ENZYMES Why care about enzymes Enzymes are necessary

ENZYMES

Why care about enzymes? • Enzymes are necessary for a variety of reactions – for photosynthesis, cell respiration, digestion, etc. • There are reactions in your body that would take over 200 years to complete if it wasn’t for enzymes • Enzymes lower activation energy or the amount of energy required to start a chemical reaction.

Chemical Reactions • When you eat, the food is broken down through chemical reactions performed by our metabolisms. • To write a chemical reaction: reactants products • Reactants – what is put in/used & comes before the arrow • Products – what is made/produced & comes after the arrow – The arrow, , means yields, makes, creates, etc. For example… Chicken + noodles + broth chicken noodle soup reactants products 2 H 2 + O 2 H O 2 H 2 & O are the reactants, H O is the product
Catalysts • Catalysts – anything which speeds up a reaction, written above the yield( ) sign catalyst Reactants Products • Enzymes – proteins that are catalysts in many chemical reactions, they speed up reactions occurring in the body

Things to know about enzymes • Enzymes are proteins that speed up reactions • Enzymes are specific – only able to be used for one type of reaction. The shape of the enzyme determines the specific reaction it catalyzes (speeds up) • Enzymes are reusable – they can be used over & over for the particular type of reaction • Enzymes lower activation energy or the amount of energy required to start a chemical reaction.
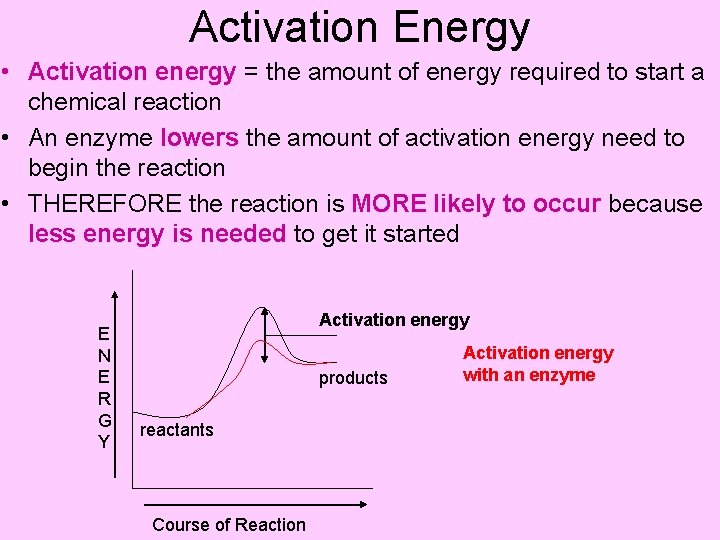
Activation Energy • Activation energy = the amount of energy required to start a chemical reaction • An enzyme lowers the amount of activation energy need to begin the reaction • THEREFORE the reaction is MORE likely to occur because less energy is needed to get it started E N E R G Y Activation energy products reactants Course of Reaction Activation energy with an enzyme
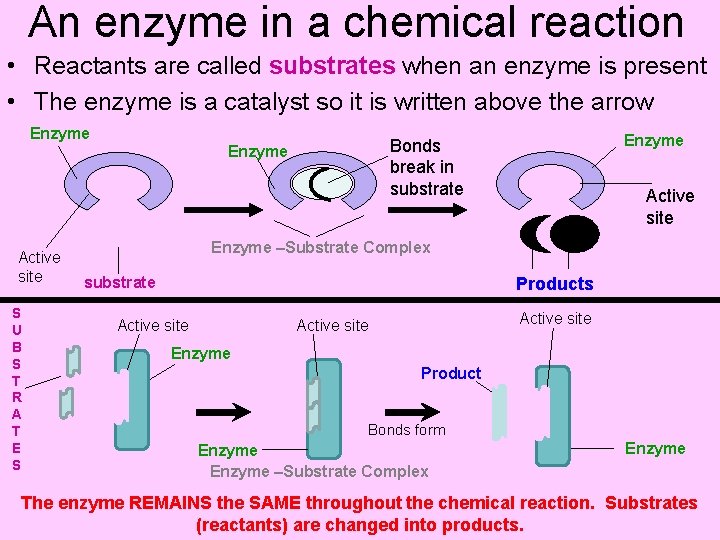
An enzyme in a chemical reaction • Reactants are called substrates when an enzyme is present • The enzyme is a catalyst so it is written above the arrow Enzyme Active site S U B S T R A T E S Enzyme Bonds break in substrate Enzyme Active site Enzyme –Substrate Complex substrate Products Active site Enzyme Product Bonds form Enzyme –Substrate Complex Enzyme The enzyme REMAINS the SAME throughout the chemical reaction. Substrates (reactants) are changed into products.

Things to remember!!! • Active sites are found on the ENZYME • An enzyme & substrate together are known as an enzyme –substrate complex • Due to the enzyme shaping itself to fit a substrate, this is called induced-fit or lock & key (a key fits only one type of lock, just as an enzyme only fits one type of substrate)

How well enzymes work are effected by 3 things: • p. H – different enzymes work best at certain p. H’s between 0 -14 • Enzyme concentration – adding more enzyme helps speed up the reaction more. ***However, at a certain concentration adding more has no greater effect. • Enzymes work best at particular temperatures. If the temperature becomes too high the enzyme becomes denatured (broken down) and will no longer speed up the reaction
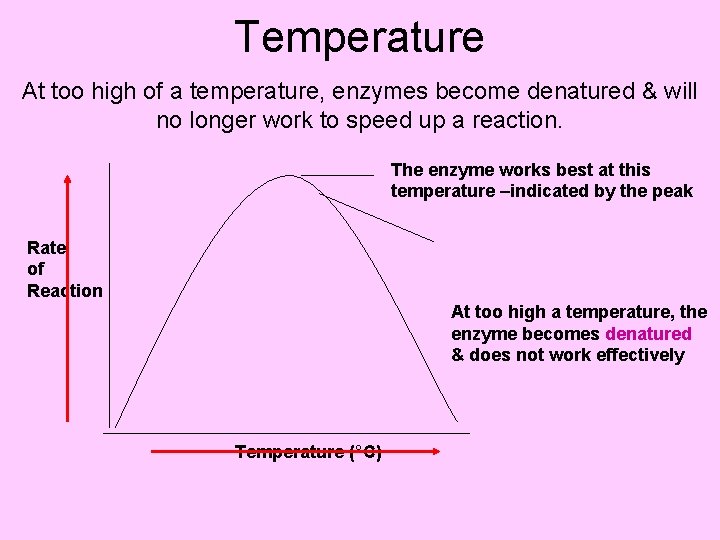
Temperature At too high of a temperature, enzymes become denatured & will no longer work to speed up a reaction. The enzyme works best at this temperature –indicated by the peak Rate of Reaction At too high a temperature, the enzyme becomes denatured & does not work effectively Temperature (°C)
- Slides: 10