Enzymes Kinetics and Inhibition Enzymes n How do

Enzymes Kinetics and Inhibition

Enzymes n How do we characterize an enzyme? – Biological catalyst – Highly specific – Mostly proteins

Enzyme Classification n Common name – Not always informative about reaction catalyzed » Ex. Chymotrypsin n Systematic name – Enzyme Commission – 6 major classes » Ex. Transferases n Classification number – EC 2. 7. 3. 2 2=transferase, 7=phosphate, 3=nitrogen group as acceptor, 2=unique

Enzyme Classification

Enzymes n What are enzyme cofactors? – Non-protein component – Organic group or metal When is a cofactor a prosthetic group? n Holoenzyme versus apoenzyme n

Enzymes n How do metal ion cofactors function? – Bridging group, coordination complex – Stabilize conformation in active form n Vitamins are parts of coenzymes – Thiamine pyrophosphate – Nicotinamide adenine dinucleotide – Pyridoxal phosphate

Enzymes and Free Energy n What does G tell us about the nature of a reaction? n What does it mean if G is = 0? n What is true if G is positive?
![Calculating Free Energy A+B C+D n G = G 0 + RT ln[C][D] / Calculating Free Energy A+B C+D n G = G 0 + RT ln[C][D] /](http://slidetodoc.com/presentation_image_h2/6cac537fc4f0a80be41b5fb53764d409/image-8.jpg)
Calculating Free Energy A+B C+D n G = G 0 + RT ln[C][D] / [A][B] ln n G 0 = standard free energy change; A, B, C, D present at 1 M n G 0/ = at p. H 7 n
![Calculating Free Energy At equilibrium 0 = G 0/+ RT ln[C][D] / ln [A][B] Calculating Free Energy At equilibrium 0 = G 0/+ RT ln[C][D] / ln [A][B]](http://slidetodoc.com/presentation_image_h2/6cac537fc4f0a80be41b5fb53764d409/image-9.jpg)
Calculating Free Energy At equilibrium 0 = G 0/+ RT ln[C][D] / ln [A][B] n Since Keq = [C][D]/ [A][B] n We can get n – G 0/ = -2. 303 RT log 10 Keq
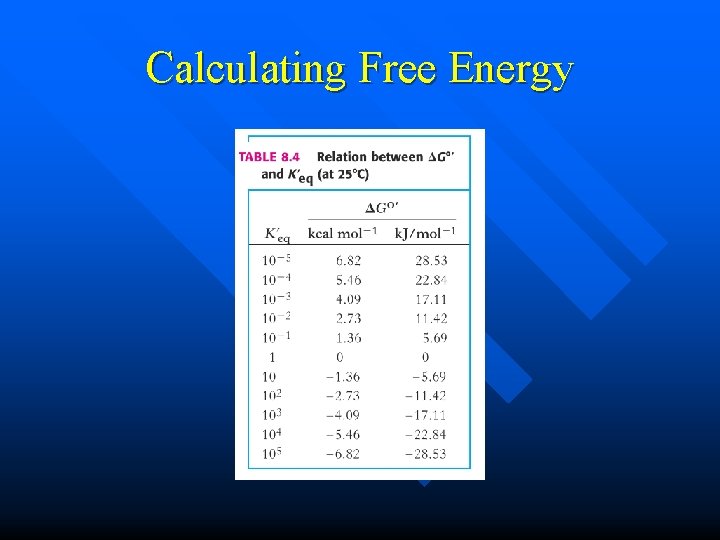
Calculating Free Energy

Enzymes n Do not change equilibrium of reactions, speed up both forward and reverse reactions n Equilibrium depends upon free energy difference between products and reactants
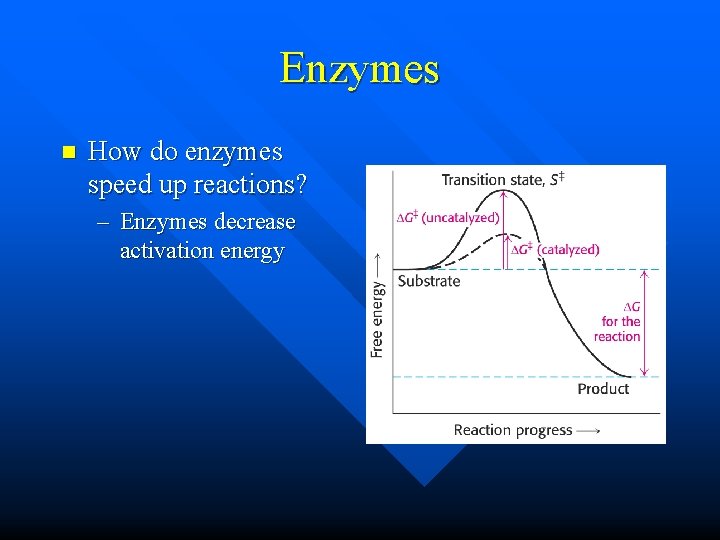
Enzymes n How do enzymes speed up reactions? – Enzymes decrease activation energy
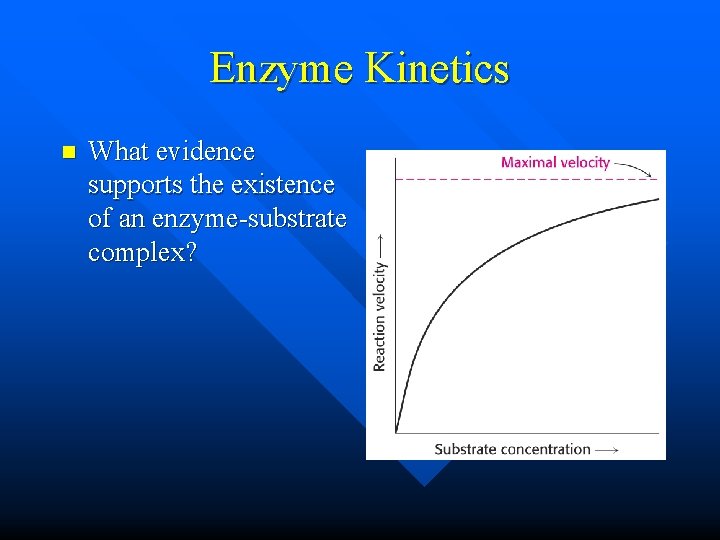
Enzyme Kinetics n What evidence supports the existence of an enzyme-substrate complex?
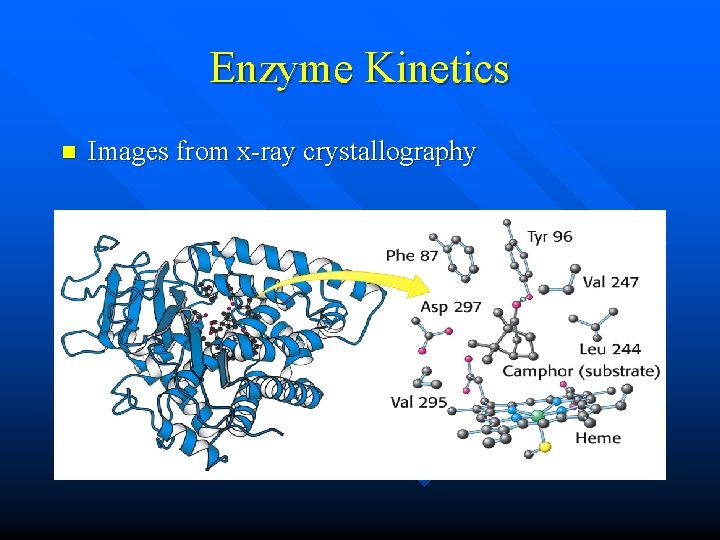
Enzyme Kinetics n Images from x-ray crystallography
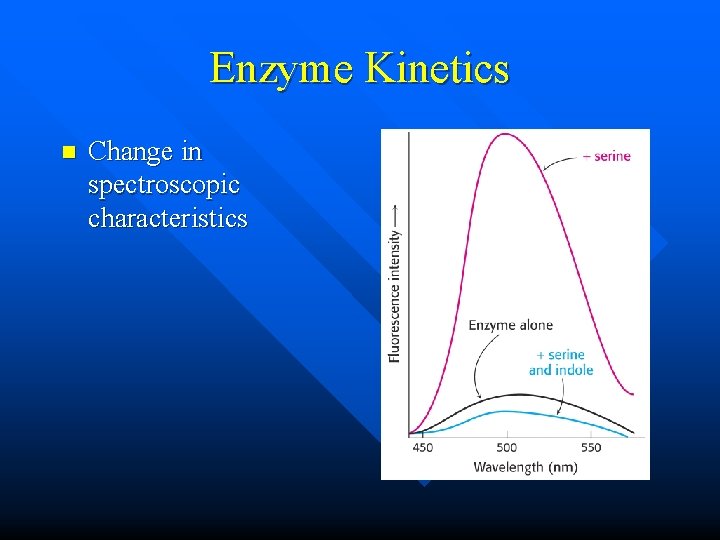
Enzyme Kinetics n Change in spectroscopic characteristics

Enzyme Kinetics n What do we know about the active site of an enzyme? – where substrate binds – three-dimensional cleft – small part of enzyme – substrate attached by weak bonds
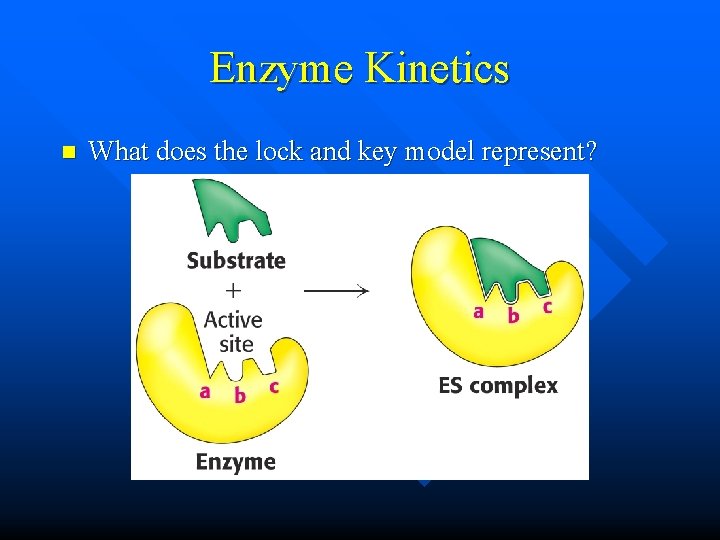
Enzyme Kinetics n What does the lock and key model represent?
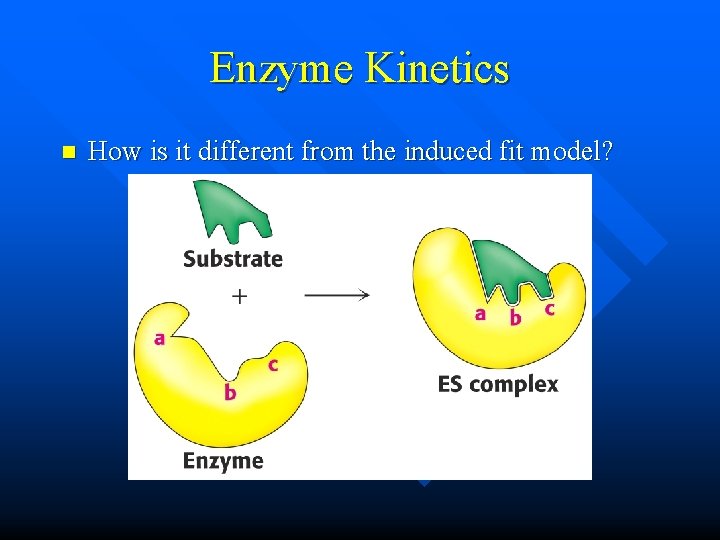
Enzyme Kinetics n How is it different from the induced fit model?

Michaelis –Menten Equation n Enzyme E combines with substrate S to form enzyme substrate complex ES and ES breaks down to form E and product P E + S k 1 k-1 ES k 2 P+E

Michaelis-Menten Equation n Initial velocity equals rate of breakdown of ES § V 0 = k 2 [ES] § Must define ES in other terms § Formation of ES = k 1[E][S] § Breakdown of ES = (k-1 + k 2 )[ES] § At steady state: k 1[E][S] = (k-1 + k 2 )[ES]
![Michaelis-Menten Equation n Rearrange equation § [E][S]/[ES] = (k-1 + k 2 )/k 1 Michaelis-Menten Equation n Rearrange equation § [E][S]/[ES] = (k-1 + k 2 )/k 1](http://slidetodoc.com/presentation_image_h2/6cac537fc4f0a80be41b5fb53764d409/image-21.jpg)
Michaelis-Menten Equation n Rearrange equation § [E][S]/[ES] = (k-1 + k 2 )/k 1 § Define new term Km § Michaelis-Menten constant § Km = (k-1 + k 2 )/k 1 we now substitute in above and solve for [ES] § [ES] =[E][S]/Km § [E] = [E]T – [ES]
![Michaelis-Menten Equation n Substitute for[E] § [ES] =([E]T – [ES])[S]/Km § Solve for [ES] Michaelis-Menten Equation n Substitute for[E] § [ES] =([E]T – [ES])[S]/Km § Solve for [ES]](http://slidetodoc.com/presentation_image_h2/6cac537fc4f0a80be41b5fb53764d409/image-22.jpg)
Michaelis-Menten Equation n Substitute for[E] § [ES] =([E]T – [ES])[S]/Km § Solve for [ES] § [ES] = [E]T [S]/[S] + Km Going back to previous equation v 0=k 2[ES] n V 0 = k 2 [E]T [S]/[S] + Km n Maximal rate Vmax is when [ES] =[E]T n V 0 = Vmax [S]/[S] + Km n
![Michaelis-Menten Equation V 0 = Vmax [S]/[S] + Km n When initial velocity = Michaelis-Menten Equation V 0 = Vmax [S]/[S] + Km n When initial velocity =](http://slidetodoc.com/presentation_image_h2/6cac537fc4f0a80be41b5fb53764d409/image-23.jpg)
Michaelis-Menten Equation V 0 = Vmax [S]/[S] + Km n When initial velocity = ½ maximal velocity n § Km = [S]
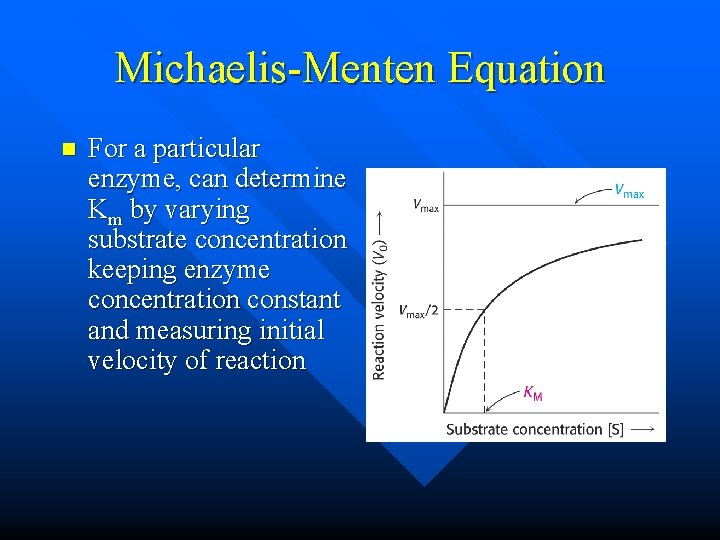
Michaelis-Menten Equation n For a particular enzyme, can determine Km by varying substrate concentration keeping enzyme concentration constant and measuring initial velocity of reaction
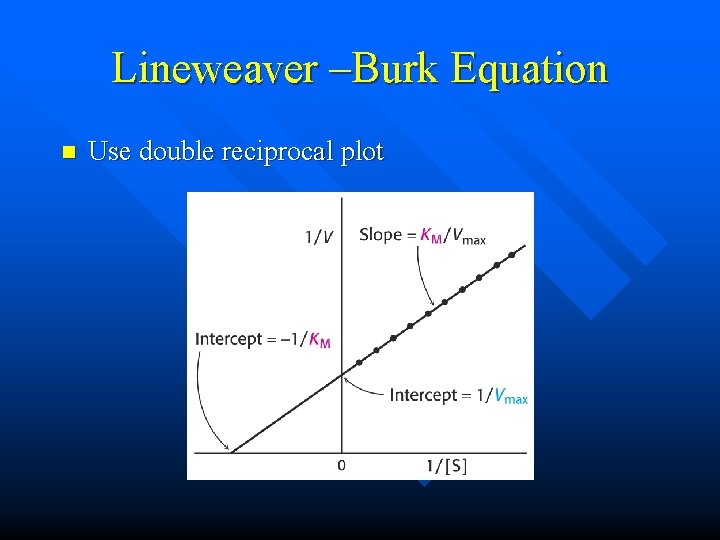
Lineweaver –Burk Equation n Use double reciprocal plot

Enzyme Kinetics n What do we know about Km? – Not a fixed value – Varies with structure of substrate – Varies with p. H, temperature, ionic strength – Each substrate has characteristic Km – Km usually between 10 -1 and 10 -6 M – Derived from rate constants

Enzyme Kinetics n If k-1 is considerable larger than k 2 so that Km = k-1/k 1 this represents the dissociation constant of ES complex Therefore, Km tells us about strength of ES complex high Km = weak bonding; low Km = strong bonding

Enzyme Kinetics n What does Vmax represent? § Turnover number § What is the kinetic constant or kcat? § Kcat = Vmax/[E]0 = initial conc. Enzyme § # of substrate molecules converted into product in unit time when enzyme is saturated § When [S] Km, kcat/Km can be used as measure of catalytic efficiency
- Slides: 28