Enzymes Chapter 6 What Are Enzymes Catalysts substances

Enzymes Chapter 6

What Are Enzymes? • Catalysts • substances that speed up a reaction • Enzymes – biological catalysts • Proteins • Reactions are too slow; need to speed up in order to maintain life

Reactions • For reactions to energy barrier must be overcome • Exergonic reactions • proceed only after the reactants are pushed over energy barrier • Ex: Propane + oxygen • Need to have a spark to start reaction • Pushed over energy barrier • Activation Energy (Ea) • The amount of energy needed to start the reaction
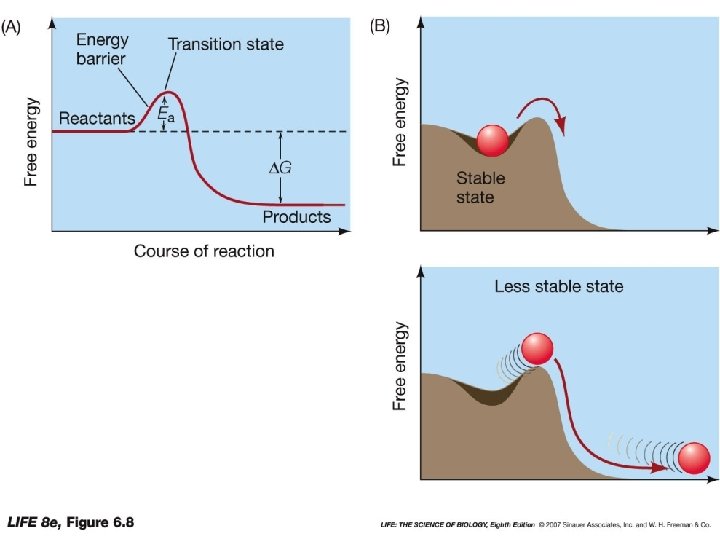

Activation Energy (Ea) • Activation Energy • Amount of energy needed to start the reaction • Transition-state species: • Energy needed to change reactants into unstable molecular forms • Chemical bonds stretch & become unstable • Activation energy can come from reactants kinetic energy from motion • Does not work in living systems • Enzymes • lower the energy barrier by bringing the reactants together.

Enzymes • ENZYMES lower the energy barrier • Bring the reactants into close proximity to each other • Biological catalysts are highly specific • Enzyme recognizes & binds to only one or a few closely related reactants • Names of enzymes represent their function & usually end in “-ase”

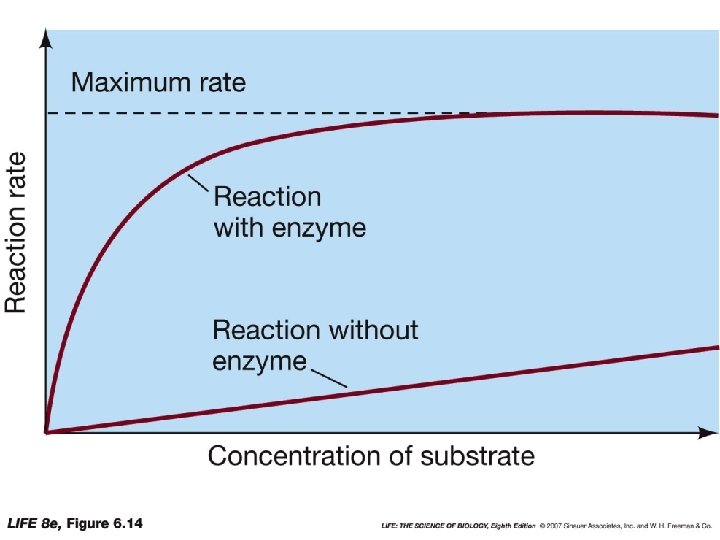
Enzymes • Substrates • Reactants in enzyme-catalyzed reaction • Active Site • Where catalysis takes place • Enzyme-Substrate Complex (ES) • The binding of a substrate to the active site • E+S ES E+P

How Enzymes Work • Chemical interaction occur in the ES complex • Break old bonds & form new ones • Enzymes use the following mechanisms Enzymes orient substances • Orient to ensure correct bonds are formed Enzymes induce strain in the substance • Stretches the bonds to create unstable transition state Enzyme temporarily add chemical groups to substrates
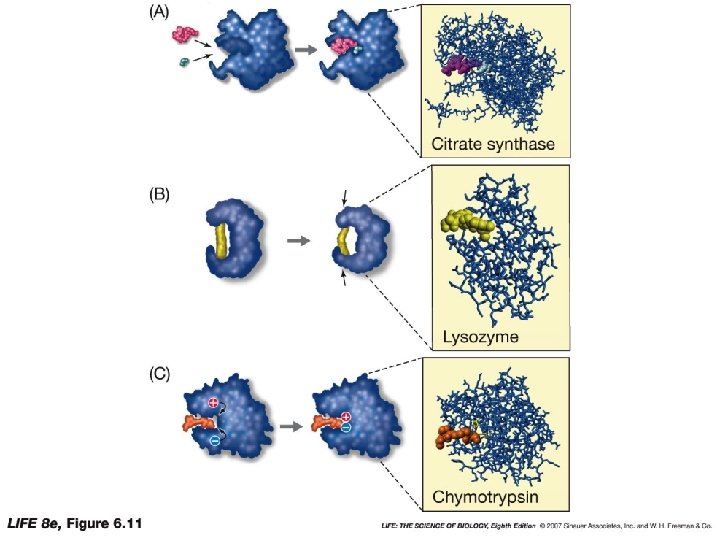

How enzymes work • Enzyme • Contain hundreds of amino acids usually with a single fold • Active site is quite small (6 -12 amino acids) • Helps select the right substrates • Enzyme changes shape when substrates bind: induced fit • Ensures the substrates are properly positioned

Enzymes • Some enzymes require “partners” to function • Prosthetic group • Non-amino acids atoms or molecules that are permanently bound • Cofactors • Inorganic ions that bind to enzymes (essential to function) • Copper, zinc, iron • Coenzymes • Required for certain actions of enzymes • Carbon-containing atom • Help add and/or remove chemical groups from substrates • ATP & ADP are necessary for reactions; considered coenzymes • Vitamins

Metabolic Pathway • Chemical reactions that occur one right after the other • Product of one is the reactant for the next • Pathways are interconnected; helps maintain homeostasis • Systems biology • Study of metabolic pathways

Inhibitors • Slow down the rate of enzymecatalyzed reactions • Natural inhibitors • Regulates metabolism • Artificial inhibitors • Treat diseases, kills pests

Inhibitors • Irreversible inhibitors • Covalently bonds • permanently inactivating the enzymes • Nerve gas • Reversible • Bonds non-covalently to active site • Prevents substrates from bonding • Competitive inhibitor • Binds elsewhere causing enzyme to change shape & alter active site • Noncompetitive inhibitor

Irreversible inhibitor
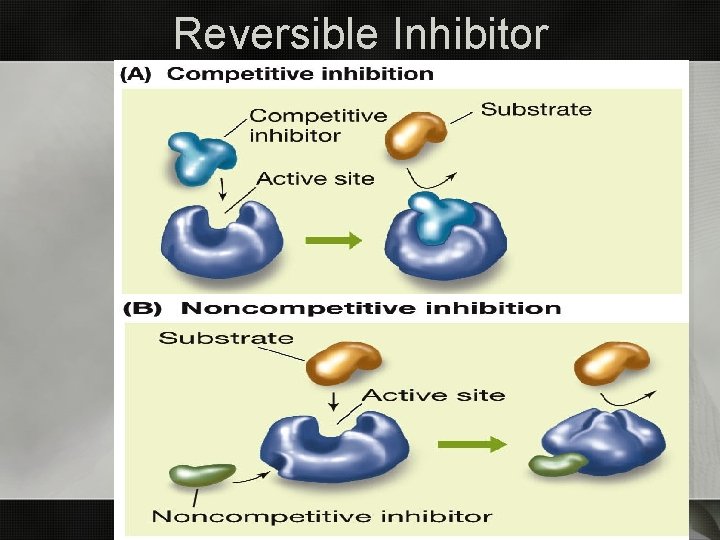
Reversible Inhibitor

Affects of enzyme activity • Environment • p. H • Each have an ideal p. H • Activity decreases the more acidic or basic a solution becomes • Temperature • High temperature non-covalent bonds break • Enzymes denature

• Isozymes: • enzymes that catalyze the same reaction but have different properties, such as optimal temperature. • Organisms can use isozymes to adjust to temperature changes. • Enzymes in humans have higher optimal temperature than enzymes in most bacteria—a fever can denature the bacterial enzymes.
- Slides: 20