ENZYMES Part 2 Ms Rahinaz Department of Biochemistry

ENZYMES - Part 2 Ms. Rahinaz Department of Biochemistry Yenepoya Medical College, Yenepoya. 1

CONTENTS 1. Enzyme Inhibition 2. Clinical applications of enzymes 2

Definitions : ENZYME INHIBITION Enzyme inhibitor : Is as a substance which binds to the enzyme, and brings about a decrease in the velocity of the enzyme-catalyzed reaction. Enzyme inhibition : Is the reduction of enzyme activity by binding of inhibitor to the enzyme. 3

ENZYME INHIBITION……. . Importance of enzyme inhibitors : 1. Used to elucidate the mechanism of enzyme action. 2. Many drugs and poisons are enzyme inhibitors. E. g. , : Group of Drugs : Anticancer, antibiotics, antivirals, immunosuppresents. 4

ENZYME INHIBITION……. . Features of an Inhibitor : Enzyme + Inhibitor Enzyme Inhibition 1. Where does the inhibitor bind on the enzyme? Inhibitor may bind (i) to the active site or (ii) to a site other than the active site. it may be to a § Free enzyme molecule or § ES-complex. 5

Features of an Inhibitor……… (2. ) What does it bind ? Binds to specific R-groups of amino acid residues, involved in , § catalysis, § substrate-binding or § maintenance of functional conformation of the enzyme. 6

Features of an Inhibitor……… (3. ) What type of interactions between enzyme and inhibitor ? (i) Weak, non-covalent bonds - Reversible. q Ionic bond, q H-bond, q Hydrophobic bond. (ii) Strong covalent bond - irreversible. 7

Features of an Inhibitor……… (4. ) What is the product of enzyme inhibitor interaction? Enzyme – Inhibitor complex is formed. i. e. , EI complex 8
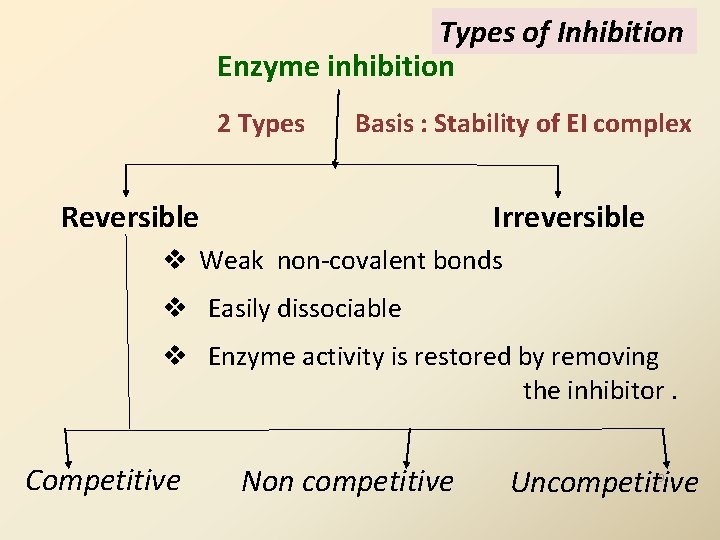
Types of Inhibition Enzyme inhibition 2 Types Basis : Stability of EI complex Reversible Irreversible v Weak non-covalent bonds v Easily dissociable v Enzyme activity is restored by removing the inhibitor. Competitive Non competitive Uncompetitive 9

I. Reversible inhibition: 3 types a) Competitive b) Non-competitive c) Uncompetitive 10
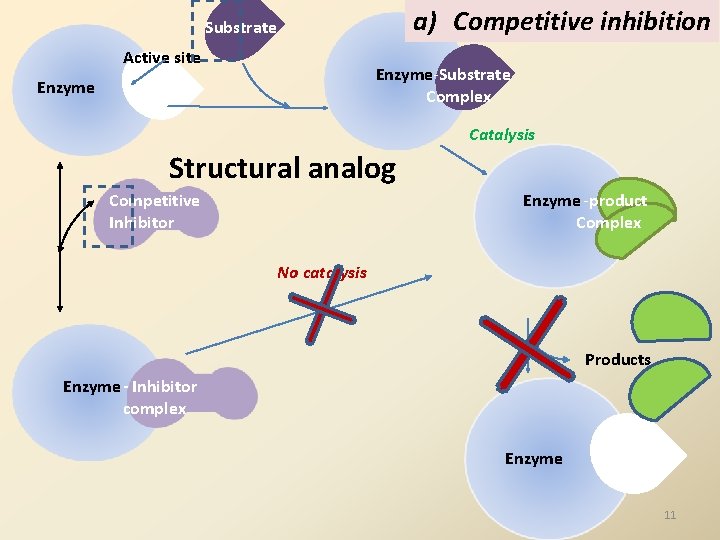
a) Competitive inhibition Substrate Active site Enzyme-Substrate Complex Enzyme Catalysis Structural analog Enzyme -product Complex Competitive Inhibitor No catalysis Products Enzyme - Inhibitor complex Enzyme 11
![Active site Enzyme Substrate S S S Increase in [S] Inhibition is reversed Enzyme Active site Enzyme Substrate S S S Increase in [S] Inhibition is reversed Enzyme](http://slidetodoc.com/presentation_image/37cf3e51a87031364867deef4ccd1863/image-12.jpg)
Active site Enzyme Substrate S S S Increase in [S] Inhibition is reversed Enzyme is active Competitive Inhibitor Enzyme -product Complex Catalysis Enzyme - Inhibitor complex Products No catalysis EScomplex EI complex 1. Competitive inhibition Enzyme 12

1. Competitive inhibition Definition : Competitive inhibition is a type of • reversible inhibition in which • the inhibitor is a structural analog of the substrate • and hence competes with the substrate for the active site. 13
![Features of an competitive Inhibitor……… • At high [S], ‘S’ displaces the ‘I’ molecule Features of an competitive Inhibitor……… • At high [S], ‘S’ displaces the ‘I’ molecule](http://slidetodoc.com/presentation_image/37cf3e51a87031364867deef4ccd1863/image-14.jpg)
Features of an competitive Inhibitor……… • At high [S], ‘S’ displaces the ‘I’ molecule from the substrate binding site and thus overcome the inhibition. Kinetics of an competitive Inhibition Kinetics - Effect on Vmax and Km 14
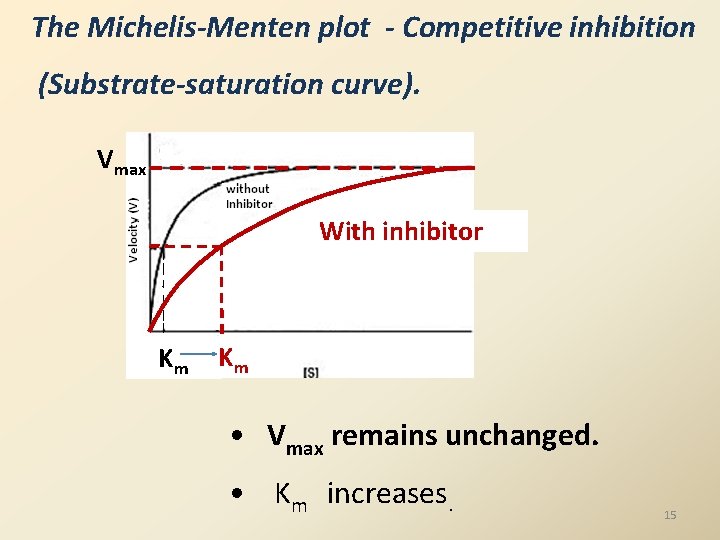
The Michelis-Menten plot - Competitive inhibition (Substrate-saturation curve). Vmax With inhibitor Km Km • Vmax remains unchanged. • Km increases. 15
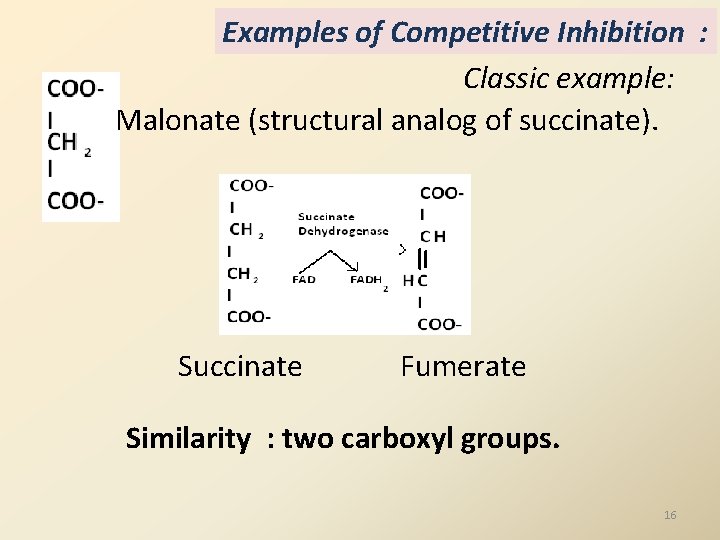
Examples of Competitive Inhibition : Classic example: Malonate (structural analog of succinate). Succinate Fumerate Similarity : two carboxyl groups. 16
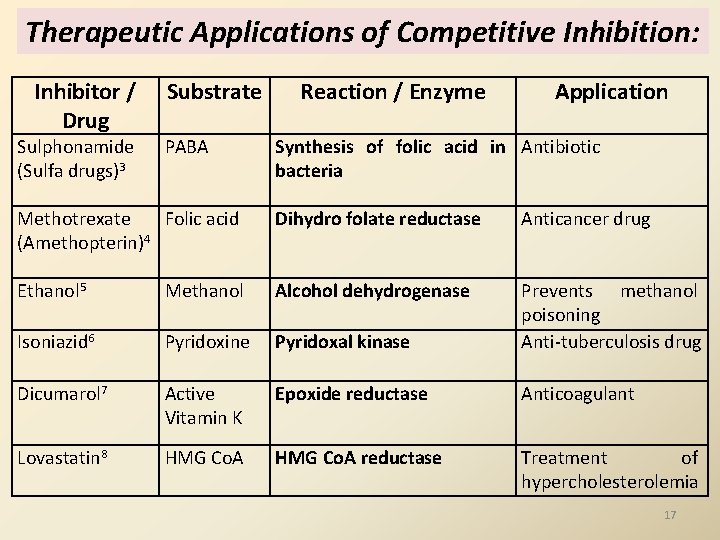
Therapeutic Applications of Competitive Inhibition: Inhibitor / Drug Sulphonamide (Sulfa drugs)3 Substrate PABA Reaction / Enzyme Application Synthesis of folic acid in Antibiotic bacteria Methotrexate Folic acid (Amethopterin)4 Dihydro folate reductase Anticancer drug Ethanol 5 Methanol Alcohol dehydrogenase Isoniazid 6 Pyridoxine Pyridoxal kinase Prevents methanol poisoning Anti-tuberculosis drug Dicumarol 7 Active Vitamin K Epoxide reductase Anticoagulant Lovastatin 8 HMG Co. A reductase Treatment of hypercholesterolemia 17
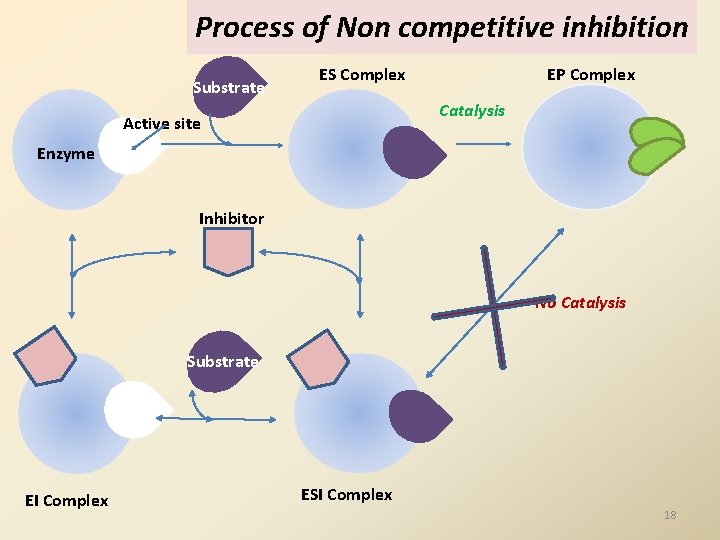
Process of Non competitive inhibition Substrate ES Complex EP Complex Catalysis Active site Enzyme Inhibitor No Catalysis Substrate EI Complex ESI Complex 18

2. Non competitive inhibition Definition : Uncompetitive inhibition is a type of • reversible inhibition in which • the inhibitor is NOT a structural analog of the substrate • and hence DOES NOT compete with the substrate for the active site. 19
![Features of an NON competitive Inhibitor……… • At high [S], ‘S’ cannot displace the Features of an NON competitive Inhibitor……… • At high [S], ‘S’ cannot displace the](http://slidetodoc.com/presentation_image/37cf3e51a87031364867deef4ccd1863/image-20.jpg)
Features of an NON competitive Inhibitor……… • At high [S], ‘S’ cannot displace the ‘I’ molecule from the substrate binding site and thus cannot overcome the inhibition. Kinetics - Effect on Vmax and Km 20
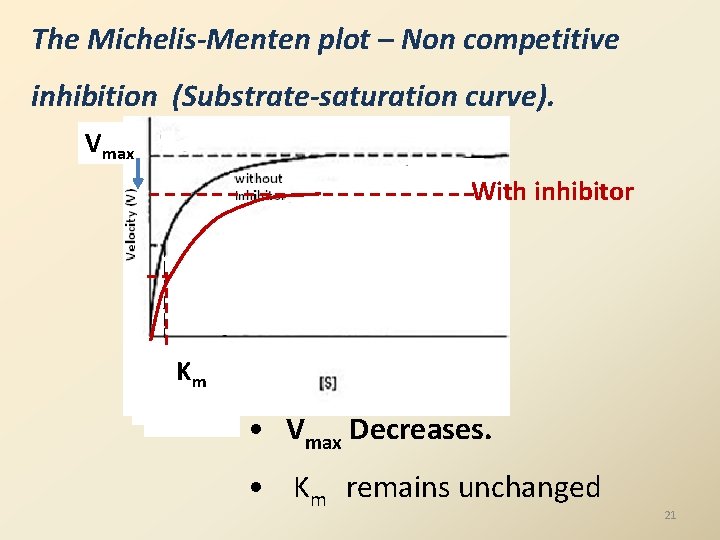
The Michelis-Menten plot – Non competitive inhibition (Substrate-saturation curve). Vmax With inhibitor KKm m • Vmax Decreases. • Km remains unchanged 21

Examples for NON competitive inhibitors i. Trypsin inhibitors occur in Soyabean and Ascaris parasites (worms). ii. -1 Antitrypsin which inhibits serine proteases (such as trypsin). 22
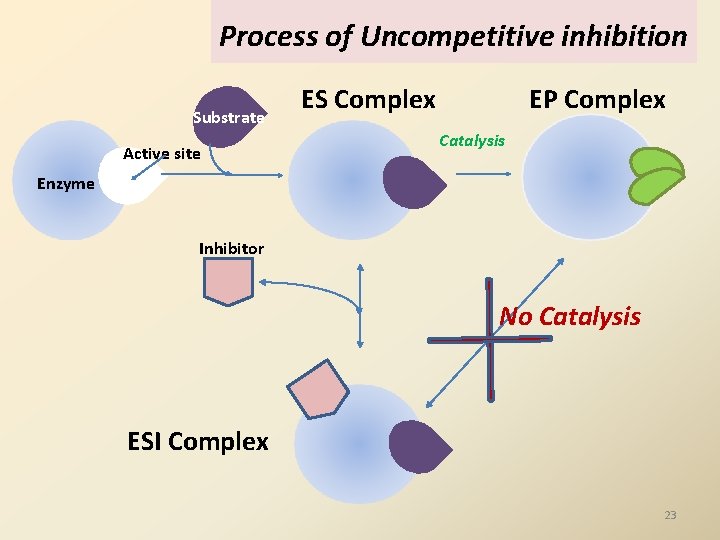
Process of Uncompetitive inhibition Substrate Active site ES Complex EP Complex Catalysis Enzyme Inhibitor No Catalysis ESI Complex 23

2. Uncompetitive inhibition Definition : Uncompetitive inhibition is a type of • reversible inhibition in which • the inhibitor binds only to the ES complex • Increased [S], cannot overcome the inhibition. 24
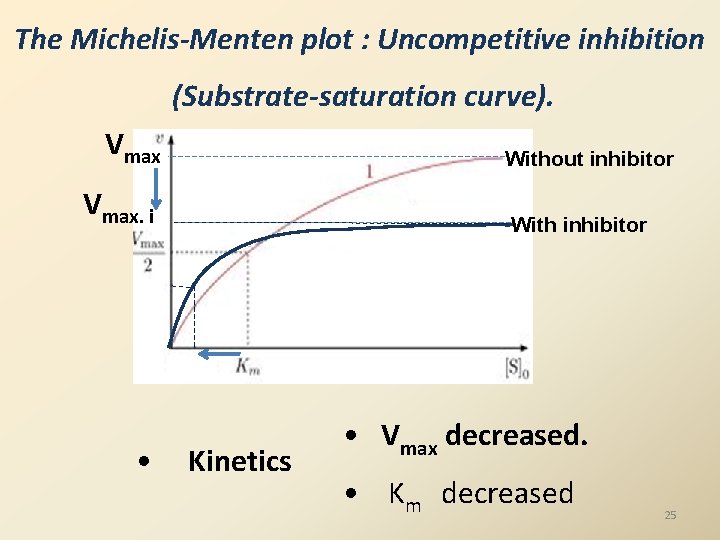
The Michelis-Menten plot : Uncompetitive inhibition (Substrate-saturation curve). Vmax Without inhibitor Vmax. i • With inhibitor Kinetics • Vmax decreased. • Km decreased 25

Example for Uncompetitive Inhibition Phenylalanine uncompetitively inhibits enzyme placental alkaline phosphatase. Summary of effect of reversible inhibitors competitive Stability Inhibitor Structural analog of Substrate Non competitive Reversible Not a structural analog Uncompetitiv e Not a structural analog
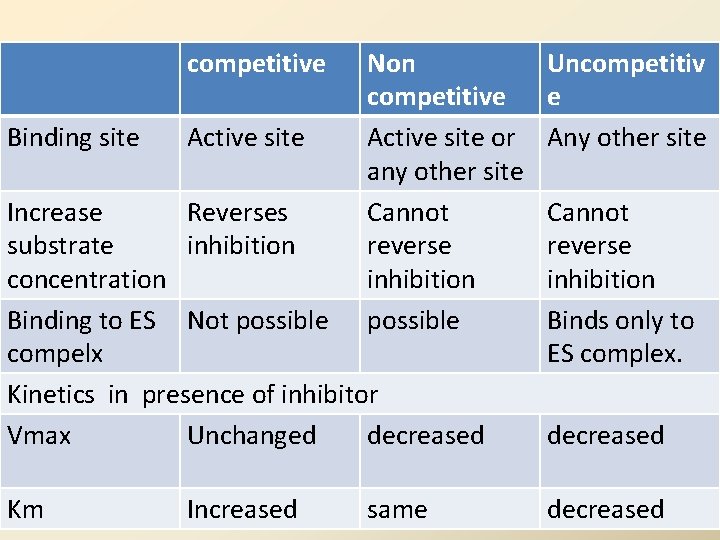
competitive Binding site Active site Non competitive Active site or any other site Cannot reverse inhibition possible Uncompetitiv e Any other site Increase Reverses Cannot substrate inhibition reverse concentration inhibition Binding to ES Not possible Binds only to Type of inhibitor K V m max compelx ES complex. Competitive Increased No effect Kinetics in presence of inhibitor effect Decreased Vmax. Non - competitive Unchanged Nodecreased Uncompetitive Km Increased Decreased same Decreased decreased 27

The inhibitor II. Irreversible inhibition: üBinds to the enzyme tightly üBy covalent bonds üForms a stable complex. üEnzyme-inhibitor complex does not dissociate significantly. Examples: A variety of poisons (toxic substances), oxidizing agents. 28

Examples for Irreversible inhibition A variety of poisons (toxic substances), oxidizing agents -irreversible inhibitors. INHIBITOR ENZYME Fluoride Enolase Cyanide Cytochrome oxidase of electron transport chain Heavey metal ions Hg 2+, Pb 2+, Ag+, Arsenite GROUPS ON THE ENZYME Removes the Mg 2+ the active site IMPORTANCE / APPLICATION Fluoride inhibits glycolysis. It is therefore from added to the sample of blood collected for blood sugar estimation. “Respiratory poison”, since it inhibits cellular respiration. Covalent bond formation Heavy metal with -SH groups poisoning. ion 29

Examples for Irreversible inhibition……… INHIBITOR IMPORTANCE / APPLICATION Organophosphorus Acetyl Choline Main component of compounds esterase – required Serine group in the insecticides, Also for nerve active site commonly used for Eg: Malathion, conduction and committing suicide. Parathione cause paralysis Diisopropyl fluorophosphates ENZYME Acetyl esterase GROUPS ON THE ENZYME Choline Serine group in the “Nerve gas” active site (DIPF / DFP / Sarin) Iodoacetate Phosphoglyceralde Covalent bond Inhibits glycolysis. hyde formation with -SH Dehydrogenase groups in the active site of the enzyme. 30
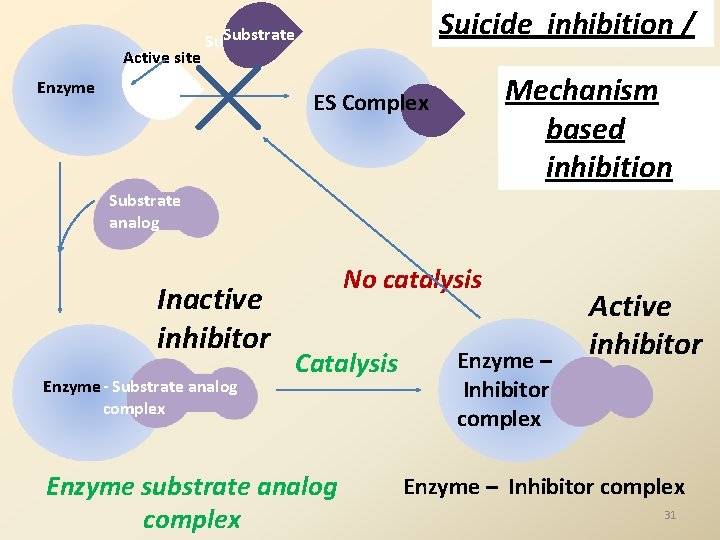
Active site Suicide inhibition / Substrate Enzyme Mechanism based inhibition ES Complex Substrate analog Inactive inhibitor Enzyme - Substrate analog complex No catalysis Catalysis Enzyme substrate analog complex Enzyme – Inhibitor complex Active inhibitor Enzyme – Inhibitor complex 31

Suicide inhibition or Mechanism based inactivation: Is a special type of irreversible inhibition The inhibitor is a substrate analog is inactive initially binds to the active site of the enzyme. the Inhibitor is modified. Modified inhibitor is an effective inhibitor binds irreversibly and inhibits further reactions of the same enzyme. 32
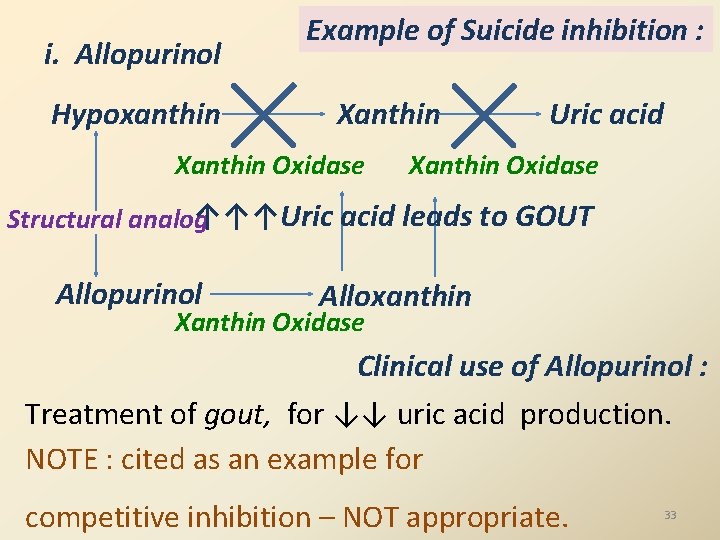
i. Allopurinol Hypoxanthin Example of Suicide inhibition : Xanthin Oxidase Uric acid Xanthin Oxidase ↑↑↑Uric acid leads to GOUT Structural analog Allopurinol Alloxanthin Xanthin Oxidase Clinical use of Allopurinol : Treatment of gout, for ↓↓ uric acid production. NOTE : cited as an example for competitive inhibition – NOT appropriate. 33

Clinical Applications of Enzymes: 3 Types A. Diagnostic B. Therapeutic C. Laboratory A. Diagnostic applications Diagnostic importance of plasma enzymes/ Plasma enzymes : 2 types Clinical Enzymology: Functional enzymes : Blood clotting factors Non functional enzymes : LDH; CPK 34
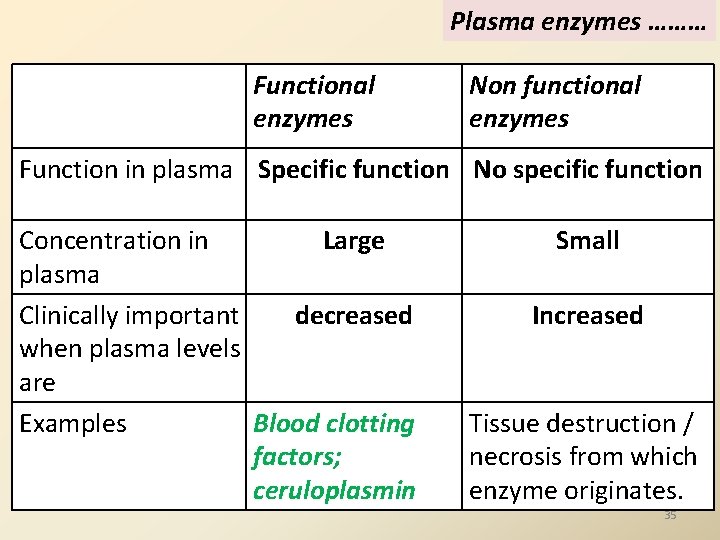
Plasma enzymes ……… Functional enzymes Non functional enzymes Function in plasma Specific function No specific function Concentration in Large plasma Clinically important decreased when plasma levels are Examples Blood clotting factors; ceruloplasmin Small Increased Tissue destruction / necrosis from which enzyme originates. 35
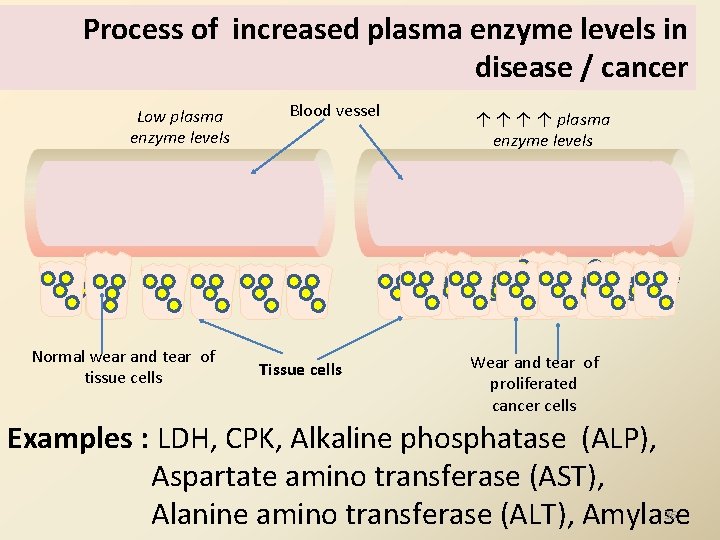
Process of increased plasma enzyme levels in disease / cancer Low plasma enzyme levels Normal wear and tear of tissue cells Blood vessel Tissue cells ↑ ↑ plasma enzyme levels Wear and tear of proliferated cancer cells Examples : LDH, CPK, Alkaline phosphatase (ALP), Aspartate amino transferase (AST), Alanine amino transferase (ALT), Amylase 36
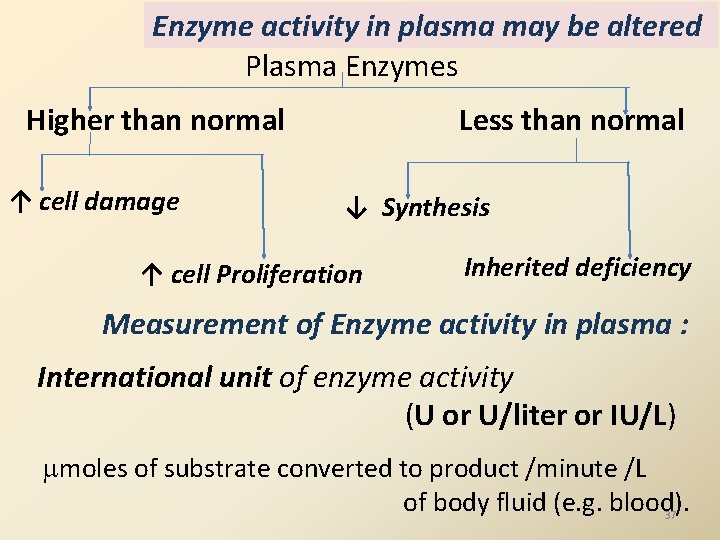
Enzyme activity in plasma may be altered Plasma Enzymes Higher than normal ↑ cell damage Less than normal ↓ Synthesis ↑ cell Proliferation Inherited deficiency Measurement of Enzyme activity in plasma : International unit of enzyme activity (U or U/liter or IU/L) moles of substrate converted to product /minute /L of body fluid (e. g. blood). 37
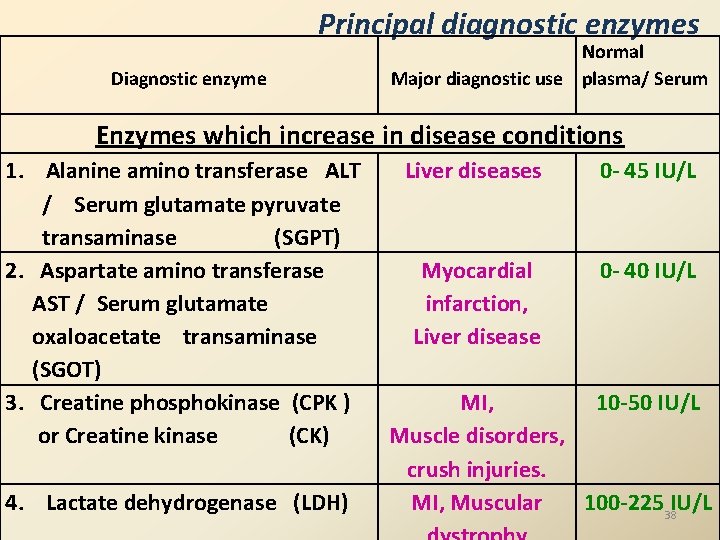
Principal diagnostic enzymes Diagnostic enzyme Normal Major diagnostic use plasma/ Serum Enzymes which increase in disease conditions 1. Alanine amino transferase ALT / Serum glutamate pyruvate transaminase (SGPT) 2. Aspartate amino transferase AST / Serum glutamate oxaloacetate transaminase (SGOT) 3. Creatine phosphokinase (CPK ) or Creatine kinase (CK) 4. Lactate dehydrogenase (LDH) Liver diseases 0 - 45 IU/L Myocardial infarction, Liver disease 0 - 40 IU/L MI, 10 -50 IU/L Muscle disorders, crush injuries. MI, Muscular 100 -22538 IU/L
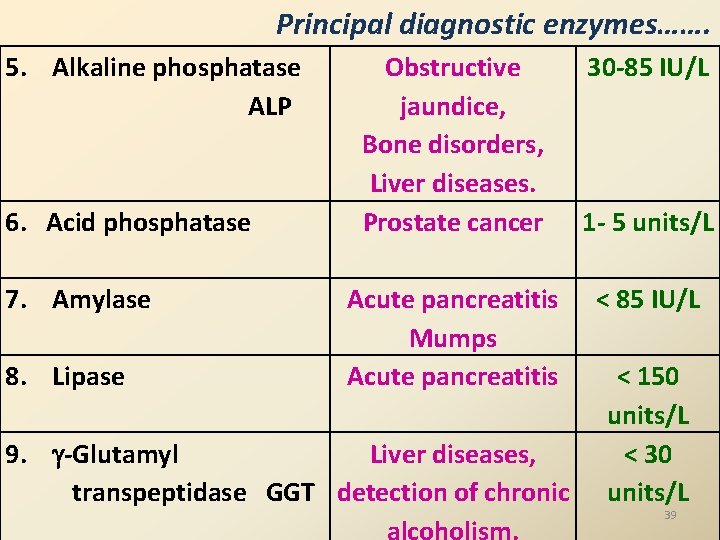
Principal diagnostic enzymes……. 5. Alkaline phosphatase ALP 6. Acid phosphatase 7. Amylase 8. Lipase Obstructive jaundice, Bone disorders, Liver diseases. Prostate cancer Acute pancreatitis Mumps Acute pancreatitis 9. -Glutamyl Liver diseases, transpeptidase GGT detection of chronic alcoholism. 30 -85 IU/L 1 - 5 units/L < 85 IU/L < 150 units/L < 30 units/L 39

Principal diagnostic enzymes……. Enzymes which decrease in disease conditions 10. Ceruloplasmin (serum Wilson’s disease ferroxidase) (enzyme level is decreased) 25 -43 mg/d. L. ENZYME PROFILE IN DISEASES (PLASMA ENZYME PATTERNS IN DISEASE) For accurate diagnosis of a particular disease more than one serum enzyme are routinely estimated, instead of a single enzyme. 40

Disease Myocardial Infarction Liver disease Enzyme profile in diseases. Enzymes 1) CPK-2 (CK-MB) 2) AST 3) LDH-1 (H 4) 1) ALT 2) AST 3) ALP- increase is seen in obstructive jaundice / infective hepatitis / alcoholic hepatitis / hepato-cellular carcinoma. 4) GGT- is useful in the diagnosis of alcoholic liver disease 41 5) LDH-5 (M 4)

Enzyme profile in diseases………. Muscle disorders 1) CK-3 (MM) 2) LDH-5 (M 4) 3) AST Bone diseases 1) Alkaline Phosphatase • Paget’s disease • Rickets • Osteomalacia • Osteoblastoma. 42

B. THERAPEUTIC APPLICATIONS OF ENZYMES OR THERAPEUTIC ENZYMES: Few human diseases, specific enzymes are administered to patients for treatment purpose. Examples : 1. Streptokinase / urokinase / tissue plasminogen activator (t. PA) - are proteases. Use in Treatment : dissolve intravascular clots in Myocardial Infarction (MI) and Deep Vein Thrombsis (DVT). 43
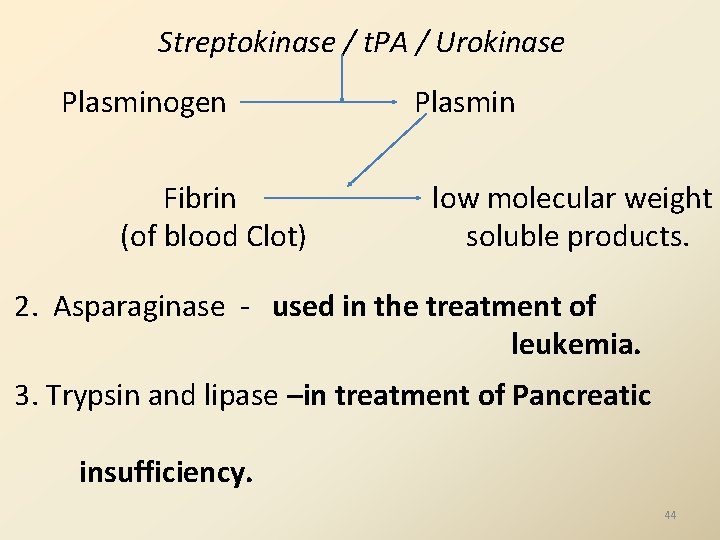
Streptokinase / t. PA / Urokinase Plasminogen Fibrin (of blood Clot) Plasmin low molecular weight soluble products. 2. Asparaginase - used in the treatment of leukemia. 3. Trypsin and lipase –in treatment of Pancreatic insufficiency. 44

C. ENZYMES AS LABORATORY REAGENTS a) Estimation of concentrations of certain substances in body fluids (plasma, CSF etc), b) DNA analysis techniques and c) ELISA technique Principle Specific enzyme Reagents Product Substances Colored compound Measured by colorimetry 45

Examples Substances estimated Enzymes used Plasma glucose Glucose oxidase and peroxidase Serum cholesterol Cholesterol oxidase Serum triglycerides Lipase Blood Urease 46

b) DNA analysis techniques: Restriction endonucleases are useful tools DNA polymerases in DNA analysis DNA ligases techniques. Use : diagnosis of genetic diseases, infective diseases, etc. 47

c) ELISA technique Utilizes enzymes as tools along with antibodies in the measurement of certain proteins -hormones, viruses, etc. 48

THANK YOU 49
- Slides: 49