Enzyme Kinetics Score Student B Student C 0
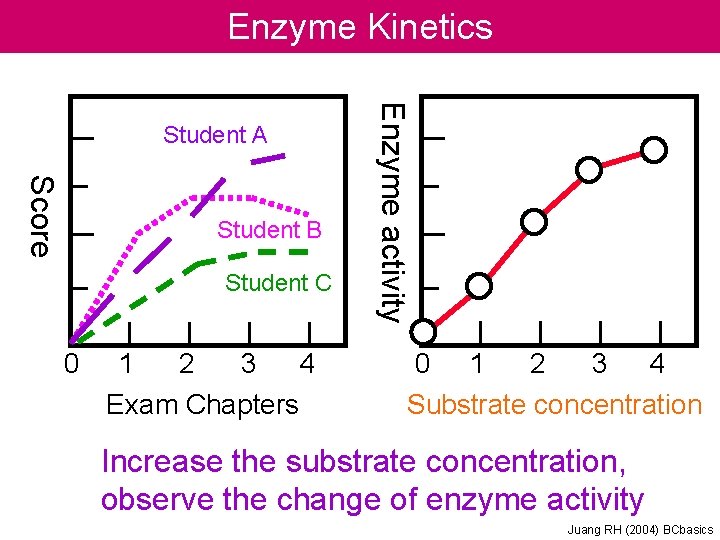
Enzyme Kinetics Score Student B Student C 0 1 2 3 4 Exam Chapters Enzyme activity Student A 0 1 2 3 4 Substrate concentration Increase the substrate concentration, observe the change of enzyme activity Juang RH (2004) BCbasics
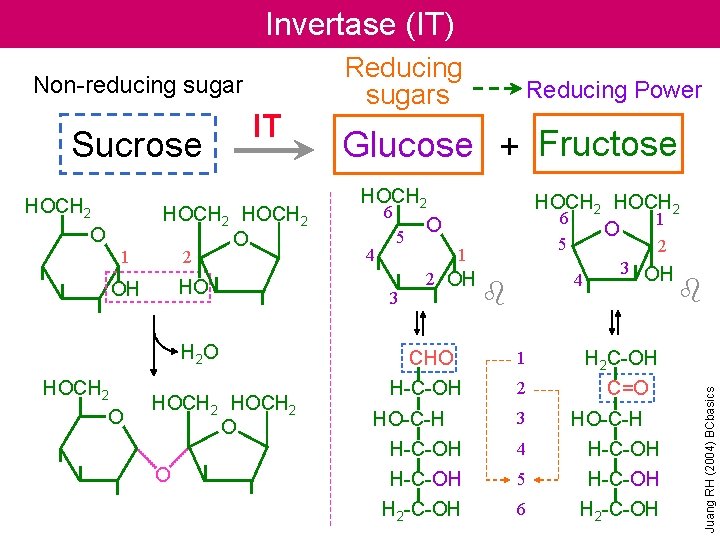
Invertase (IT) Sucrose HOCH 2 O 1 IT HOCH 2 O 2 HO OH H 2 O HOCH 2 O O Reducing Power Glucose + Fructose HOCH 2 6 O 4 5 3 HOCH 2 6 1 O 5 1 2 OH CHO H-C-OH HO-C-H 2 4 b 1 2 3 3 OH b H 2 C-OH C=O HO-C-H H-C-OH 4 H-C-OH 5 H-C-OH H 2 -C-OH 6 H 2 -C-OH Juang RH (2004) BCbasics Non-reducing sugar Reducing sugars
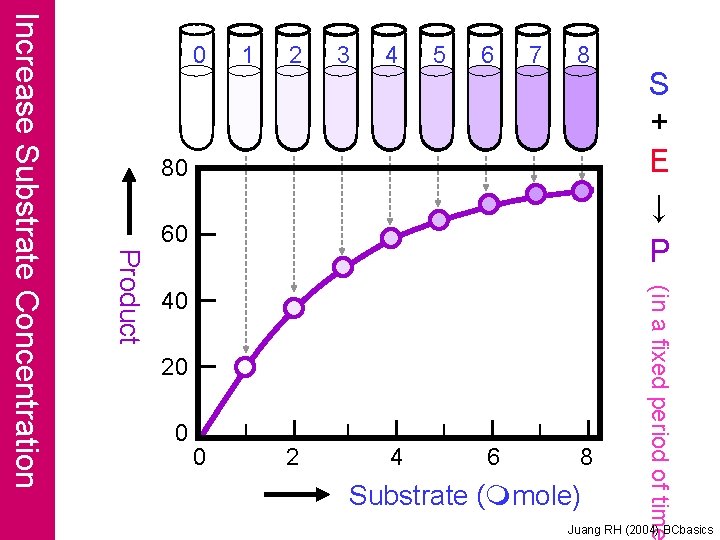
1 2 3 4 5 6 7 8 80 60 40 20 0 0 2 4 6 8 Substrate (mmole) S + E ↓ P (in a fixed period of time Product Increase Substrate Concentration 0 Juang RH (2004) BCbasics
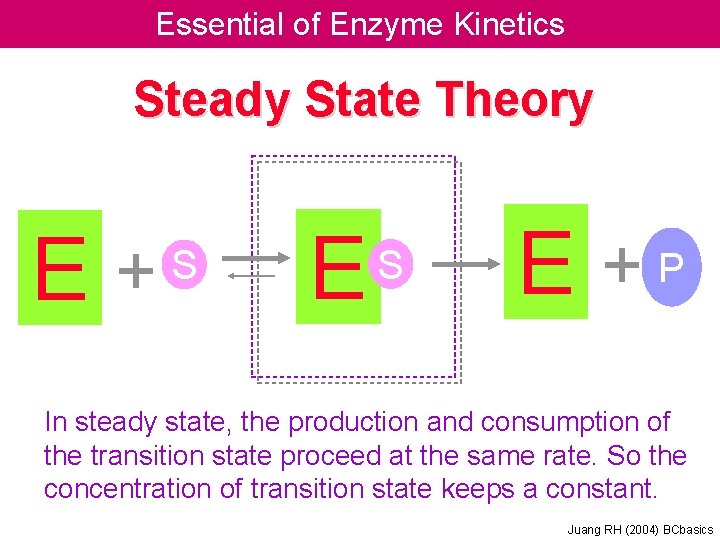
Essential of Enzyme Kinetics Steady State Theory E + S E +P In steady state, the production and consumption of the transition state proceed at the same rate. So the concentration of transition state keeps a constant. Juang RH (2004) BCbasics
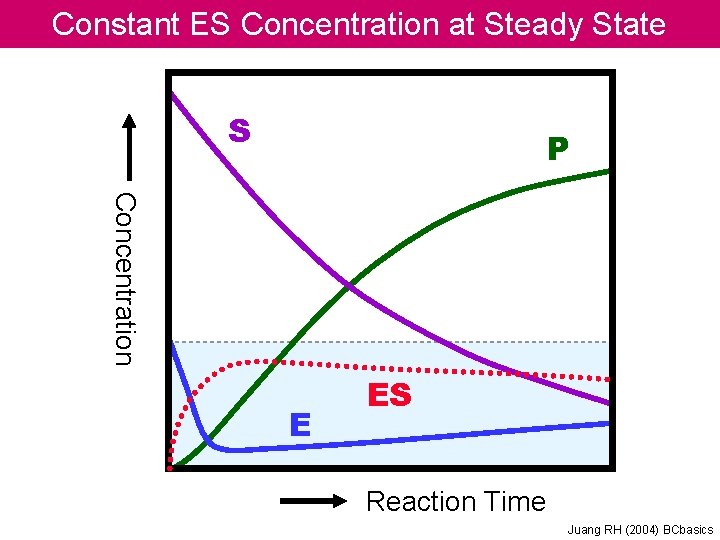
Constant ES Concentration at Steady State S P Concentration E ES Reaction Time Juang RH (2004) BCbasics
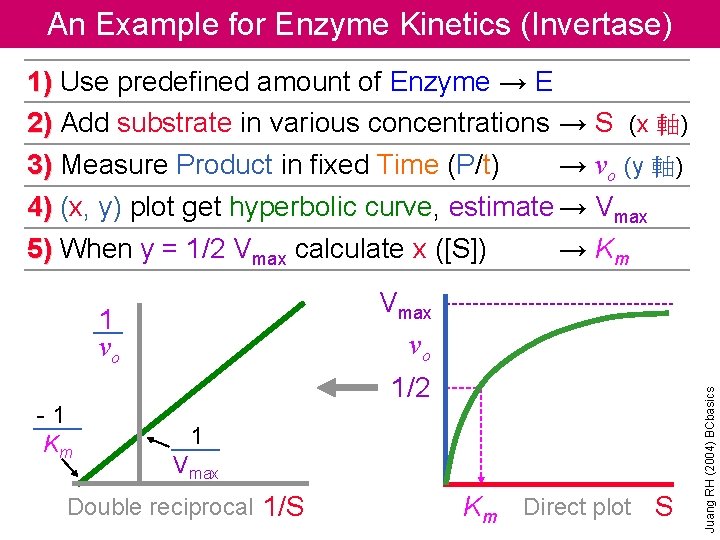
An Example for Enzyme Kinetics (Invertase) 1) Use predefined amount of Enzyme → E 2) Add substrate in various concentrations → S (x 軸) 3) Measure Product in fixed Time (P/t) → vo (y 軸) 4) (x, y) plot get hyperbolic curve, estimate → Vmax 5) When y = 1/2 Vmax calculate x ([S]) → Km 1 vo -1 Km 1/2 1 Vmax Double reciprocal 1/S Km Direct plot S Juang RH (2004) BCbasics Vmax vo
![A Real Example for Enzyme Kinetics Data Substrate Product Velocity Double reciprocal [S] Absorbancev A Real Example for Enzyme Kinetics Data Substrate Product Velocity Double reciprocal [S] Absorbancev](http://slidetodoc.com/presentation_image/2deb61d5626ff1c25142850a7c5f26f1/image-7.jpg)
A Real Example for Enzyme Kinetics Data Substrate Product Velocity Double reciprocal [S] Absorbancev (mmole/min) 0. 25 0. 21 → 0. 42 0. 50 0. 36 → 0. 72 1. 0 0. 40 → 0. 80 2. 0 0. 46 → 0. 92 no 1 2 3 4 1/S 4 2 1 0. 5 1/v 2. 08 1. 56 1. 35 1. 16 1. 0 v 0. 5 0 0 1 2 [S] 2. 0 1/v 1. 0 0 -4 -3. 8 -2 0 1/[S] 2 4 Juang RH (2004) BCbasics Double reciprocal Direct plot (1) The product was measured by spectroscopy at 600 nm for 0. 05 per mmole (2) Reaction time was 10 min
- Slides: 7