Enzymes o Catalyst A substance that or the


Enzymes o Catalyst: A substance that ↑ or ↓ the rate of a chemical reactions without consumed or affect the end point of the reaction. o Enzymes: - Thermo-labile Organic catalyst Needed in traces in the body remain unchanged during the reaction Characters: 1 - Protein in nature, forming colloids of high molecular weight 2 - highly specific 3 - Produced by living cells but can act outside them 4 - acts intra- or extra-cellular

Enzymes o Enzymes Terminology: according to the following systems enzymes are named 1 - For hydrolases: name of the substrate (substance acted upon the enzyme) + ase e. g. urease, lipase, sucrase, lactase. 2 - For other enzymes: name of substrate + mechanism of action (succinate dehydrogenase) that removes hydrogen from succinic acid.

Chemical nature of enzymes o Enzymes are protein in nature, they are divided chemically into: 1 - Simple protein: consisting wholly of proteins (pepsin, amylase, lipase) 2 - Conjugated protein: (Holoenzymes) a. Protein part (apoenzymes) thermo-labile, Nondialiasable b. Non-ptn part: Loosely attached (coenzyme) or firmly attached (prothetic group)
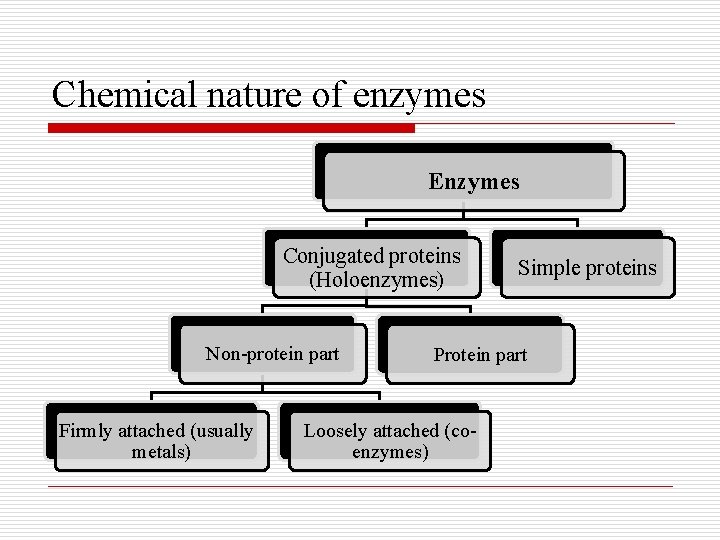
Chemical nature of enzymes Enzymes Conjugated proteins (Holoenzymes) Non-protein part Firmly attached (usually metals) Simple proteins Protein part Loosely attached (coenzymes)
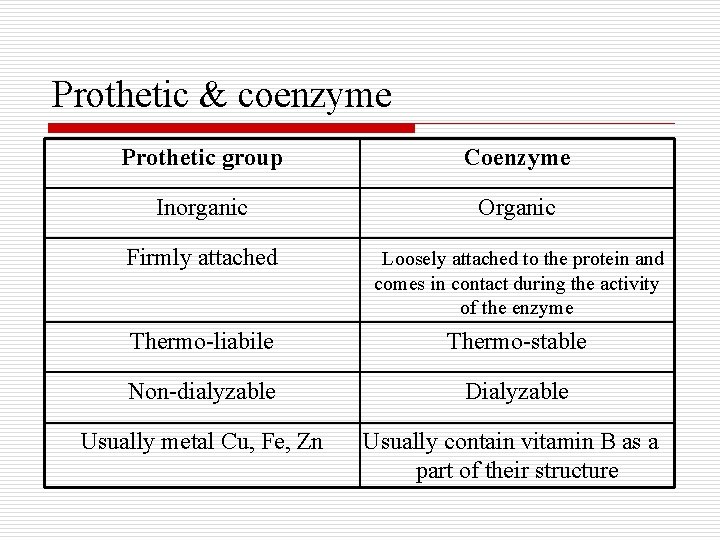
Prothetic & coenzyme Prothetic group Coenzyme Inorganic Organic Firmly attached Loosely attached to the protein and comes in contact during the activity of the enzyme Thermo-liabile Thermo-stable Non-dialyzable Dialyzable Usually metal Cu, Fe, Zn Usually contain vitamin B as a part of their structure

Examples of coenzymes o NAD: nicotinamide adenine dineuclotide. Nicotinamide + adenine + 2 ribose + 2 phosphoric acid. It acts as a hydrogen carrier. o NADP: nicotinamide adenine dineuclotide phosphate. Nicotinamide + adenine + 2 ribose + 3 phosphoric acid. It acts as a hydrogen carrier. o Co. Q: (ubiquinone): it is the quinone used in electron transport and oxidative phosphorylation. o Co. ASH: it is the coenzyme responsible for activation of fatty acids. o FMN: flavin mononeuclotide. Flavin + ribitol +phosphoric acid. It acts as a hydrogen carrier. o FAD: favin adenine dineuclotide. Riboflavin + phosphate + AMP. It acts as a hydrogen carrier.

Mechanism of enzyme action o Lowering of activation energy - for a reaction to occur, the substrate should be placed at a high energy level i. e. activation energy which is to be supplied to the reaction. - Enzymes are lower this activation energy. - Example: activation energy required for hyrolysis of sucrose is 26, 000 calories/mol, when sucrase enzyme is used the activation energy is reduced to 9, 000 calories/mol.
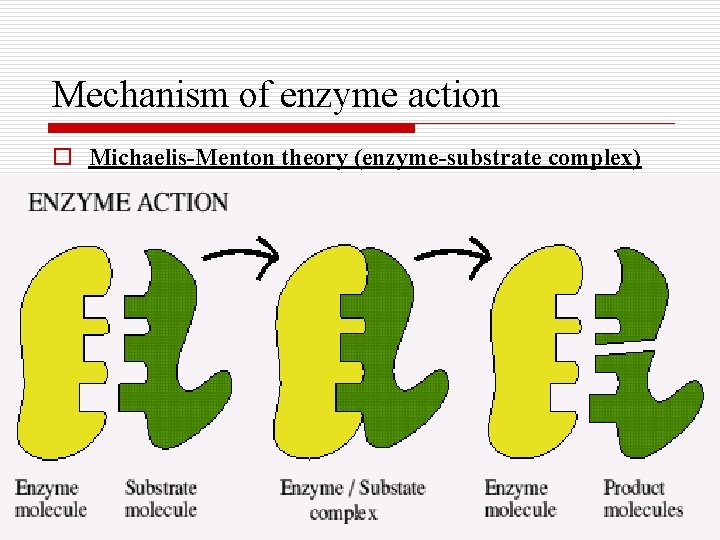
Mechanism of enzyme action o Michaelis-Menton theory (enzyme-substrate complex)

Mechanism of enzyme action o Active center: Certain areas of the enzyme will be functionally more important and named the active site. This area can unite with the substrate forming an E-S- complex.
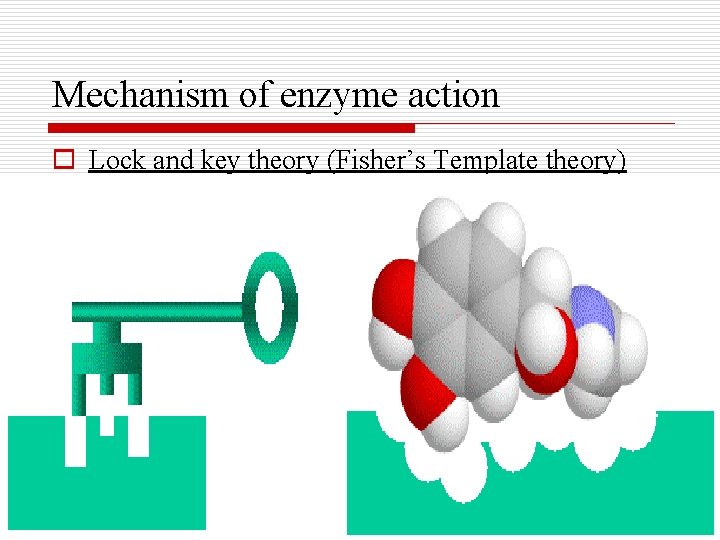
Mechanism of enzyme action o Lock and key theory (Fisher’s Template theory)

Mechanism of enzyme action o Some enzymes are secreted in an inactive form (extracellular enzymes), these are known as zymogen or proenzymes. The active site of these zymogen are masked by polypeptide chains. These proenzymes must be activated first by removal of these polypeptide chains by either: HCl p. H change : pepsinogen → pepsin Autocatalysis: trypsinogen → trypsin Kinases: trypsinogen → trypsin kinases

Factors affecting enzyme activity o Nature of the enzyme (isoenzyme or isozyme) - Catalyze the same reaction. - Differ in structure, properties, and the rate of enzymatic activity. - The best example is lactate dehydrogenase (LDH), which is by electrophoresis is separated into 5 isoenzymes.

Lactate dehydrogenase (LDH) o Consists of 4 polypeptide chains( tetramers) of 2 types H, M o Type I: HHHH→ heart Type II: HHHM Type III: HHMM Type IV : HMMM Type IV : MMMM → muscle and liver
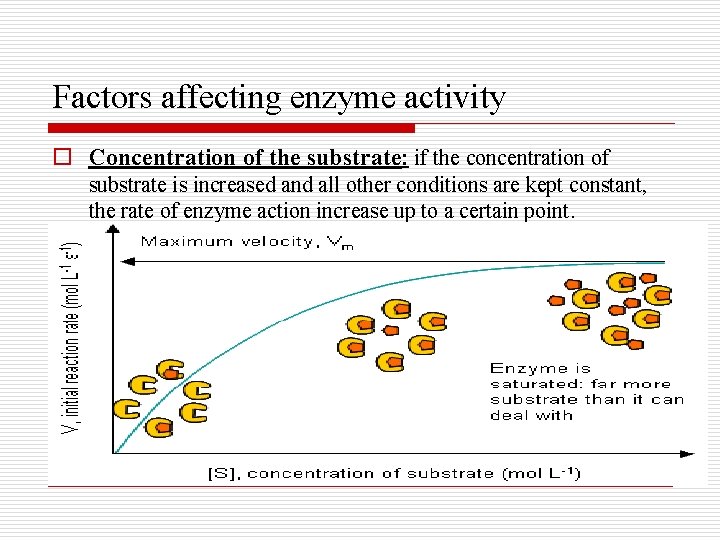
Factors affecting enzyme activity o Concentration of the substrate: if the concentration of substrate is increased and all other conditions are kept constant, the rate of enzyme action increase up to a certain point.
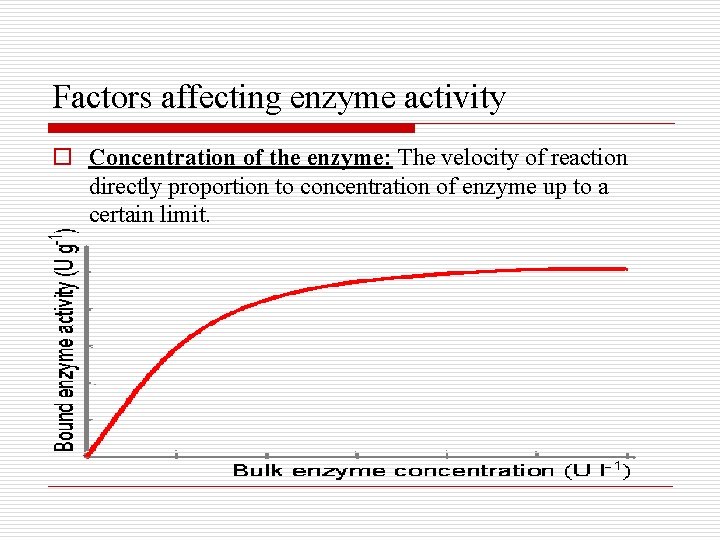
Factors affecting enzyme activity o Concentration of the enzyme: The velocity of reaction directly proportion to concentration of enzyme up to a certain limit.
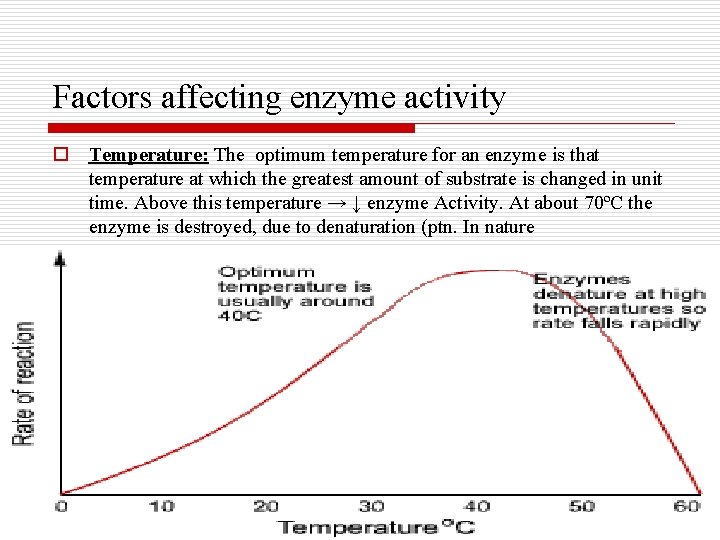
Factors affecting enzyme activity o Temperature: The optimum temperature for an enzyme is that temperature at which the greatest amount of substrate is changed in unit time. Above this temperature → ↓ enzyme Activity. At about 70ºC the enzyme is destroyed, due to denaturation (ptn. In nature
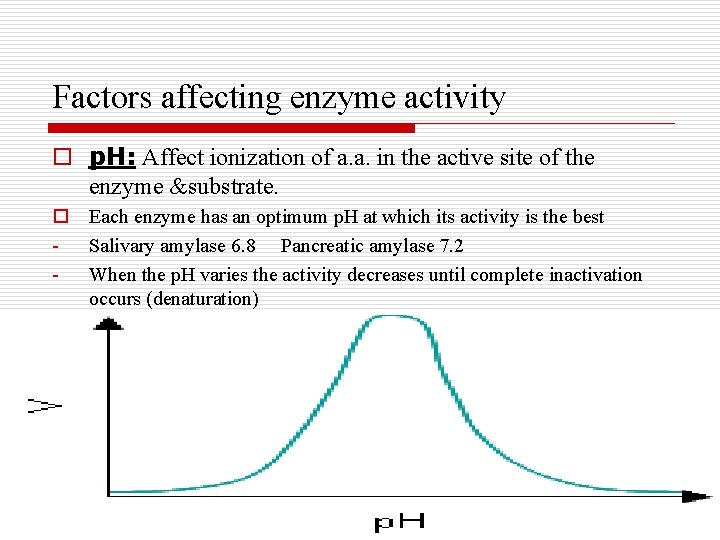
Factors affecting enzyme activity o p. H: Affect ionization of a. a. in the active site of the enzyme &substrate. o Each enzyme has an optimum p. H at which its activity is the best - Salivary amylase 6. 8 Pancreatic amylase 7. 2 - When the p. H varies the activity decreases until complete inactivation occurs (denaturation)

Factors affecting enzyme activity o concentration of the reaction products A→ B + C - the velocity of the reaction is inversely proportion to the Enzyme concentration of the reaction products. q Effect of time - activity of the enzyme is directly proportion to time within limits.

Factors affecting enzyme activity q Enzyme activators: - are inorganic ions which increases the activity of an enzyme. - choloride ions activate amylase enzyme - Mg ions activate kinase enzyme - Ca ions activate thrombokinase enzyme

Factors affecting enzyme activity o Enzyme inhibitors: A. 1) 2) - Non specific inhibitors; affect a wide variety of enzymes Agents which precipitate or denaturate protein. Sulphahydryl inhibtors: Oxidizing agents as K ferricynaide → oxidizing SH → S— S Alkalyating agents as iodoacetic acid → block SH Salts of heavy metals (Hg, Pb) → mercaptides
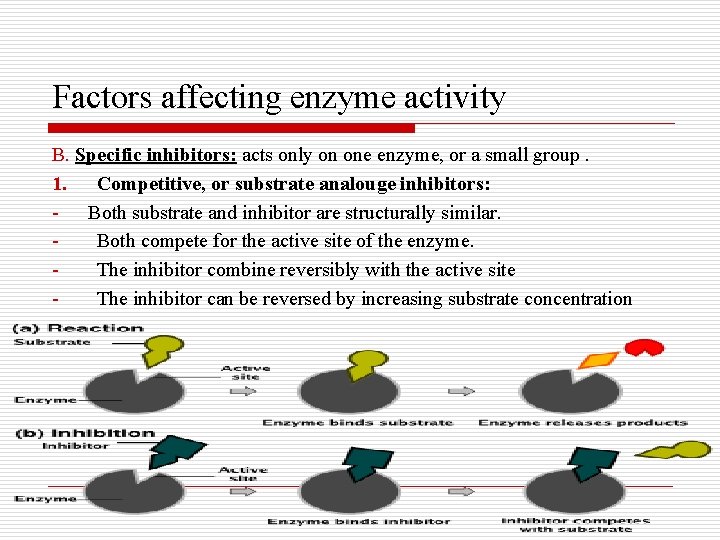
Factors affecting enzyme activity B. Specific inhibitors: acts only on one enzyme, or a small group. 1. Competitive, or substrate analouge inhibitors: Both substrate and inhibitor are structurally similar. Both compete for the active site of the enzyme. The inhibitor combine reversibly with the active site The inhibitor can be reversed by increasing substrate concentration
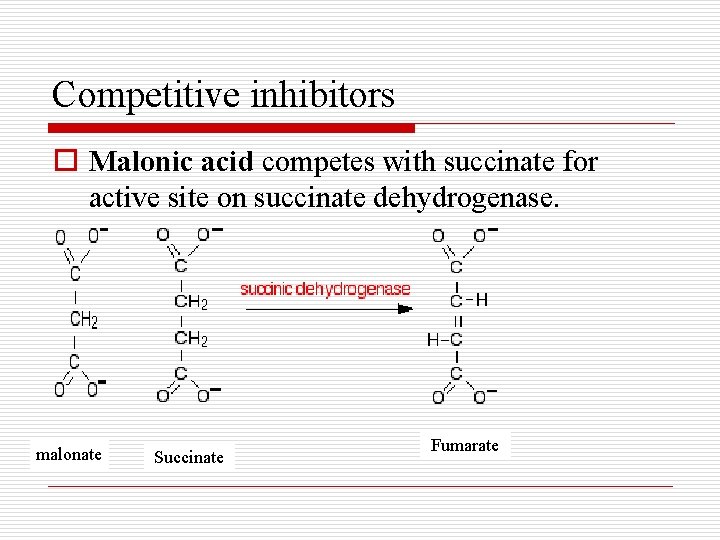
Competitive inhibitors o Malonic acid competes with succinate for active site on succinate dehydrogenase. malonate Succinate Fumarate
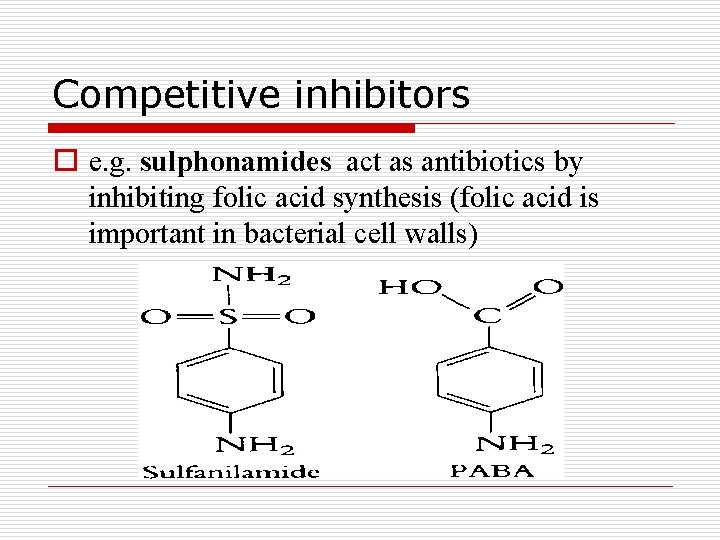
Competitive inhibitors o e. g. sulphonamides act as antibiotics by inhibiting folic acid synthesis (folic acid is important in bacterial cell walls)
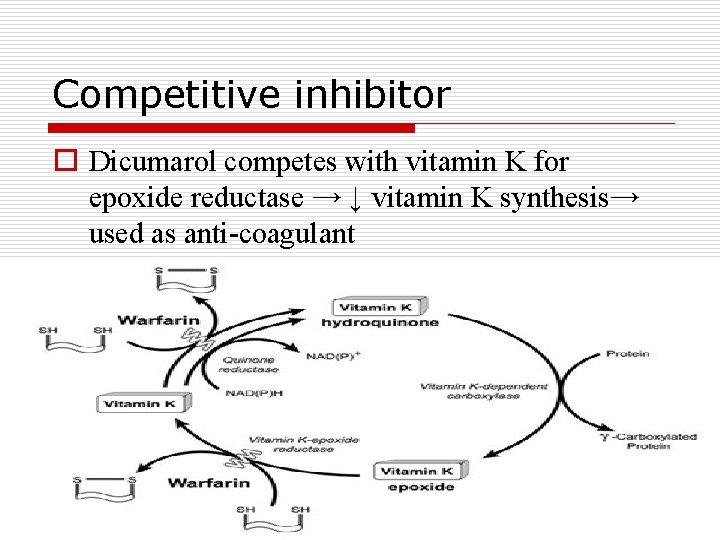
Competitive inhibitor o Dicumarol competes with vitamin K for epoxide reductase → ↓ vitamin K synthesis→ used as anti-coagulant
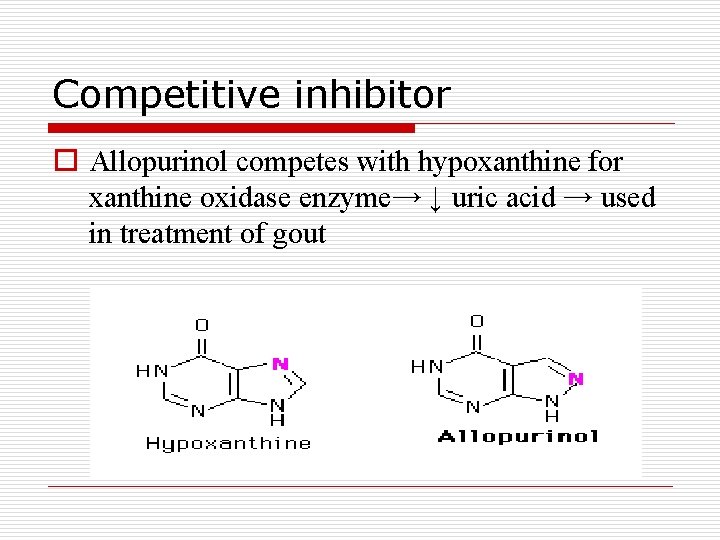
Competitive inhibitor o Allopurinol competes with hypoxanthine for xanthine oxidase enzyme→ ↓ uric acid → used in treatment of gout

Non-competitive inhibitors o No structural similarity between the substrate and inhibitors. o No competition for the active site 1 - Allosteric modifiers Are small organic molecules which have regulatory role on enzyme activity.

Allosteric modification o The inhibitor binds with the apoenzyme (allosteric site) far from the active site o Reversible, when the modifier is consumed (not by increasing the substrate concentration)→ return to the normal

Allosteric modification o. Mechanism: The reversible binding of the allosteric inhibitor produce conformational change in the apo-enzyme, so binding of the substrate to the active site become difficult. o Example; ATP & citrate are allosteric inhibitors of PFK 1, G -6 -P is allosteric inhibitor of hexokinase.

Allosteric modifiers o Sometimes, binding of the allosteric modifiers to the apo-enzyme produce conformational change, making the active site more fit for the substrate (Allosteric activators). o Example; AMP, ADP are allosteric activators of PFK 1
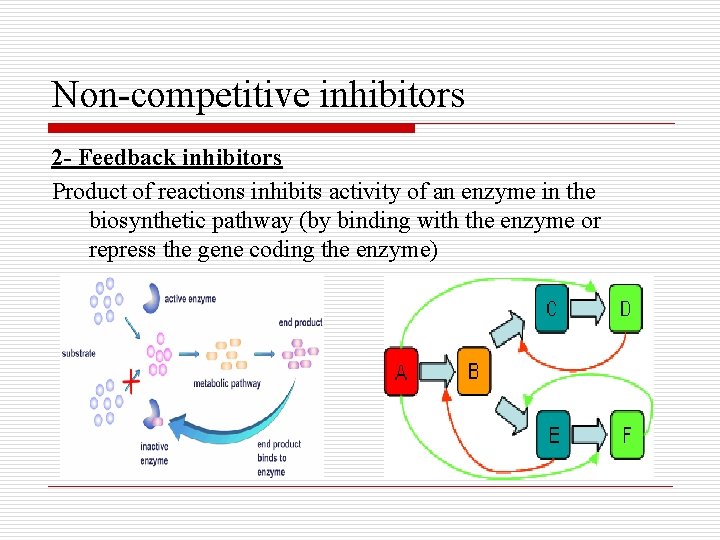
Non-competitive inhibitors 2 - Feedback inhibitors Product of reactions inhibits activity of an enzyme in the biosynthetic pathway (by binding with the enzyme or repress the gene coding the enzyme)

Non-competitive inhibitors 3 - inhibitors blocking activatos and coenzymes -Agents which block the coenzyme will stop enzyme action i. e. phenylhydrazine block aldhyde group of PLP (coenzyme for transaminase, decarboxylases) -Agents which block the prothetic group will stop enzyme action i. e. -cynaide and CO block iron of heme in cytochrome oxidase.

Non-competitive inhibitors 4 - Antienzymes & antibodies: -Antienzymes: as ascaris worm secrete anti-pepsin and anti-trypsin to prevent digestion of worm by these enzymes. -Antibodies: if enzymes are injected, the immune system of the body produce antibodies to inactivate these enzymes.

- Slides: 34