Enzymes AS Revision Session 19 th February What

Enzymes AS Revision Session 19 th February
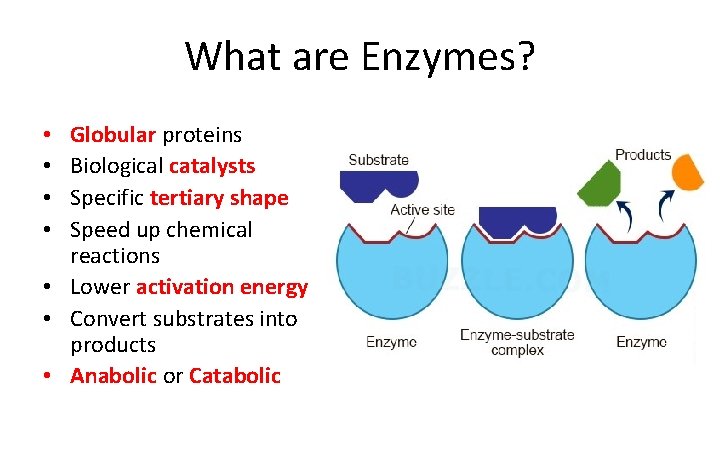
What are Enzymes? Globular proteins Biological catalysts Specific tertiary shape Speed up chemical reactions • Lower activation energy • Convert substrates into products • Anabolic or Catabolic • •
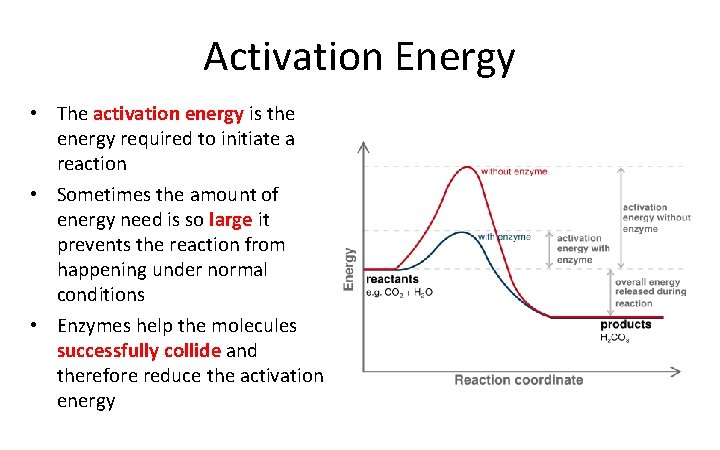
Activation Energy • The activation energy is the energy required to initiate a reaction • Sometimes the amount of energy need is so large it prevents the reaction from happening under normal conditions • Enzymes help the molecules successfully collide and therefore reduce the activation energy
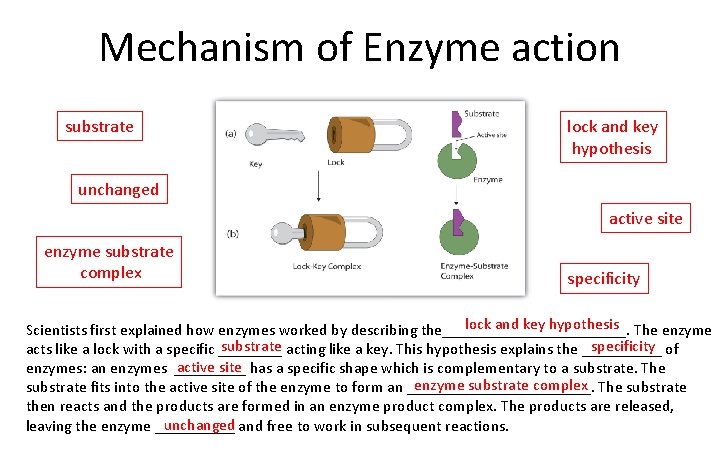
Mechanism of Enzyme action substrate lock and key hypothesis unchanged active site enzyme substrate complex specificity lock and key hypothesis The enzyme Scientists first explained how enzymes worked by describing the____________. substrate acting like a key. This hypothesis explains the _____ specificity of acts like a lock with a specific ____ active site has a specific shape which is complementary to a substrate. The enzymes: an enzymes _____ enzyme substrate complex The substrate fits into the active site of the enzyme to form an ____________. then reacts and the products are formed in an enzyme product complex. The products are released, unchanged and free to work in subsequent reactions. leaving the enzyme _____
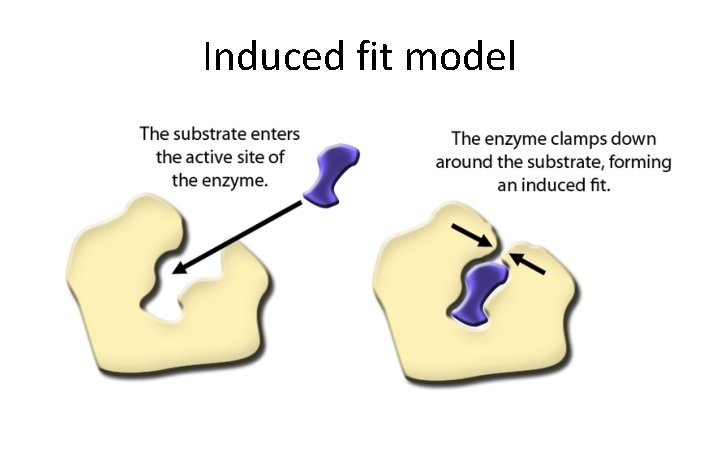
Induced fit model
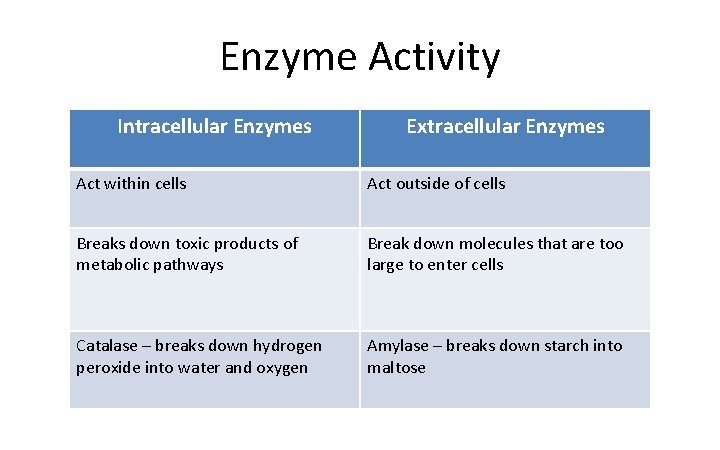
Enzyme Activity Intracellular Enzymes Extracellular Enzymes Act within cells Act outside of cells Breaks down toxic products of metabolic pathways Break down molecules that are too large to enter cells Catalase – breaks down hydrogen peroxide into water and oxygen Amylase – breaks down starch into maltose
Factors affecting enzyme activity 1. Temperature 1. p. H 1. Concentrations
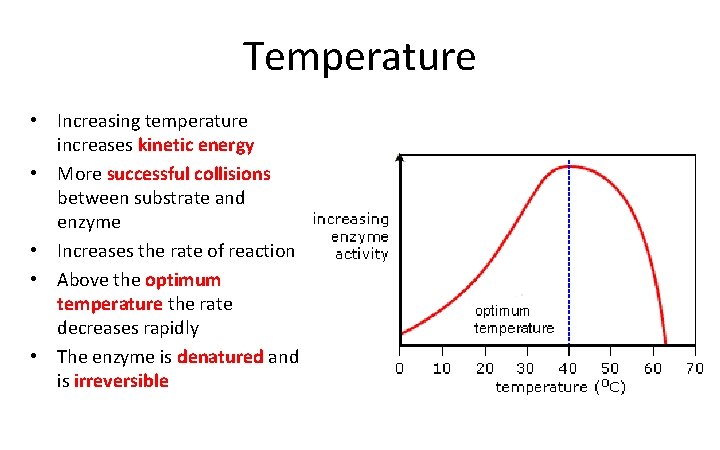
Temperature • Increasing temperature increases kinetic energy • More successful collisions between substrate and enzyme • Increases the rate of reaction • Above the optimum temperature the rate decreases rapidly • The enzyme is denatured and is irreversible
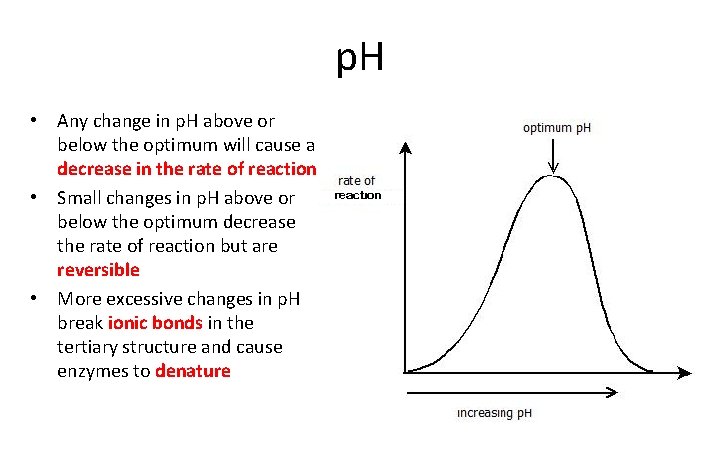
p. H • Any change in p. H above or below the optimum will cause a decrease in the rate of reaction • Small changes in p. H above or below the optimum decrease the rate of reaction but are reversible • More excessive changes in p. H break ionic bonds in the tertiary structure and cause enzymes to denature

Substrate Concentration • Increasing substrate concentration increases the rate of reaction. • This is because more substrate molecules will be colliding with enzyme molecule so more enzyme substrate complexes are formed • Plateau reached when all enzyme active sites are occupied – the enzymes are saturated • Enzyme becomes the limiting factor

Enzyme Concentration • Increasing enzyme concentration will increase the rate of reaction • More available active sites • More enzyme substrate complexes are formed • However this only continues to rise up until the Vmax • No substrate left • No more enzyme substrate complexes can be formed, the substrate concentration is the limiting factor
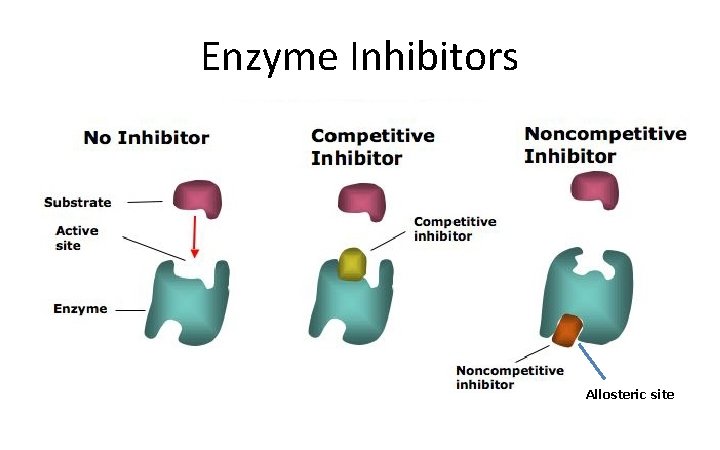
Enzyme Inhibitors Allosteric site

Cofactors, coenzymes and prosthetic groups • Non protein “helpers” • They transfer atoms or groups or form part of the active site • Cofactors are inorganic molecules derived from minerals e. g Cl- in amylase • Coenzymes are organic molecule derived from vitamins • Where cofactors are more tightly bound to the enzyme (permenant feature), they are referred to as a prosthetic group e. g Zn 2+ for carbonic anhydrase


ANABOLIC

PROTEASE

ACTIVATION ENERGY

DENATURE

COFACTOR

LOCK AND KEY HYPOTHESIS

ACTIVE SITE

COMPETITIVE INHIBITOR

AMYLASE

ENZYME SUBSTRATE COMPLEX

Post MCQ and Questionnaire
- Slides: 25