Enzymes as catalysts Rate can be increased up





Enzymes as catalysts Rate can be increased up to 107 -fold. Relatively mild conditions. Highly specific. Activity can be regulated.










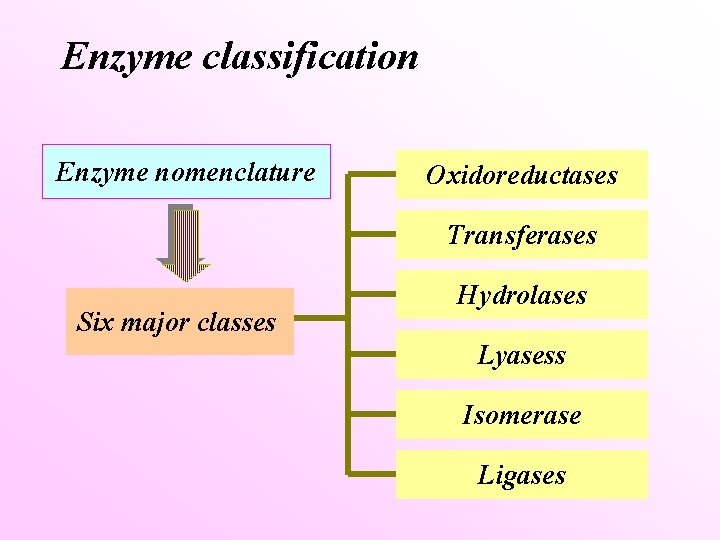
Enzyme classification Enzyme nomenclature Oxidoreductases Transferases Six major classes Hydrolases Lyasess Isomerase Ligases












Coenzymes and prosthetic groups Apoenzyme Holoenzyme Cofactors Coenzyme Prosthetic group Inorganic ions Complex organic molecule Vitamin Nicotinamide adenine dinucleotide Flavin mononucleotide Nicotinamide adenine dinucleotide phosphate Flavin adenine dinucleotide
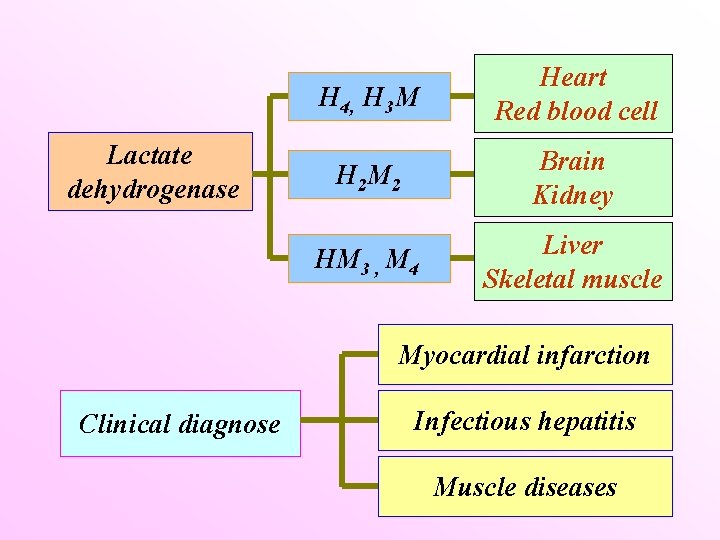
Lactate dehydrogenase H 4, H 3 M Heart Red blood cell H 2 M 2 Brain Kidney HM 3 , M 4 Liver Skeletal muscle Myocardial infarction Clinical diagnose Infectious hepatitis Muscle diseases






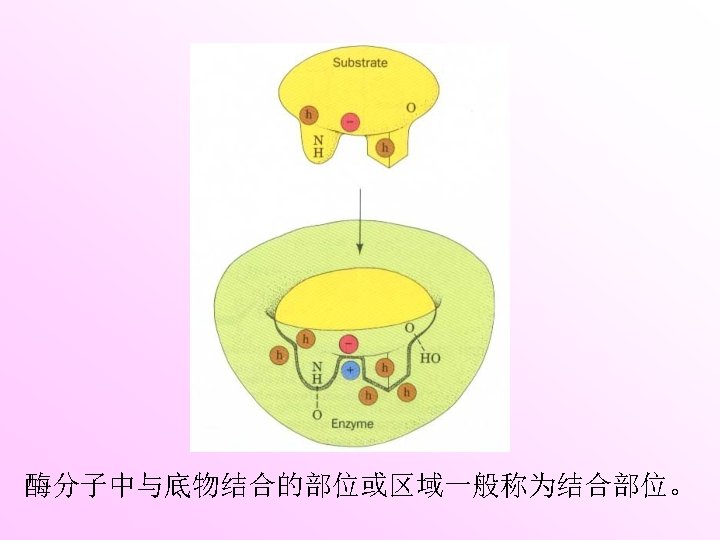



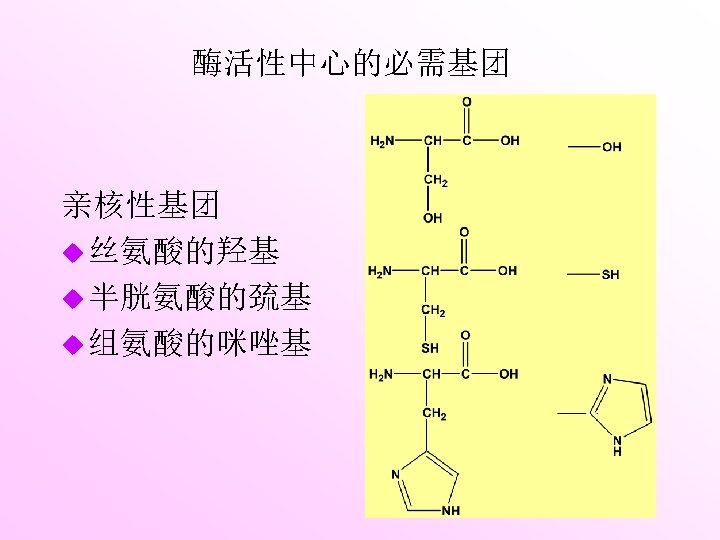

Three-dimen -sional entity Active site is the region that binds the substrate and converts it into product. Electrostatic interactions Multiple weak forces Hydrogen bonds Van der Waals bonds Covalent bonds Hydrophobic interactions
Three-dimen -sional entity Active site is the region that binds the substrate and converts it into product. Electrostatic interactions Multiple weak forces Hydrogen bonds Van der Waals bonds Covalent bonds Hydrophobic interactions
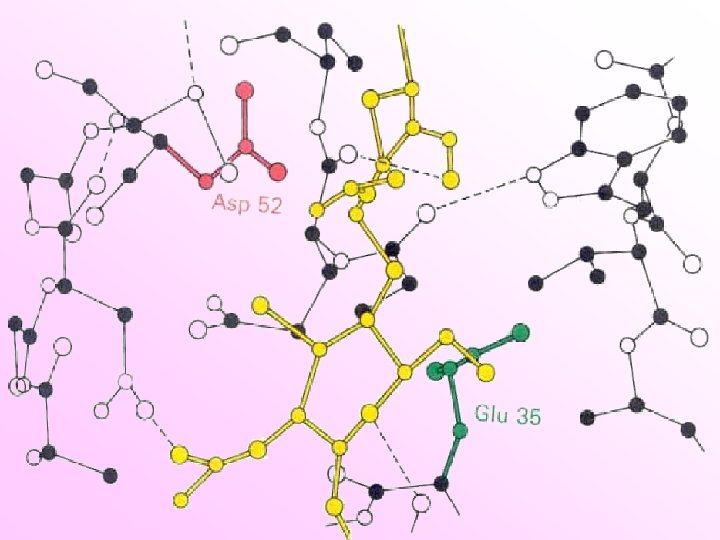



Isoenzymes Catalyze the same reaction p. I Different physical or kinetic properties p. H optimum Substrate affinity Effect of inhibitors




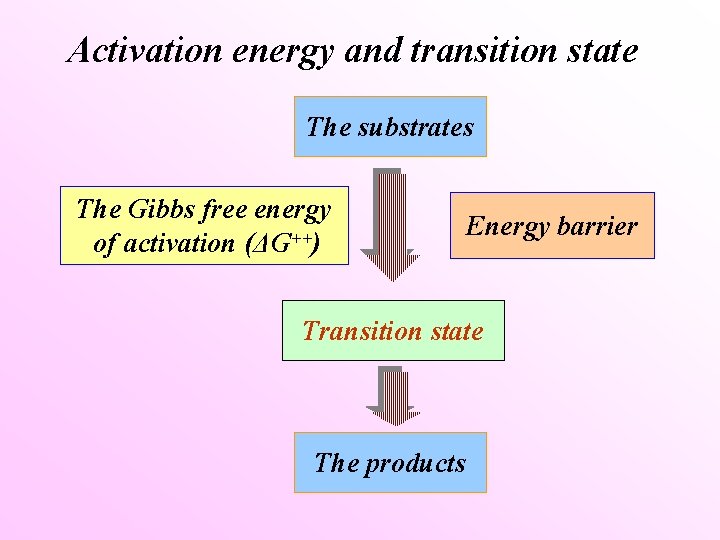
Activation energy and transition state The substrates The Gibbs free energy of activation (ΔG++) Energy barrier Transition state The products
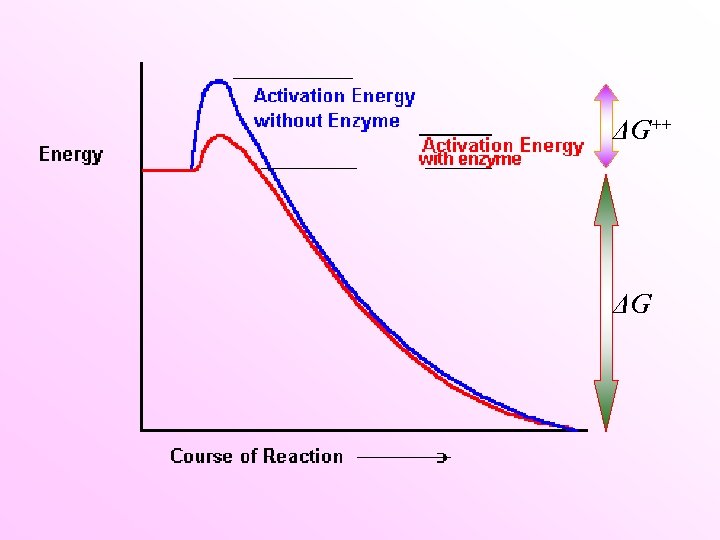

Free energy chance The change in Gibbs free energy(ΔG; k. J mol-1) dictates whether a reaction will be energetically favorable or not. Negative ΔG is thermodynamically favorable Positive ΔG is not thermodynamically favorable Concentrations of 1. 0 M ΔGo' Constants p. H of 7. 0

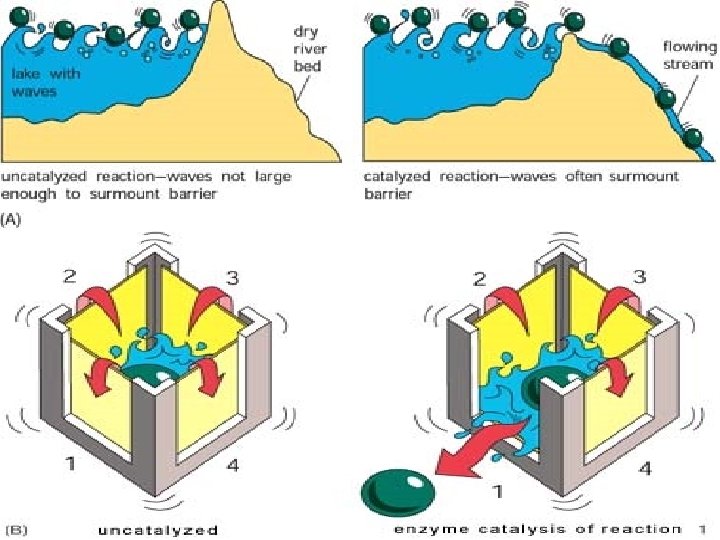
Chemical equilibria Dynamic equilibrium Equilibrium constant (K) Enzyme accelerate the attainment of the equilibrium position.
- Slides: 57