Enzyme Regulation Andy Howard Introductory Biochemistry 19 November

Enzyme Regulation Andy Howard Introductory Biochemistry 19 November 2014 Mechanisms & Regulation 11/19/2014

Enzyme Regulation happens at several levels n Even though it isn’t really an enzyme, hemoglobin can teach us how allostery in enzymes works. n But first, we’ll finish our discussion of mechanisms n 11/19/2014 Mechanisms & Regulation P. 2 of 40

Mechanisms & Regulation n Mechanisms Globins n Cysteinyl proteases n n Lysozyme TIM n n n Regulation n n n Thermodynamics Availability Allostery PTM Protein-protein interactions 11/19/2014 n n n Oxygen binding Structure R and T states Allostery Bohr effect BPG as an effector Sickle-cell anemia Mechanisms & Regulation P. 3 of 40
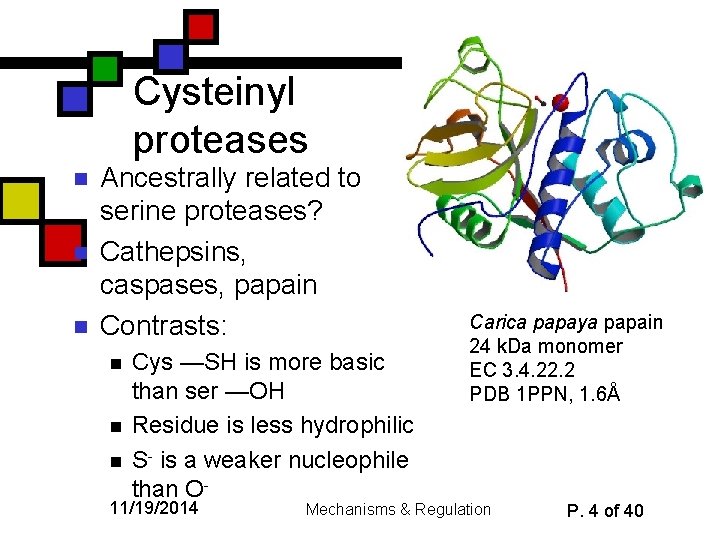
Cysteinyl proteases n n n Ancestrally related to serine proteases? Cathepsins, caspases, papain Contrasts: n n n Cys —SH is more basic than ser —OH Residue is less hydrophilic S- is a weaker nucleophile than O- 11/19/2014 Carica papaya papain 24 k. Da monomer EC 3. 4. 22. 2 PDB 1 PPN, 1. 6Å Mechanisms & Regulation P. 4 of 40
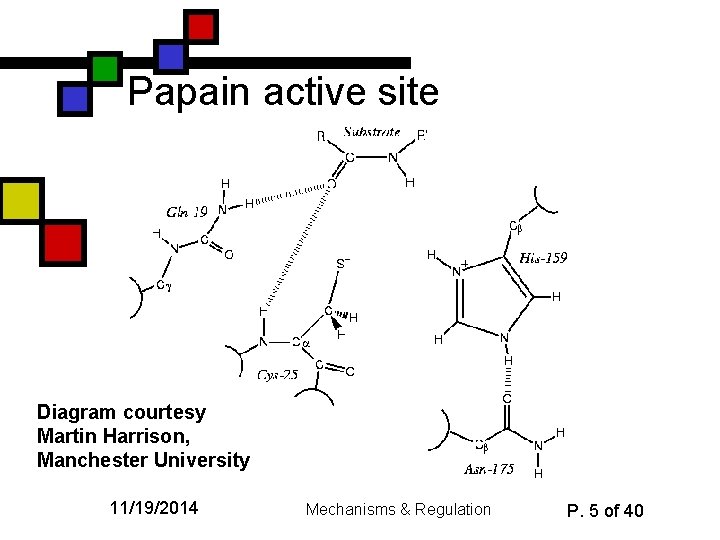
Papain active site Diagram courtesy Martin Harrison, Manchester University 11/19/2014 Mechanisms & Regulation P. 5 of 40
Hen egg-white lysozyme n n n Antibacterial protectant of growing chick embryo Hydrolyzes bacterial cell-wall peptidoglycans “hydrogen atom of structural biology” n n Commercially available in pure form Easy to crystallize and do structure work Available in multiple crystal forms HEWL PDB 2 vb 1 0. 65Å 15 k. Da Mechanism is surprisingly complex 11/19/2014 Mechanisms & Regulation P. 6 of 40

Mechanism of lysozyme n n n Strain-induced destabilization of substrate makes the substrate look more like the transition state Long arguments about the nature of the intermediates Accepted answer: covalent intermediate between D 52 and glycosyl C 1 11/19/2014 Mechanisms & Regulation P. 7 of 40
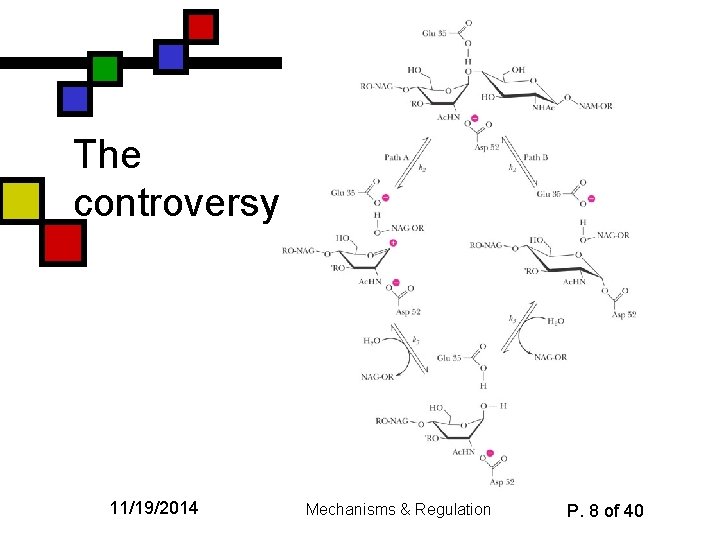
The controversy 11/19/2014 Mechanisms & Regulation P. 8 of 40
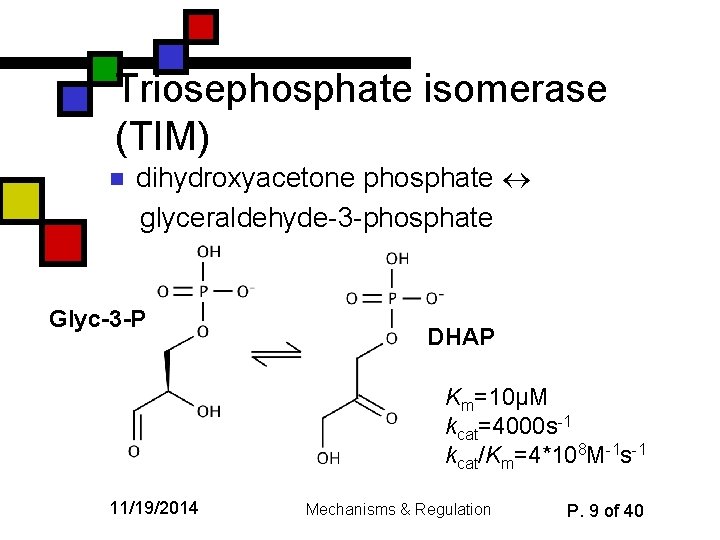
Triosephosphate isomerase (TIM) n dihydroxyacetone phosphate glyceraldehyde-3 -phosphate Glyc-3 -P DHAP Km=10µM kcat=4000 s-1 kcat/Km=4*108 M-1 s-1 11/19/2014 Mechanisms & Regulation P. 9 of 40
TIM mechanism n n n DHAP carbonyl H-bonds to neutral imidazole of his-95; proton moves from C 1 to carboxylate of glu 165 Enediolate intermediate (C—O- on C 2) Imidazolate (negative!) form of his 95 interacts with C 1—O-H) glu 165 donates proton back to C 2 See Fort’s treatment (http: //chemistry. umeche. maine. edu/ CHY 431/Enzyme 3. html) 11/19/2014 Mechanisms & Regulation P. 10 of 40

Enzymes are under several levels of control Some controls operate at the level of enzyme availability n Other controls are exerted by thermodynamics, inhibition, or allostery n 11/19/2014 Mechanisms & Regulation P. 11 of 40

Regulation of enzymes n n The very catalytic proficiency for which enzymes have evolved means that their activity must not be allowed to run amok Activity is regulated in many ways: n n n Thermodynamics Enzyme availability Allostery Post-translational modification Protein-protein interactions 11/19/2014 Mechanisms & Regulation P. 12 of 40

Thermodynamics as a regulatory force n n n Remember that Go’ is not the determiner of spontaneity: G is. Therefore: local product and substrate concentrations determine whether the enzyme is catalyzing reversible reactions to the left or to the right Rule of thumb: Go’ < -20 k. J mol-1 is irreversible 11/19/2014 Mechanisms & Regulation P. 13 of 40
![Why -20 k. J/mol? n n n ΔG = ΔGo’+ RT ln[Products]/[Reactants] We ask: Why -20 k. J/mol? n n n ΔG = ΔGo’+ RT ln[Products]/[Reactants] We ask:](http://slidetodoc.com/presentation_image/689240f07d8e7620a51b8d5e34a0b8e6/image-14.jpg)
Why -20 k. J/mol? n n n ΔG = ΔGo’+ RT ln[Products]/[Reactants] We ask: if ΔGo’ = -20 k. J/mol, what ratio of concentrations will make ΔG = 0? That’s easy to answer: 0 = -20 + RTln[products]/[reactants] For T=300. 66 K, RT=2. 5 k. J/mol, that’s 20 = 2. 5 ln[products]/[reactants] = 8, [products]/[reactants]=e 8 = 2981… How likely is that? 11/19/2014 Mechanisms & Regulation P. 14 of 40

Enzyme availability n n n The enzyme has to be where the reactants are in order for it to act Even a highly proficient enzyme has to have a nonzero concentration How can the cell control [E]tot? n n n Transcription (and translation) Protein processing (degradation) Compartmentalization 11/19/2014 Mechanisms & Regulation P. 15 of 40
Transcriptional control n n n m. RNAs have short lifetimes Therefore once a protein is degraded, it will be replaced and available only if new transcriptional activity for that protein occurs Many types of transcriptional effectors n n n Proteins can bind to their own gene Small molecules can bind to gene Promoters can be turned on or off 11/19/2014 Mechanisms & Regulation P. 16 of 40
Protein degradation n n All proteins have finite half-lives; Enzymes’ lifetimes often shorter than structural or transport proteins Degraded by slings & arrows of outrageous fortune; or Activity of the proteasome, a molecular machine that tags proteins for degradation and then accomplishes it 11/19/2014 Mechanisms & Regulation P. 17 of 40

Compartmentalization n If the enzyme is in one compartment and the substrate in another, it won’t catalyze anything Several mitochondrial catabolic enzyme act on substrates produced in the cytoplasm; these require elaborate transport mechanisms to move them in Therefore, control of the transporters confers control over the enzymatic system 11/19/2014 Mechanisms & Regulation P. 18 of 40

Allostery n n Remember we defined this as an effect on protein activity in which binding of a ligand to a protein induces a conformational change that modifies the protein’s activity Ligand may be the same molecule as the substrate or it may be a different one Ligand may bind to the same subunit or a different one These effects happen to non-enzymatic proteins as well as enzymes 11/19/2014 Mechanisms & Regulation P. 19 of 40

Substrates as allosteric effectors (homotropic) n n n Standard example: binding of O 2 to one subunit of tetrameric hemoglobin induces conformational change that facilitates binding of 2 nd (& 3 rd & 4 th) O 2’s So the first oxygen is an allosteric effector of the activity in the other subunits Effect can be inhibitory or accelerative 11/19/2014 Mechanisms & Regulation P. 20 of 40

Other allosteric effectors (heterotropic) n n n Covalent modification of an enzyme by phosphate or other PTM molecules can turn it on or off Usually catabolic enzymes are stimulated by phosphorylation and anabolic enzymes are turned off, but not always Phosphatases catalyze dephosphorylation; these have the opposite effects 11/19/2014 Mechanisms & Regulation P. 21 of 40
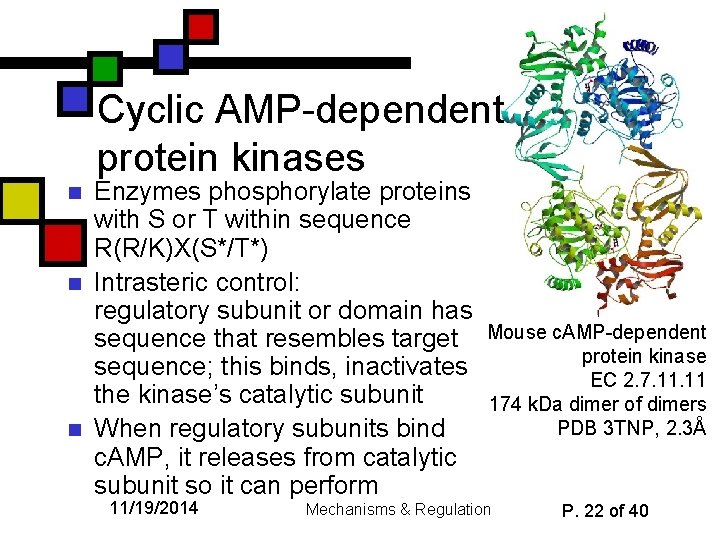
Cyclic AMP-dependent protein kinases n n n Enzymes phosphorylate proteins with S or T within sequence R(R/K)X(S*/T*) Intrasteric control: regulatory subunit or domain has sequence that resembles target sequence; this binds, inactivates the kinase’s catalytic subunit When regulatory subunits bind c. AMP, it releases from catalytic subunit so it can perform 11/19/2014 Mouse c. AMP-dependent protein kinase EC 2. 7. 11 174 k. Da dimer of dimers PDB 3 TNP, 2. 3Å Mechanisms & Regulation P. 22 of 40
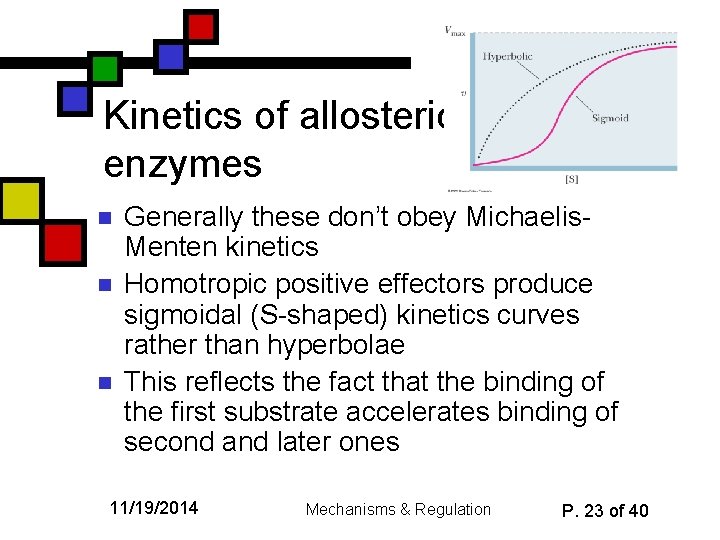
Kinetics of allosteric enzymes n n n Generally these don’t obey Michaelis. Menten kinetics Homotropic positive effectors produce sigmoidal (S-shaped) kinetics curves rather than hyperbolae This reflects the fact that the binding of the first substrate accelerates binding of second and later ones 11/19/2014 Mechanisms & Regulation P. 23 of 40

T R State transitions n n Many allosteric effectors influence the equilibrium between two conformations One is typically more rigid and inactive, the other is more flexible and active The rigid one is typically called the “tight” or “T” state; the flexible one is called the “relaxed” or “R” state Allosteric effectors shift the equilibrium toward R or toward T 11/19/2014 Mechanisms & Regulation P. 24 of 40
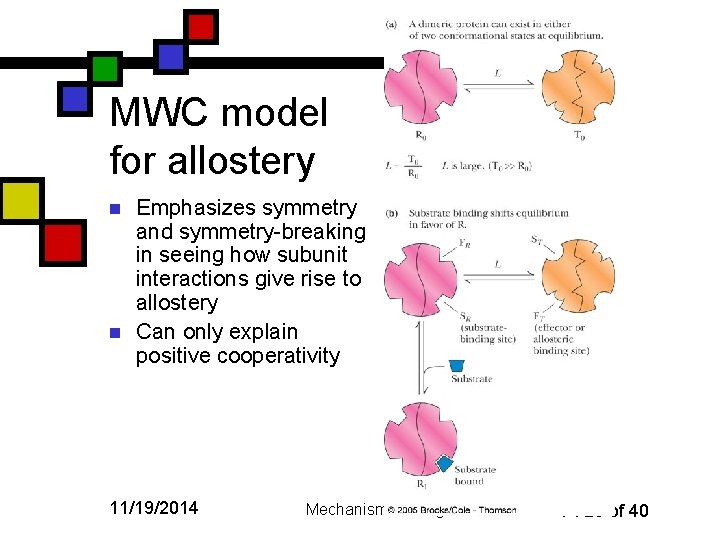
MWC model for allostery n n Emphasizes symmetry and symmetry-breaking in seeing how subunit interactions give rise to allostery Can only explain positive cooperativity 11/19/2014 Mechanisms & Regulation P. 25 of 40

Koshland (KNF) model n n Emphasizes conformational changes from one state to another, induced by binding of effector Ligand binding and conformational transitions are distinct steps … so this is a sequential model for allosteric transitions Allows for negative cooperativity as well as positive cooperativity 11/19/2014 Mechanisms & Regulation P. 26 of 40
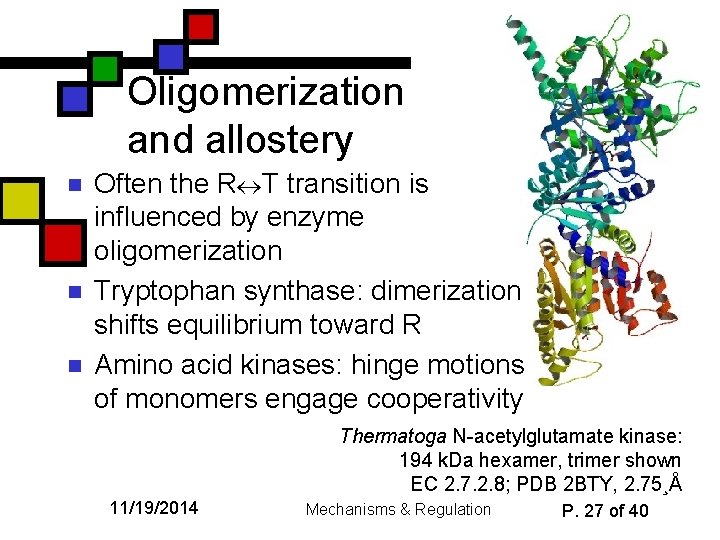
Oligomerization and allostery n n n Often the R T transition is influenced by enzyme oligomerization Tryptophan synthase: dimerization shifts equilibrium toward R Amino acid kinases: hinge motions of monomers engage cooperativity Thermatoga N-acetylglutamate kinase: 194 k. Da hexamer, trimer shown EC 2. 7. 2. 8; PDB 2 BTY, 2. 75¸Å 11/19/2014 Mechanisms & Regulation P. 27 of 40
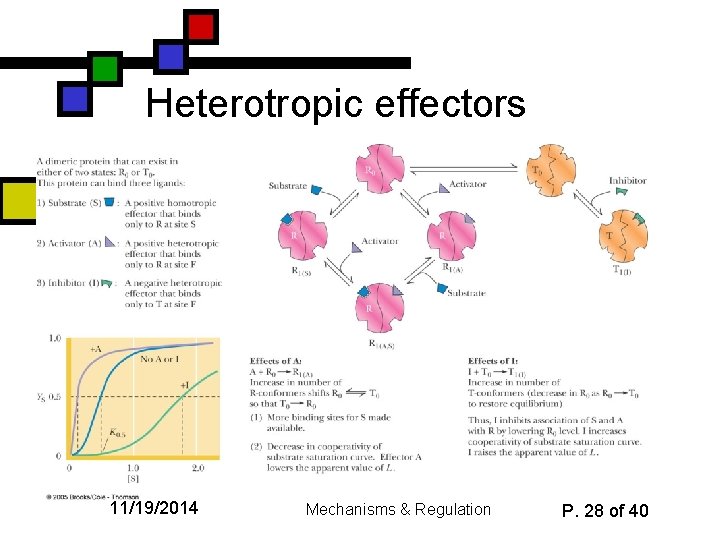
Heterotropic effectors 11/19/2014 Mechanisms & Regulation P. 28 of 40

Post-translational modification n n We’ve already looked at phosphorylation Proteolytic cleavage of the enzyme to activate it is another common PTM mode Some proteases cleave themselves (auto -catalysis); in other cases there’s an external protease involved Blood-clotting cascade involves a series of catalytic activations 11/19/2014 Mechanisms & Regulation P. 29 of 40

Zymogens n n n As mentioned earlier, this is a term for an inactive form of a protein produced at the ribosome Proteolytic post-translational processing required for the zymogen to be converted to its active form Cleavage may happen intracellularly, during secretion, or extracellularly 11/19/2014 Mechanisms & Regulation Bacillus subtilisin + prosequence 35 k. Da heterodimer EC 3. 4. 21. 62 PDB 3 CNQ, 1. 71Å P. 30 of 40
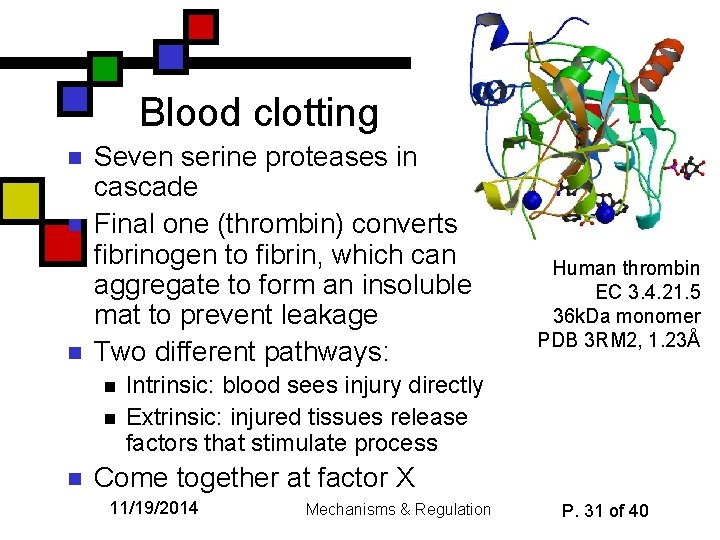
Blood clotting n n n Seven serine proteases in cascade Final one (thrombin) converts fibrinogen to fibrin, which can aggregate to form an insoluble mat to prevent leakage Two different pathways: n n n Human thrombin EC 3. 4. 21. 5 36 k. Da monomer PDB 3 RM 2, 1. 23Å Intrinsic: blood sees injury directly Extrinsic: injured tissues release factors that stimulate process Come together at factor X 11/19/2014 Mechanisms & Regulation P. 31 of 40
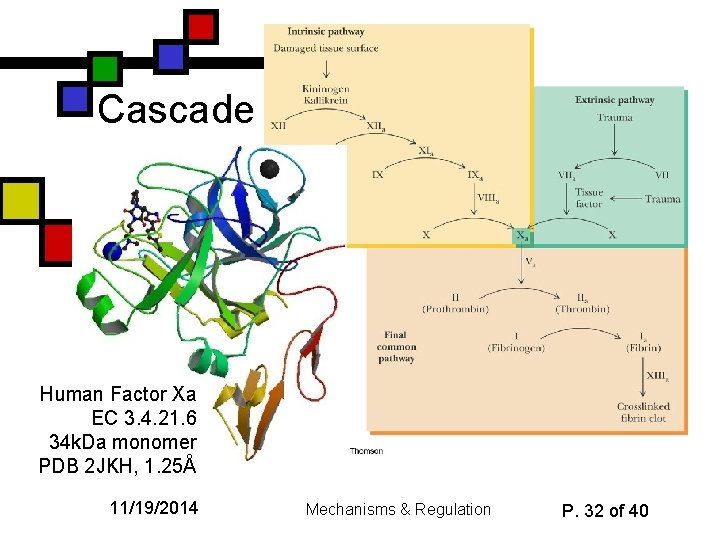
Cascade Human Factor Xa EC 3. 4. 21. 6 34 k. Da monomer PDB 2 JKH, 1. 25Å 11/19/2014 Mechanisms & Regulation P. 32 of 40

Protein-protein interactions n n n One major change in biochemistry in the last 25 years is the increasing emphasis on protein-protein interactions in understanding biological activities Many proteins depend on exogenous partners for modulating their activity up or down Example: cholera toxin’s enzymatic component depends on interaction with human protein ARF 6 Vibrio cholerae toxin A 1 subunit + 11/19/2014 human ARF 6: 37 k. Da heterodimer PDB 2 A 5 D, 1. 8Å Mechanisms & Regulation P. 33 of 40

Enzyme availability n n n The enzyme has to be where the reactants are in order for it to act Even a highly proficient enzyme has to have a nonzero concentration How can the cell control [E]tot? n n n Transcription (and translation) Protein processing (degradation) Compartmentalization 11/19/2014 Mechanisms & Regulation P. 34 of 40

Globins as aids to understanding n n Myoglobin and hemoglobin are wellunderstood non-enzymatic proteins whose properties help us understand enzyme regulation Hemoglobin is described as an “honorary enzyme” in that it “catalyzes” the reaction O 2(lung) O 2 (peripheral tissues) 11/19/2014 Mechanisms & Regulation P. 35 of 40
Setting the stage for this story n n n Myoglobin is a 16 k. Da monomeric O 2 -storage protein found in peripheral tissues Has Fe-containing prosthetic group called heme; iron must be in Fe 2+ state to bind O 2 It yields up dioxygen to various oxygenrequiring processes, particularly oxidative phosphorylation in mitochondria in rapidly metabolizing tissues 11/19/2014 Mechanisms & Regulation P. 36 of 40

Why is myoglobin needed? n Free heme will bind O 2 nicely; why not just rely on that? n Protein has 3 functions: Immobilizes the heme group n Discourages oxidation of Fe 2+ to Fe 3+ n Provides a pocket that oxygen can fit into n 11/19/2014 Mechanisms & Regulation P. 37 of 40

Setting the stage II n n n Hemoglobin (in vertebrates, at least) is a tetrameric, 64 k. Da transport protein that carries oxygen from the lungs to peripheral tissues It also transports acidic CO 2 the opposite direction Its allosteric properties are what we’ll discuss 11/19/2014 Mechanisms & Regulation P. 38 of 40

n Structure determinations Myoglobin & hemoglobin were the first 2 proteins to have their 3 -D structures determined experimentally n n Photo courtesy EMBL Myoglobin: Kendrew, 1958 Hemoglobin: Perutz, 1958 Most of the experimental tools that crystallographers rely on were developed for these structure determinations Nobel prizes for both, 1965 (small T!) 11/19/2014 Mechanisms & Regulation Photo courtesy Oregon State Library P. 39 of 40
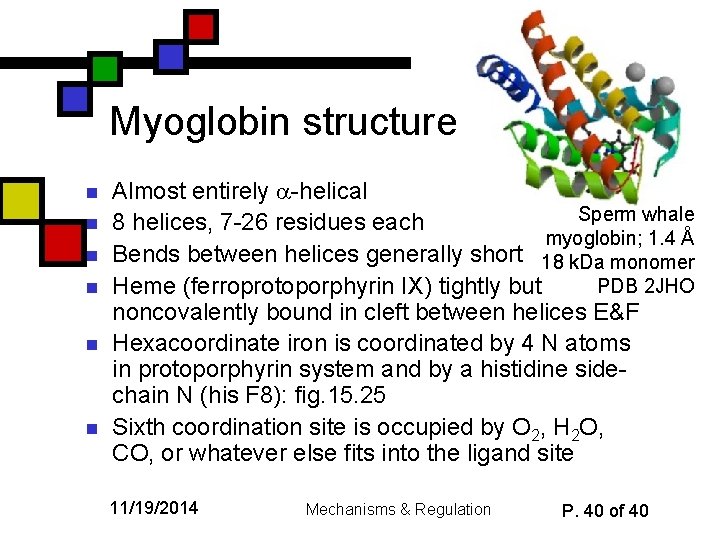
Myoglobin structure n n n Almost entirely -helical Sperm whale 8 helices, 7 -26 residues each myoglobin; 1. 4 Å Bends between helices generally short 18 k. Da monomer PDB 2 JHO Heme (ferroprotoporphyrin IX) tightly but noncovalently bound in cleft between helices E&F Hexacoordinate iron is coordinated by 4 N atoms in protoporphyrin system and by a histidine sidechain N (his F 8): fig. 15. 25 Sixth coordination site is occupied by O 2, H 2 O, CO, or whatever else fits into the ligand site 11/19/2014 Mechanisms & Regulation P. 40 of 40
- Slides: 40