Enzymes Most biological catalysts are some REALLY COOL

Enzymes Most biological catalysts are ______ (some REALLY COOL ONES are folded RNAs!!!) Catalysts Catalyst does not alter equilibrium Enzyme Nonenzymatic reaction rate (s-1) Enzymatic reaction rate (s-1) Rate enhancement Carbonic anhydrase 1. 3 x 10 -1 1 x 106 7. 7 x 106 Triose phosphate isomerase 4. 3 x 10 -6 4300 1 x 109 Staphlococcal nuclease 1. 7 x 10 -13 95 5. 6 x 1014
Enzymes E + S ES E + P Highly specific Reaction occurs in ______ of enzyme Substance acted upon = _____ Resulting species = _______ Enzyme acts on forward and reverse reactions Activity depends on protein’s native structure Regulated - by concentrations of substrate and substances other than substrate

Enzymes Cofactors Functional groups of protein enzymes are involved in acid-base reactions, covalent bond formation, charge-charge interactions BUT they are less suitable for oxid-reduc and group-transfer reactions SO they use _________ (inorganic ions) COFACTORS may be metal ions (Cu 2+, Fe 3+, Zn 2+) trace amounts of metal needed in our diets

Enzymes Cofactors COFACTORS can be organic or metalloorganic molecules --> COENZYMES Examples: NAD+ Heme Holoenzyme = Apoenzyme (inactive) + cofactor/coenzyme/metal ions
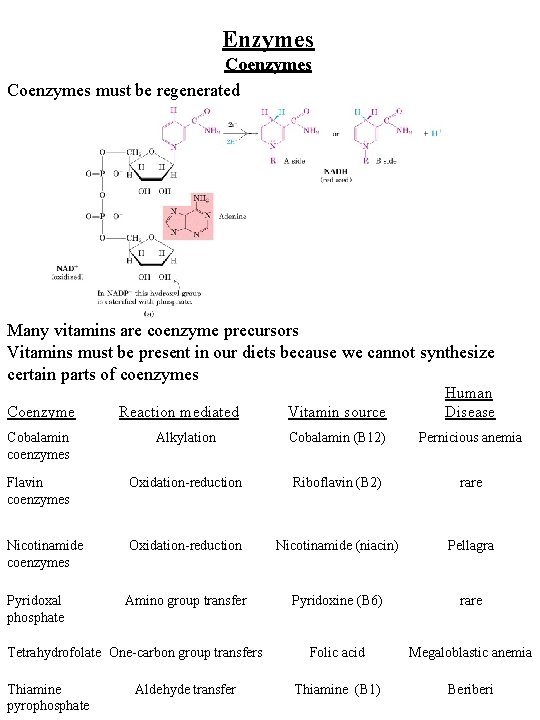
Enzymes Coenzymes must be regenerated Many vitamins are coenzyme precursors Vitamins must be present in our diets because we cannot synthesize certain parts of coenzymes Coenzyme Reaction mediated Vitamin source Human Disease Cobalamin coenzymes Alkylation Cobalamin (B 12) Pernicious anemia Flavin coenzymes Oxidation-reduction Riboflavin (B 2) rare Nicotinamide coenzymes Oxidation-reduction Nicotinamide (niacin) Pellagra Pyridoxal phosphate Amino group transfer Pyridoxine (B 6) rare Folic acid Megaloblastic anemia Thiamine (B 1) Beriberi Tetrahydrofolate One-carbon group transfers Thiamine pyrophosphate Aldehyde transfer
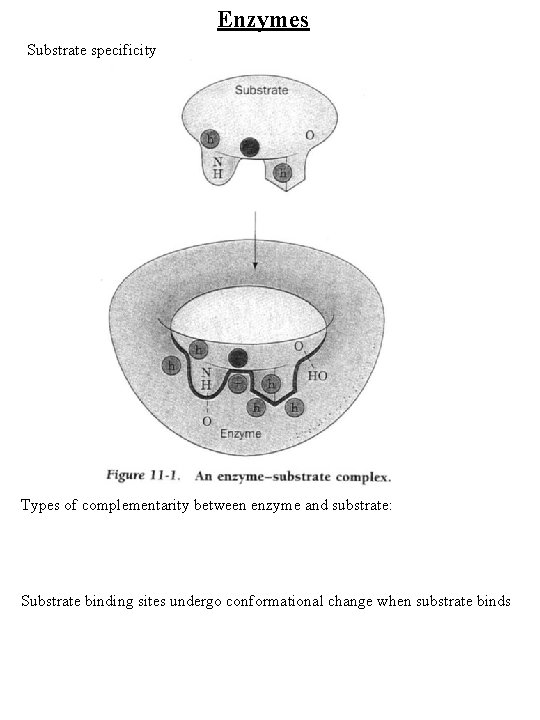
Enzymes Substrate specificity Types of complementarity between enzyme and substrate: Substrate binding sites undergo conformational change when substrate binds
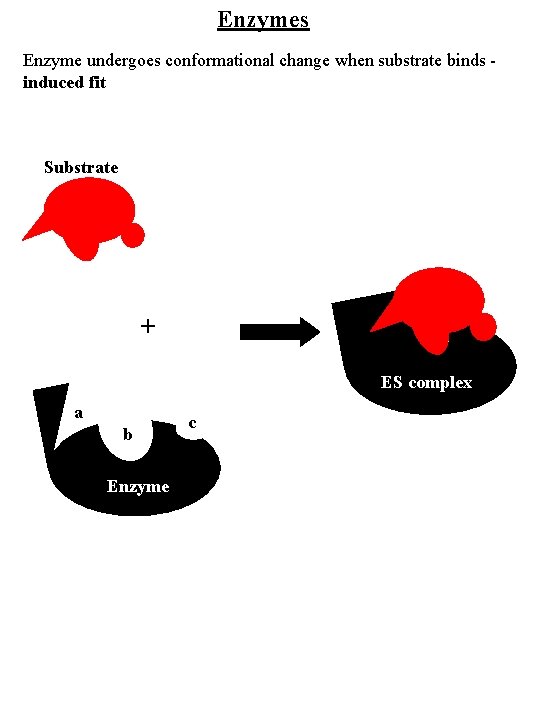
Enzymes Enzyme undergoes conformational change when substrate binds induced fit Substrate a c b a + b ES complex a b Enzyme c c
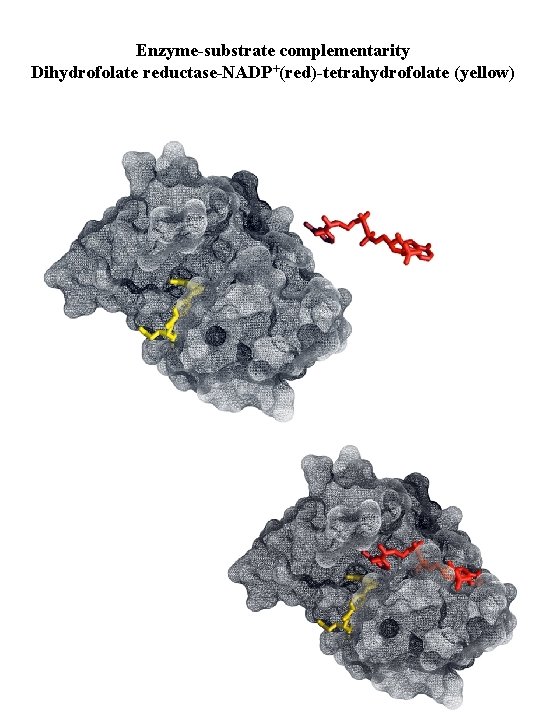
Enzyme-substrate complementarity Dihydrofolate reductase-NADP+(red)-tetrahydrofolate (yellow)

Enzymes Stereospecific Why? Inherently chiral (proteins only consist of L-amino acids) so form asymmetric active sites Example: Protein enzyme Yeast Alcohol dehydrogenase (YADH) YADH CH 3 CH 2 OH + NAD+ Ethanol O CH 3 CH + NADH + H+ Acetaldehyde
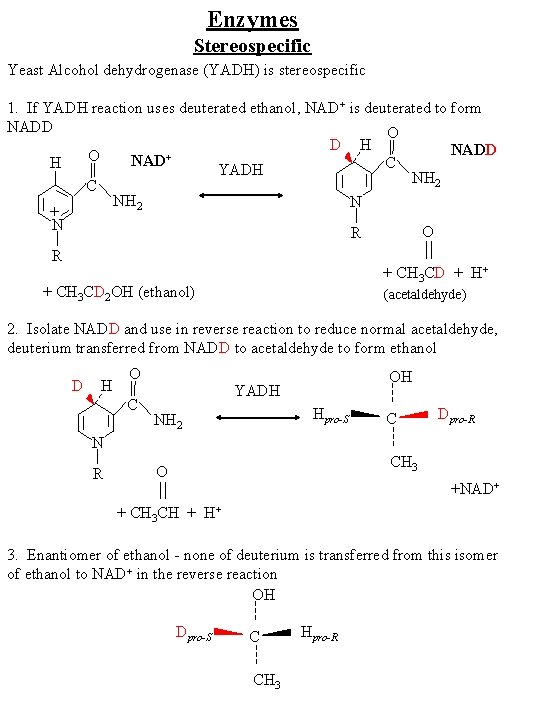
Enzymes Stereospecific Yeast Alcohol dehydrogenase (YADH) is stereospecific 1. If YADH reaction uses deuterated ethanol, NAD+ is deuterated to form NADD O D H NADD O + NAD C H YADH NH 2 C NH 2 N + N O R R + CH 3 CD + H+ + CH 3 CD 2 OH (ethanol) (acetaldehyde) 2. Isolate NADD and use in reverse reaction to reduce normal acetaldehyde, deuterium transferred from NADD to acetaldehyde to form ethanol D H O C OH YADH Hpro-S NH 2 C Dpro-R N R CH 3 O +NAD+ + CH 3 CH + H+ 3. Enantiomer of ethanol - none of deuterium is transferred from this isomer of ethanol to NAD+ in the reverse reaction OH Dpro-S C CH 3 Hpro-R
![Enzyme activity Dependent on: [metal ion], p. H, temperature, [enzyme], [substrate] Enzyme activity Dependent on: [metal ion], p. H, temperature, [enzyme], [substrate]](http://slidetodoc.com/presentation_image_h/e68d717d9ac472ca29250fac4dd3ff0b/image-11.jpg)
Enzyme activity Dependent on: [metal ion], p. H, temperature, [enzyme], [substrate]
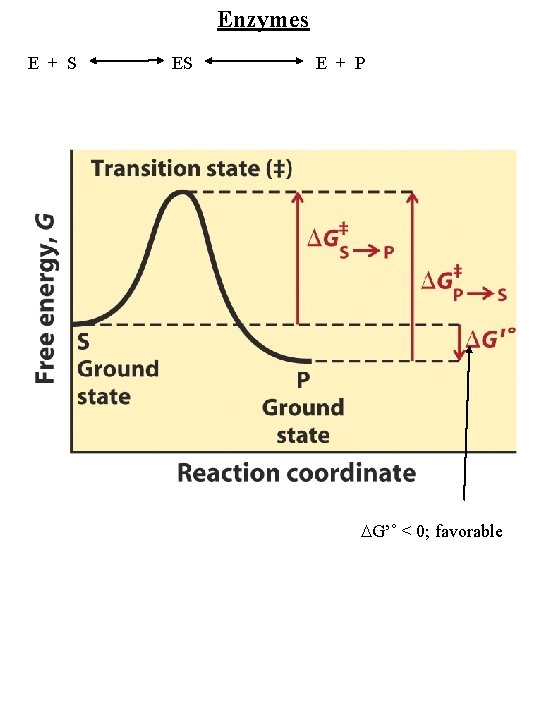
Enzymes E + S ES E + P G’˚ < 0; favorable
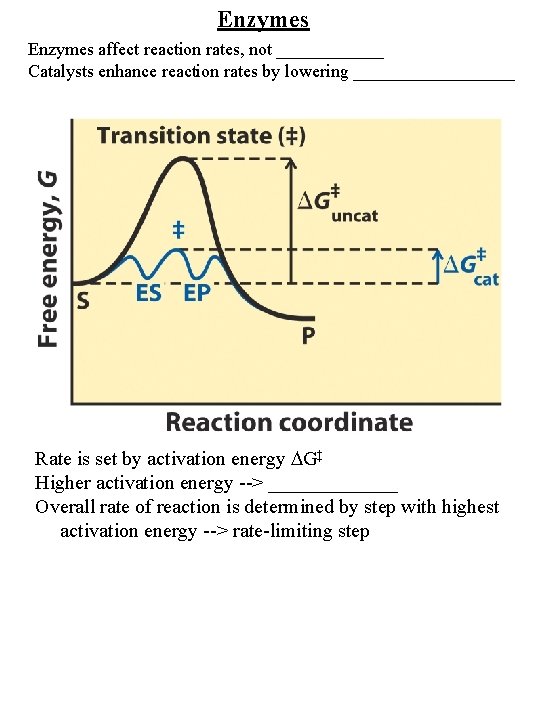
Enzymes affect reaction rates, not ______ Catalysts enhance reaction rates by lowering _________ Rate is set by activation energy G‡ Higher activation energy --> _______ Overall rate of reaction is determined by step with highest activation energy --> rate-limiting step

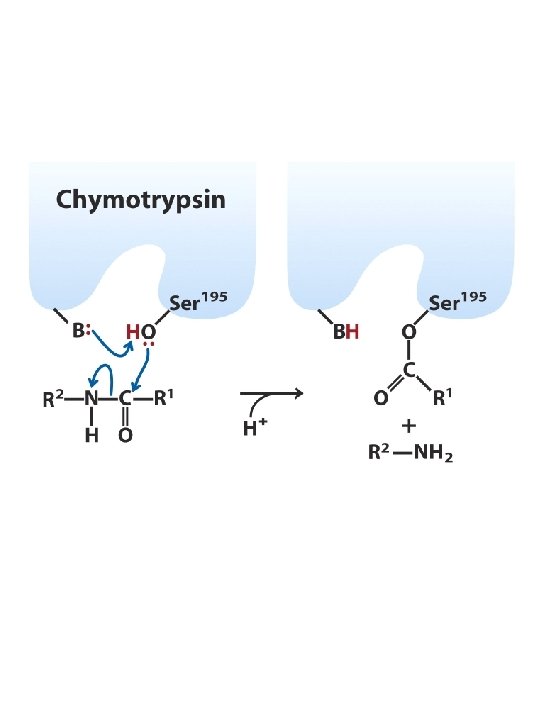
Enzymes General acid-base catalysis General acid catalysis - partial proton transfer from an acid lowers free energy of reaction’s transition state Keto Transition state R Enol R C O H CH 2 A R + H O C A C CH 2 - H H H O H CH 2 A- + General base catalysis - partial proton abstraction by a base lowers free energy of reaction’s transition state Keto Transition state R R C C O O CH 2 - H H B B Enol + + R C H+ O H CH 2 H B
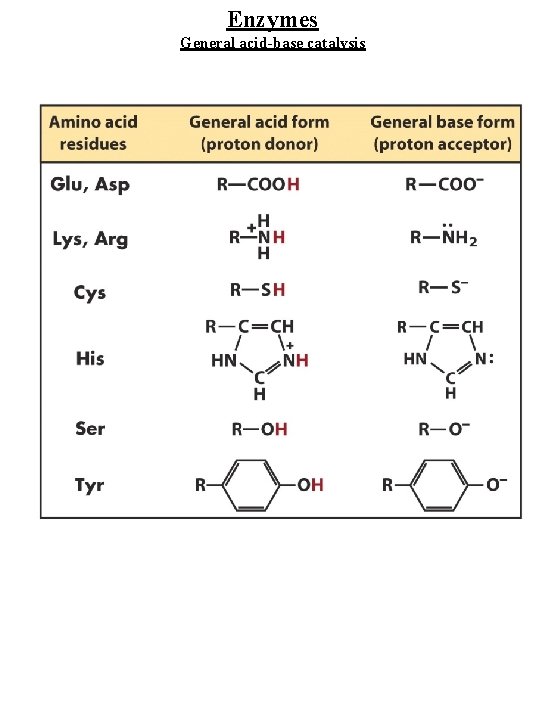
Enzymes General acid-base catalysis
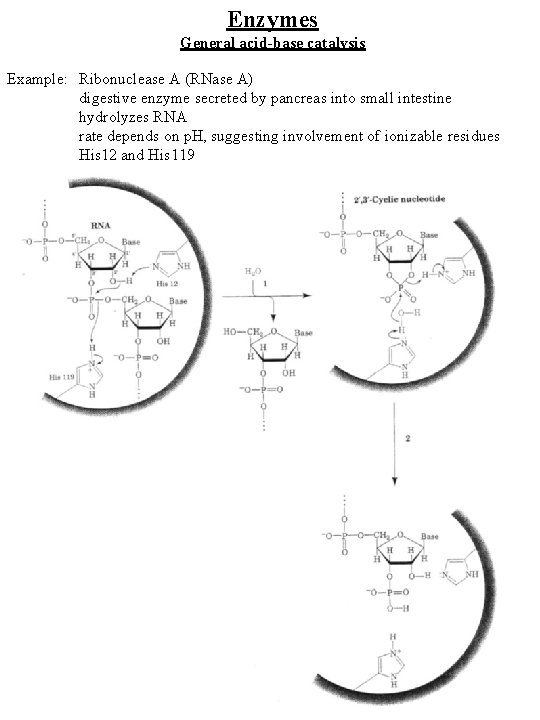
Enzymes General acid-base catalysis Example: Ribonuclease A (RNase A) digestive enzyme secreted by pancreas into small intestine hydrolyzes RNA rate depends on p. H, suggesting involvement of ionizable residues His 12 and His 119
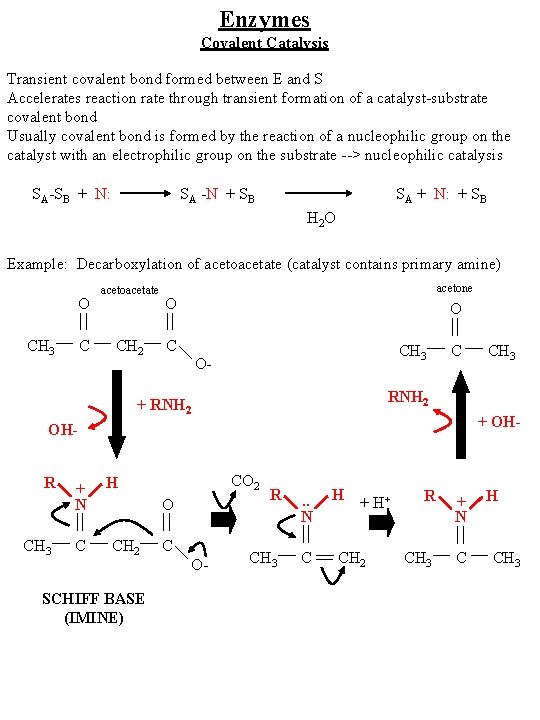
Enzymes Covalent Catalysis Transient covalent bond formed between E and S Accelerates reaction rate through transient formation of a catalyst-substrate covalent bond Usually covalent bond is formed by the reaction of a nucleophilic group on the catalyst with an electrophilic group on the substrate --> nucleophilic catalysis SA-SB + N: SA -N + SB SA + N: + SB H 2 O Example: Decarboxylation of acetoacetate (catalyst contains primary amine) O CH 3 C acetoacetate CH 2 acetone O O C CH 3 O- C RNH 2 + OH- OHR CH 3 + N C CO 2 H O CH 2 SCHIFF BASE (IMINE) CH 3 C O- R . . N CH 3 C H + H+ CH 2 R + N CH 3 C H CH 3
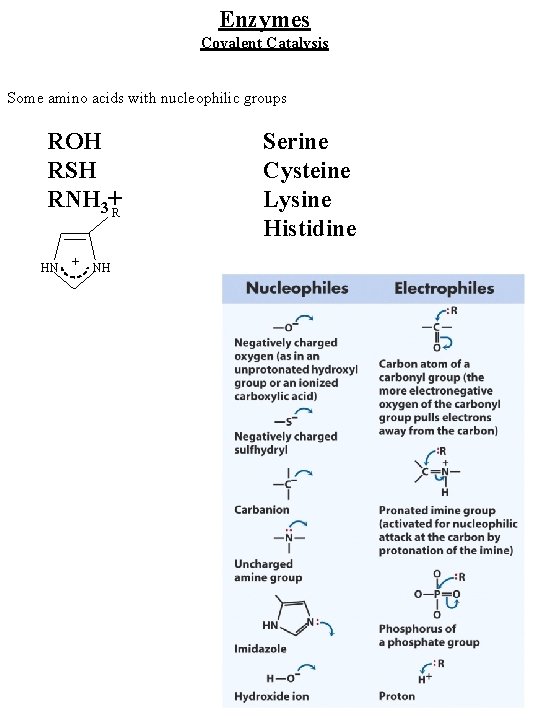
Enzymes Covalent Catalysis Some amino acids with nucleophilic groups ROH RSH RNH 3+R HN + NH Serine Cysteine Lysine Histidine
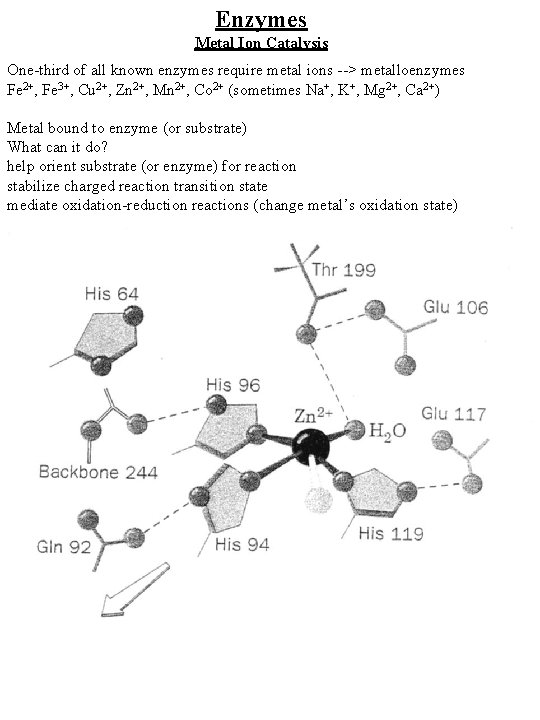
Enzymes Metal Ion Catalysis One-third of all known enzymes require metal ions --> metalloenzymes Fe 2+, Fe 3+, Cu 2+, Zn 2+, Mn 2+, Co 2+ (sometimes Na+, K+, Mg 2+, Ca 2+) Metal bound to enzyme (or substrate) What can it do? help orient substrate (or enzyme) for reaction stabilize charged reaction transition state mediate oxidation-reduction reactions (change metal’s oxidation state) Voet, p. 295 11 -11, scheme
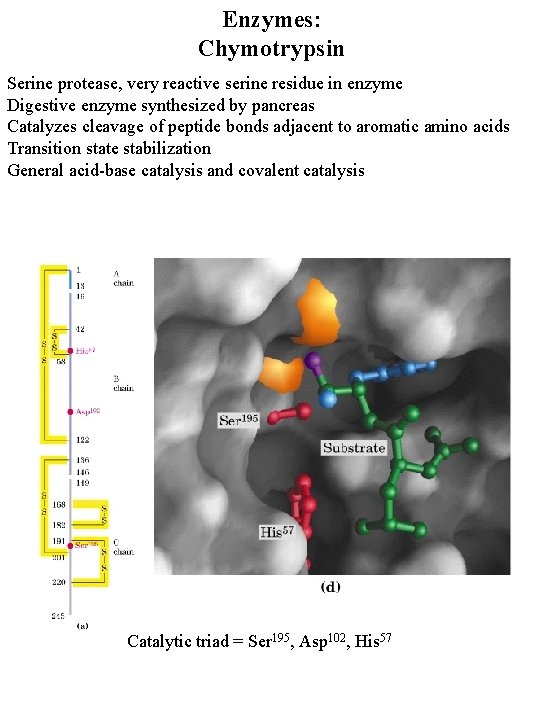
Enzymes: Chymotrypsin Serine protease, very reactive serine residue in enzyme Digestive enzyme synthesized by pancreas Catalyzes cleavage of peptide bonds adjacent to aromatic amino acids Transition state stabilization General acid-base catalysis and covalent catalysis Catalytic triad = Ser 195, Asp 102, His 57
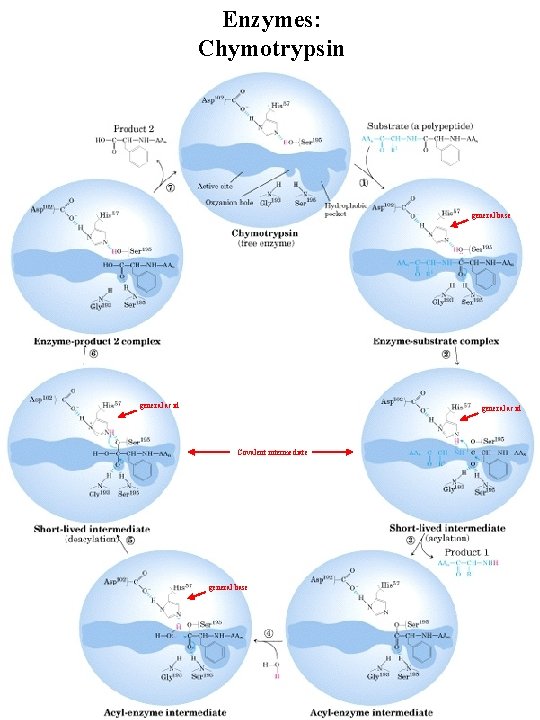
Enzymes: Chymotrypsin general base general acid Covalent intermediate general base
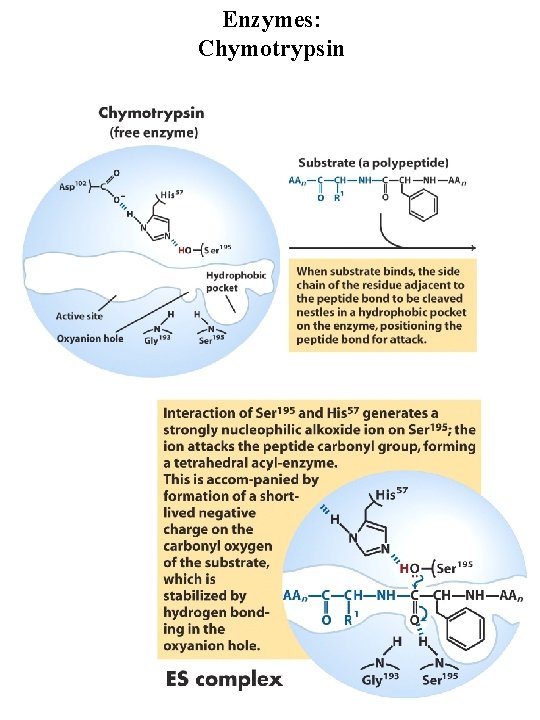
Enzymes: Chymotrypsin
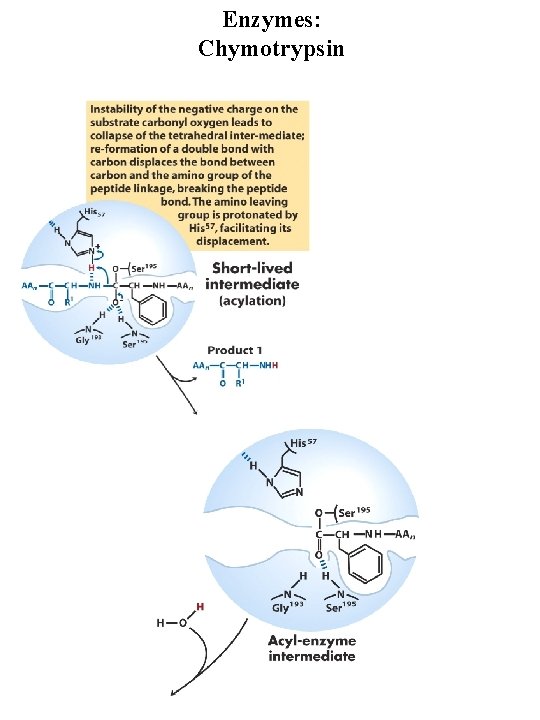
Enzymes: Chymotrypsin
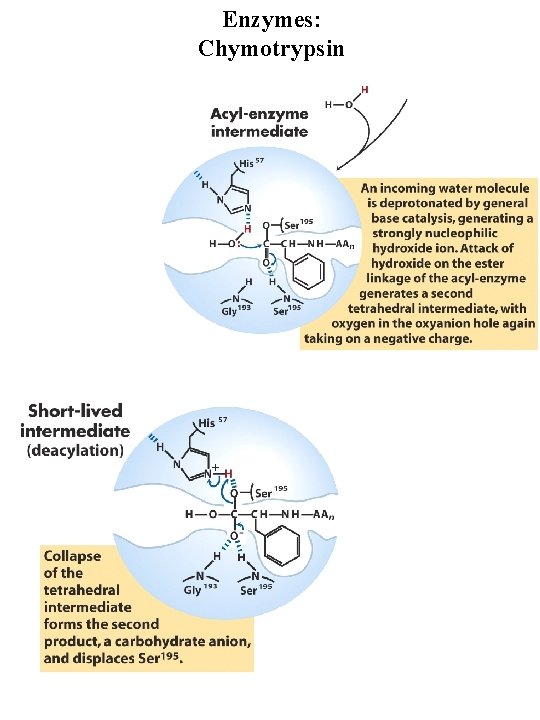
Enzymes: Chymotrypsin
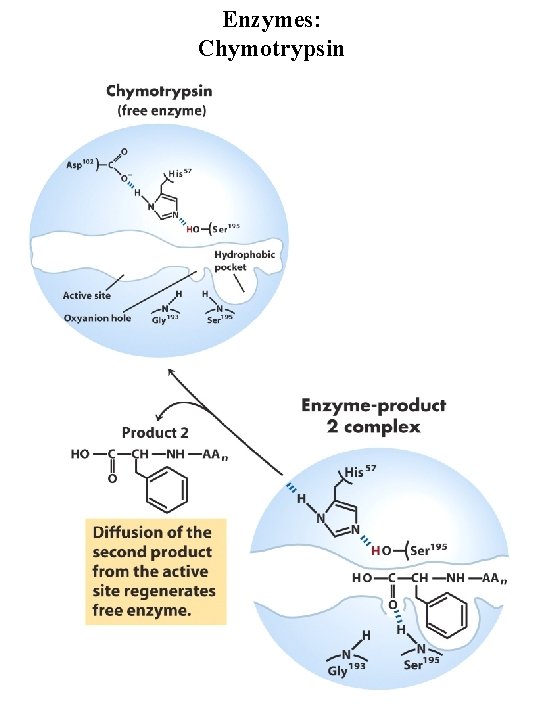
Enzymes: Chymotrypsin
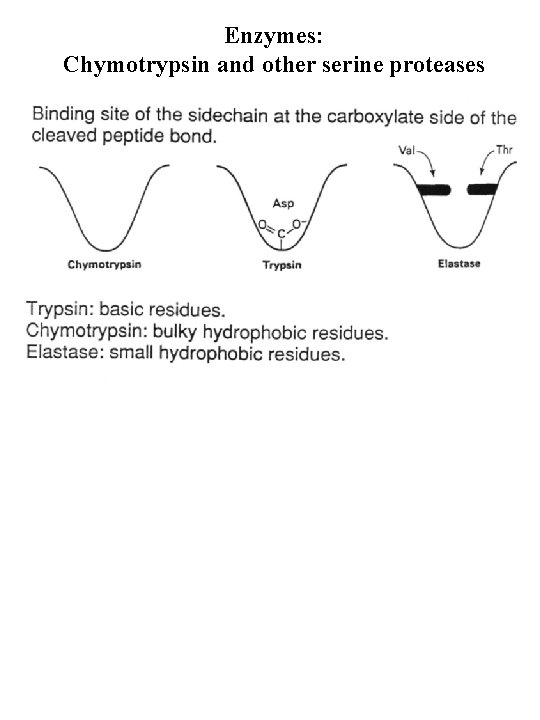
Enzymes: Chymotrypsin and other serine proteases
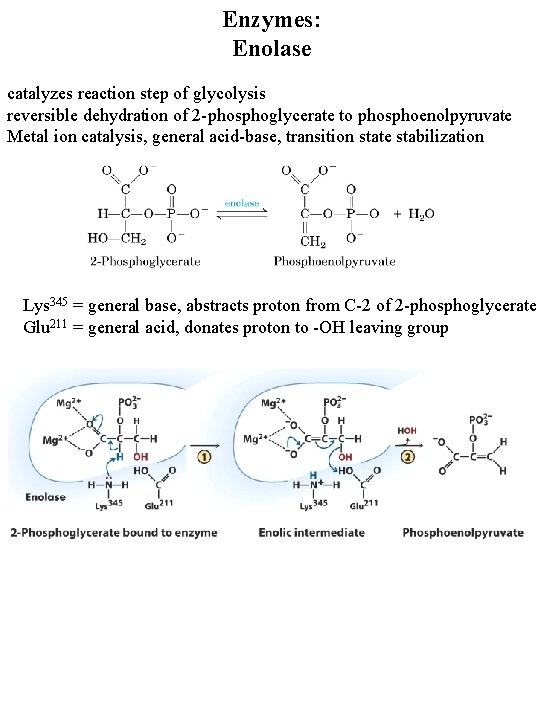
Enzymes: Enolase catalyzes reaction step of glycolysis reversible dehydration of 2 -phosphoglycerate to phosphoenolpyruvate Metal ion catalysis, general acid-base, transition state stabilization Lys 345 = general base, abstracts proton from C-2 of 2 -phosphoglycerate Glu 211 = general acid, donates proton to -OH leaving group
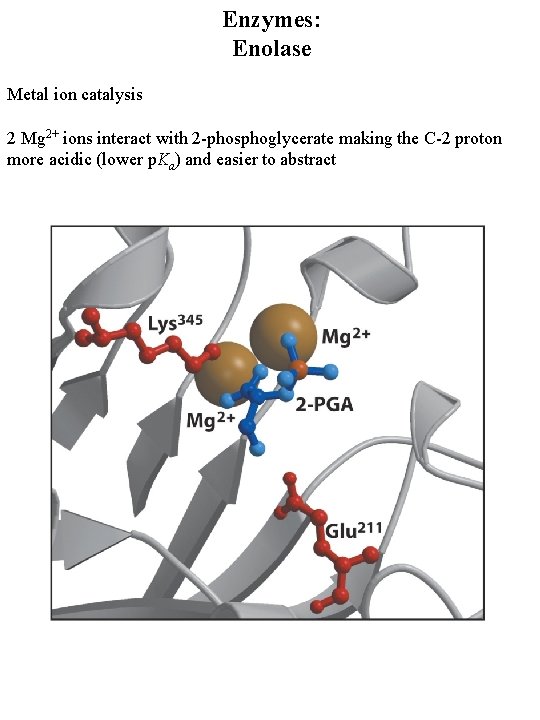
Enzymes: Enolase Metal ion catalysis 2 Mg 2+ ions interact with 2 -phosphoglycerate making the C-2 proton more acidic (lower p. Ka) and easier to abstract
- Slides: 29