Enzymes Enzymes as Biological Catalysts Nomenclature and Names

Enzymes § § § § Enzymes as Biological Catalysts Nomenclature and Names of Enzymes Nature of Enzymes Enzyme Cofactors Classification of Enzymes Mechanism of Action Enzyme Specificity Enzyme Catalyzed Reaction

Enzymes § Large molecules which most are proteins § Activity lost if denatured § Catalysts for biological reactions § § Have unique three-dimensional shapes that fit the shapes of reactants. They remain unchanged at the end of the reaction
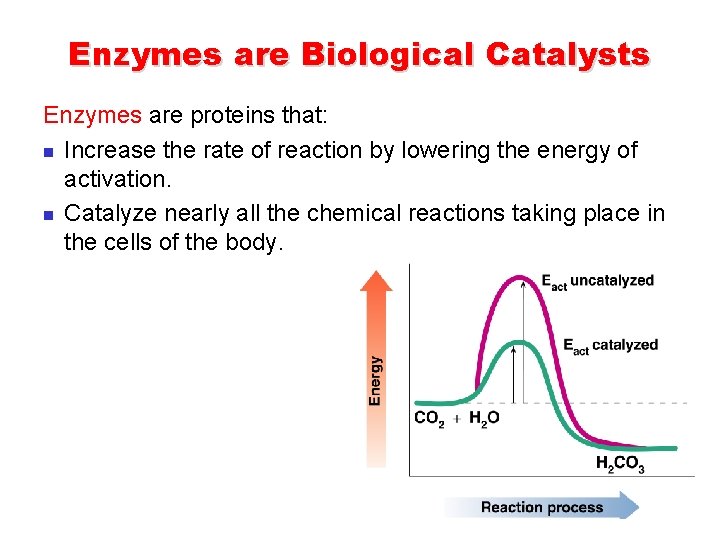
Enzymes are Biological Catalysts Enzymes are proteins that: n Increase the rate of reaction by lowering the energy of activation. n Catalyze nearly all the chemical reactions taking place in the cells of the body.

Nomenclature and Names of Enzymes The name of an enzyme: n Usually ends in –ase. n Identifies the reacting substance. n Sucrase catalyzes the hydrolysis of sucrose. n Lipase catalyzes the hydrolysis of lipids. n Describes the function of the enzyme. n Oxidases catalyze oxidation. n Hydrolases catalyze hydrolysis. n Could be a common name, particularly for the digestion enzymes such as pepsin and trypsin.

Nature of Enzymes § Simple enzymes: composed of whole protein e. g. Ribonuclease § Complex enzymes: composed of protein PLUS a relatively small organic / inorganic compound

Nature of Enzymes Holoenzyme: (Apoenzymes + Cofactor) Cofactors: small non-protein molecules that required for the catalytic activity of the enzyme termed as "helper molecules". It may be: 1) Prosthetic groups are small organic or inorganic molecules that tightly bound to Apoenzyme by covalent bond. Organic like vitamins , while inorganic like metal ions (Fe++, Zn++ or Cu++). 2) Coenzymes: Are organic molecules loosely bound in a dissociable manner to Apoenzyme. It serves as recyclable shuttles that transport substrates from the site of generation to site of utilization e. g FAD , NAD and vitamin B-complex.
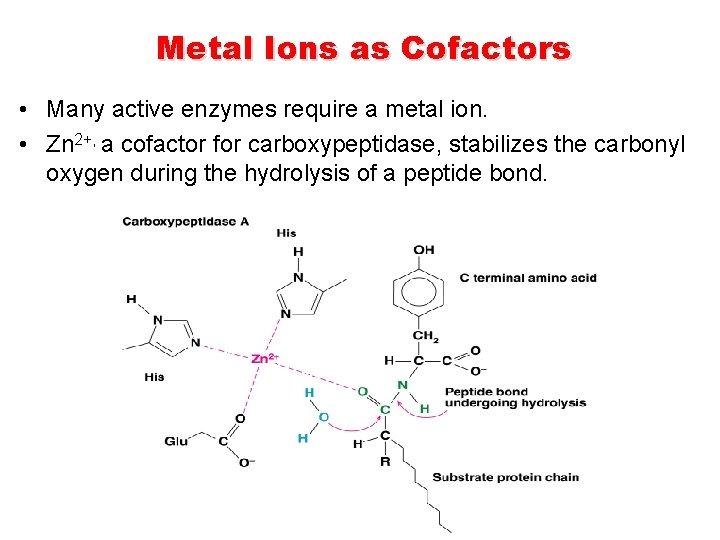
Metal Ions as Cofactors • Many active enzymes require a metal ion. • Zn 2+, a cofactor for carboxypeptidase, stabilizes the carbonyl oxygen during the hydrolysis of a peptide bond.

Some Enzymes and Their Cofactors

Classification of Enzymes are classified according to the reaction they catalyze. Class Reactions catalyzed Oxidoreductases Oxidation-reduction. Add or remove hydrogen atoms. Transferases Transfer groups or atoms between donor and acceptor molecules. Hydrolases Hydrolysis by adding water across a bond. Lyases Add/remove atoms to form a double bond. (Adding ammonia, or carbon dioxide across double bonds or remove them forming double bonds). Isomerases Rearrange atoms Ligases Two chemical groups are joined (or ligated) with the use of energy from ATP

Oxidoreductases and Transferases
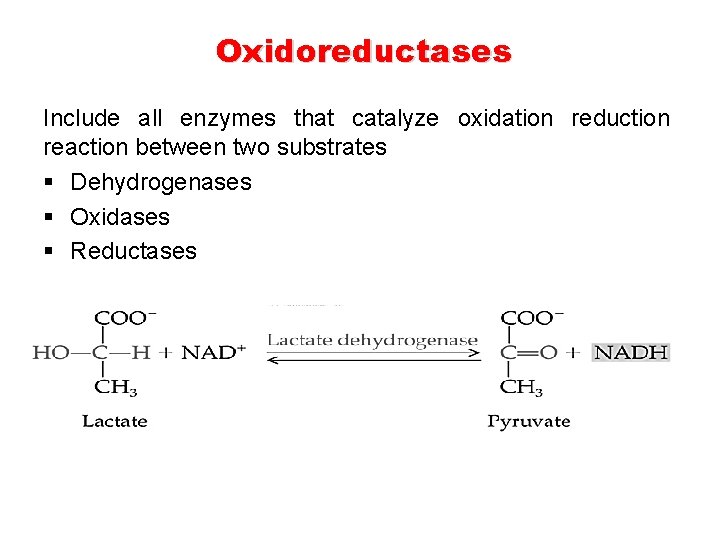
Oxidoreductases Include all enzymes that catalyze oxidation reduction reaction between two substrates § Dehydrogenases § Oxidases § Reductases
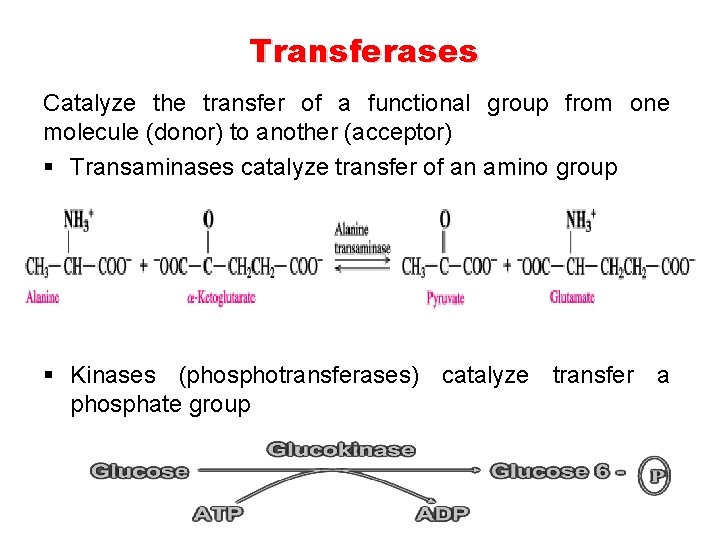
Transferases Catalyze the transfer of a functional group from one molecule (donor) to another (acceptor) § Transaminases catalyze transfer of an amino group § Kinases (phosphotransferases) catalyze transfer a phosphate group
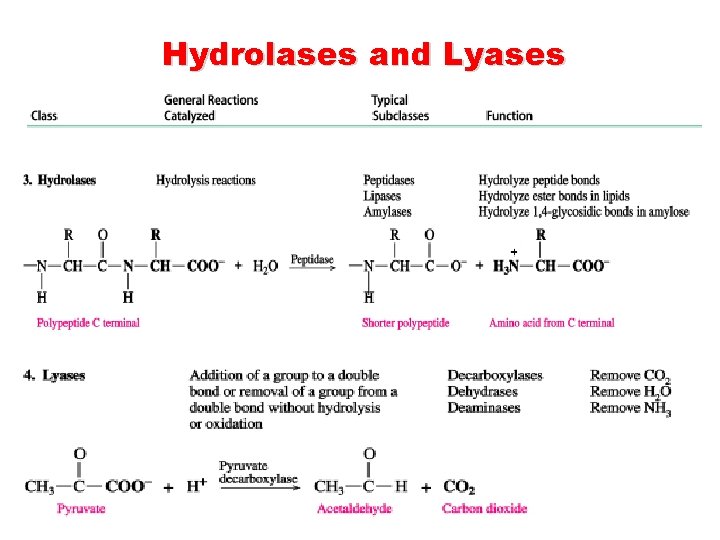
Hydrolases and Lyases
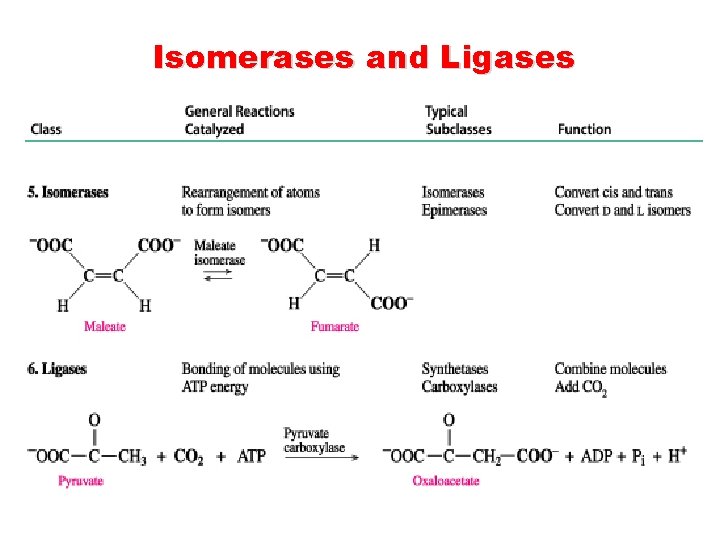
Isomerases and Ligases
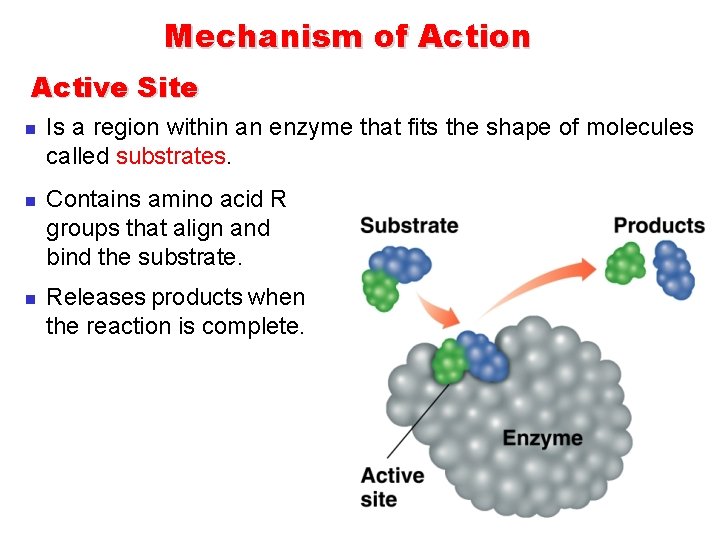
Mechanism of Action Active Site n n n Is a region within an enzyme that fits the shape of molecules called substrates. Contains amino acid R groups that align and bind the substrate. Releases products when the reaction is complete.
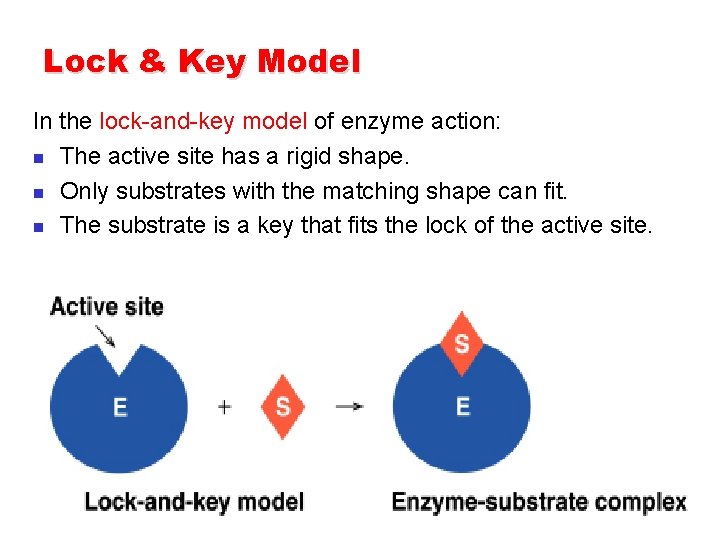
Lock & Key Model In the lock-and-key model of enzyme action: n The active site has a rigid shape. n Only substrates with the matching shape can fit. n The substrate is a key that fits the lock of the active site.
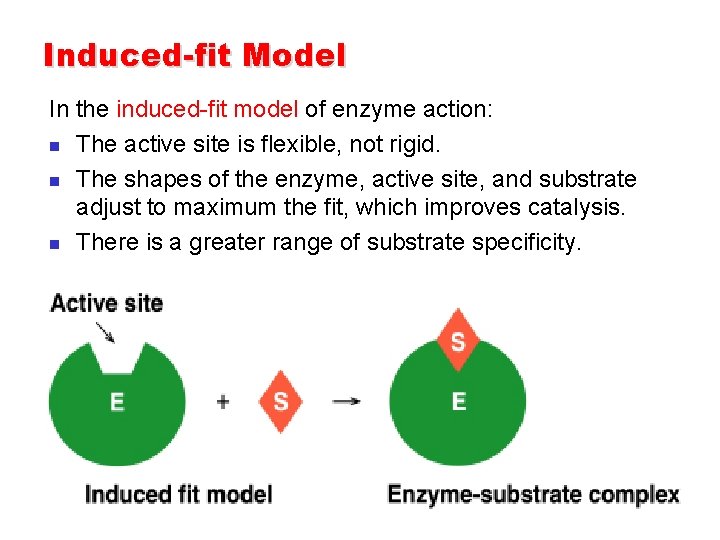
Induced-fit Model In the induced-fit model of enzyme action: n The active site is flexible, not rigid. n The shapes of the enzyme, active site, and substrate adjust to maximum the fit, which improves catalysis. n There is a greater range of substrate specificity.

Enzyme Specificity Enzymes may recognize and catalyze: n A single substrate. n A group of similar substrates. n A particular type of bond.

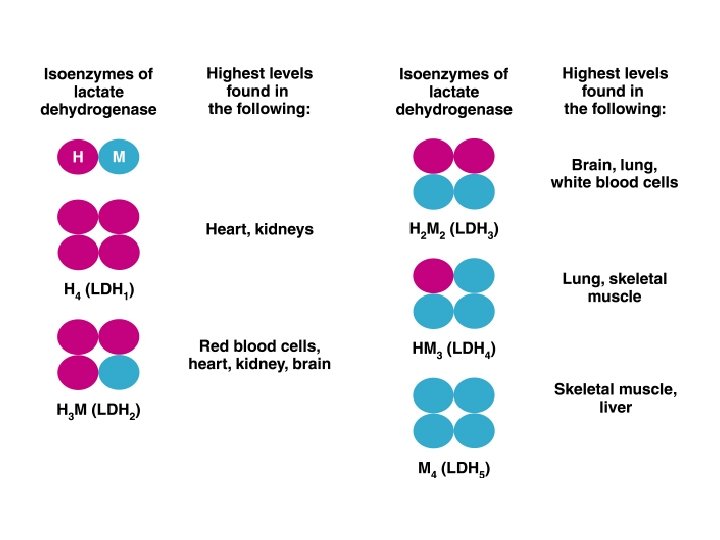
Isoenzymes n n Isoenzymes group of enzymes differs in amino acid sequence ( structure) but catalyze the same reaction in different tissues in the body. Lactate dehydrogenase, which converts lactate to pyruvate, (LDH) consists of five isoenzymes.
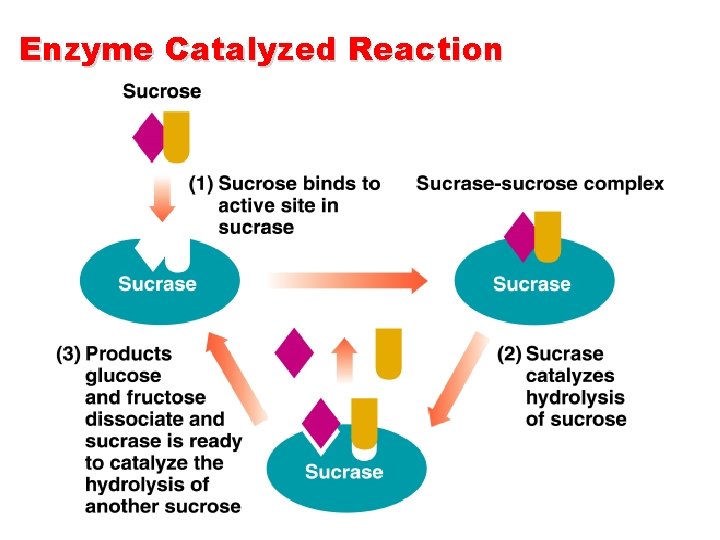
Enzyme Catalyzed Reaction

Enzyme Catalyzed Reaction n n The proper fit of a substrate (S) in an active site forms an enzyme-substrate (ES) complex. E+S ES Within the ES complex, the reaction occurs to convert substrate to product (P). ES E+P The products, which are no longer attracted to the active site, are released. Overall, substrate is convert to product. E+S ES E+P
![Enzyme Catalyzed Reaction n K 1 [E][S]=K-1[ES] +K 2 [ES] n Keq = K Enzyme Catalyzed Reaction n K 1 [E][S]=K-1[ES] +K 2 [ES] n Keq = K](http://slidetodoc.com/presentation_image_h2/3e275add6731972c49da3b50a4f3fcad/image-23.jpg)
Enzyme Catalyzed Reaction n K 1 [E][S]=K-1[ES] +K 2 [ES] n Keq = K 1 [E] [S]-K-1[ES] -K 2[ES]= 0 Michaelis-Menten equation V° = Vmax [S]/[S]+Km Michaelis-Menten Constant Km = K 2 + K-1 / K 1
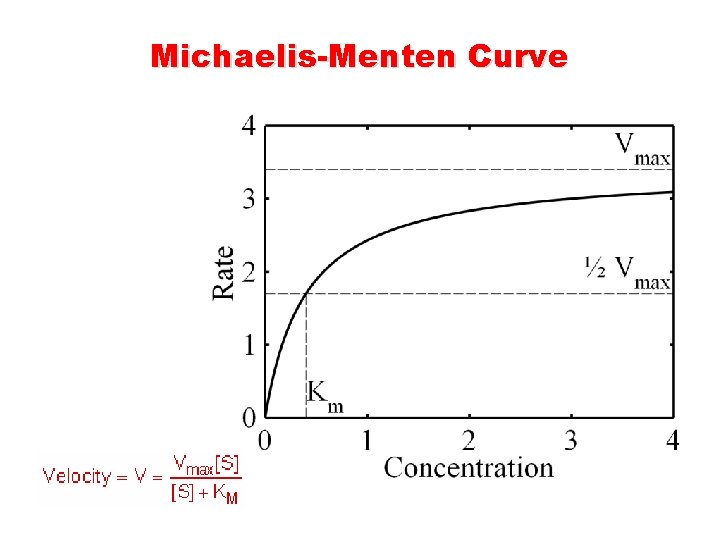
Michaelis-Menten Curve
![Substrate Saturation of an Enzyme A. Low [S] B. 50% [S] or Km C. Substrate Saturation of an Enzyme A. Low [S] B. 50% [S] or Km C.](http://slidetodoc.com/presentation_image_h2/3e275add6731972c49da3b50a4f3fcad/image-25.jpg)
Substrate Saturation of an Enzyme A. Low [S] B. 50% [S] or Km C. High, saturating [S]
- Slides: 25