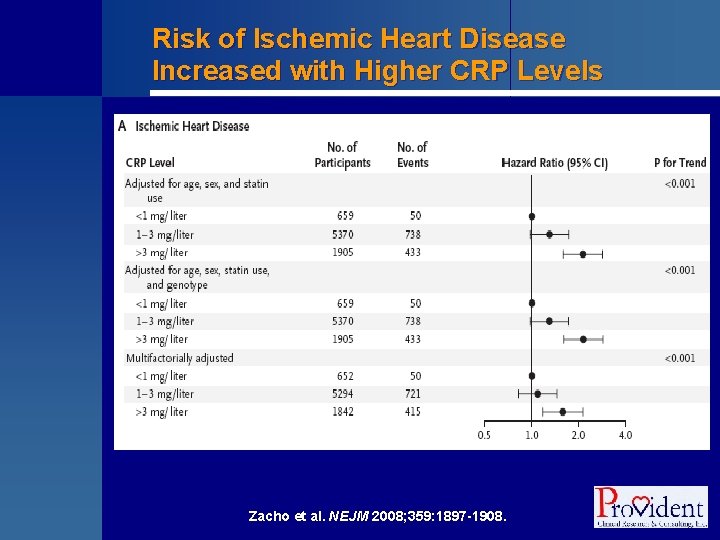
Risk of Ischemic Heart Disease Increased with Higher CRP Levels Zacho et al. NEJM 2008; 359: 1897 -1908.
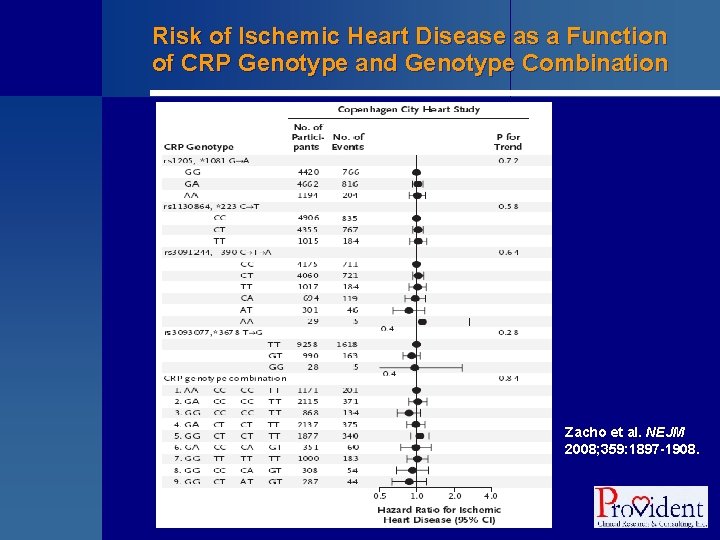
Risk of Ischemic Heart Disease as a Function of CRP Genotype and Genotype Combination Zacho et al. NEJM 2008; 359: 1897 -1908.
- Slides: 84