Enzyme and Cell Immobilization and its Applications Course

Enzyme and Cell Immobilization and its Applications Course title: Industrial Production of Secondary Metabolites By Professor Dr. Zeb Saddiqe Department of Botany LCWU

Introduction • Enzymes are the biological catalysts that promote chemical reactions in living organisms • They have the ability to catalyze reactions under very mild conditions with high degree of substrate specificity thus decreasing the formation of byproducts • Enzymes can catalyze reactions in different states, individual molecules in solution, in aggregates with other entities and as attached to surfaces

Immobilized enzymes • “Immobilized enzyme” refers to “enzymes physically confined or localized in a certain defined region of space with retention of their catalytic activities, and which can be used repeatedly and continuously” OR • Imprisonment of enzyme in a distinct support/matrix. The support/matrix allows exchange of medium that contains substrate of effecter or inhibitor molecules. • The substrate passes over the immobilized enzyme and is converted to products

Technological properties of Immobilized enzymes • • • • Advantages Increased functional efficiency Enhanced reproducibility Catalyst Reuse Easier Reactor Operation Easier Product Separation Wider choice of Reactor Minimum reaction time Less chance of contamination in products High stability High enzyme substrate ratio Enzyme-free products The ability to stop the reaction rapidly by removing the enzyme form the reaction solution Disadvantages • Limited industrial applications • Loss or reduction in activity in some enzymes • Some enzymes become unstable • Diffusion limitations • Additional Cost • High cost for isolation, purification and recovery of active enzyme. • Does not give required results if one of the substrate is insoluble • There may be diffusion problems for the substrate to access the enzyme under certain conditions

Applications of immobilization technique • Industrial production: e. g. , antibiotics, amino acids, beverages etc. • Biomedical applications: treatment, diagnosis, drug delivery • Food industry: production of jams, jellies and syrups • Waste water management: treatment of sewage and industrial effluents • Production of biodiesel: from vegetable oils • Textile industry: bio-polishing, desizing of fabrics • Detergent industry: immobilization of lipase for effective dirt removal

Applications of Immobilized Enzymes • The greatest industrial importance of immobilized enzymes is the ease with which they can be separated from reaction mixtures • Hence, in contrast to systems involving soluble enzymes - the reaction can be stopped by physical removal of the immobilized enzyme – without requiring such procedures as heat inactivation which might affect the products of the reaction • Furthermore, the enzyme will still be active and largely uncontaminated, so can be used again • For these reasons, immobilized enzymes are ideal for use in continuously operated processes

Supports/Matrix used in immobilization The matrix that holds the enzyme should be: cheap and easily available. Should not react with medium and enzyme. Three types of matrix are used: Natural polymers: alginate, chitosan and chitin, collagen, carrageenan, gelatin, cellulose, starch, pectin 2. Synthetic polymers: ion exchange resins/polymers [polyvinyl chloride (PVC), UV activated Polyethylene glycol (PEG)] 3. Inorganic materials: ceramics, silica, glass, activated carbon, charcoal • • 1.
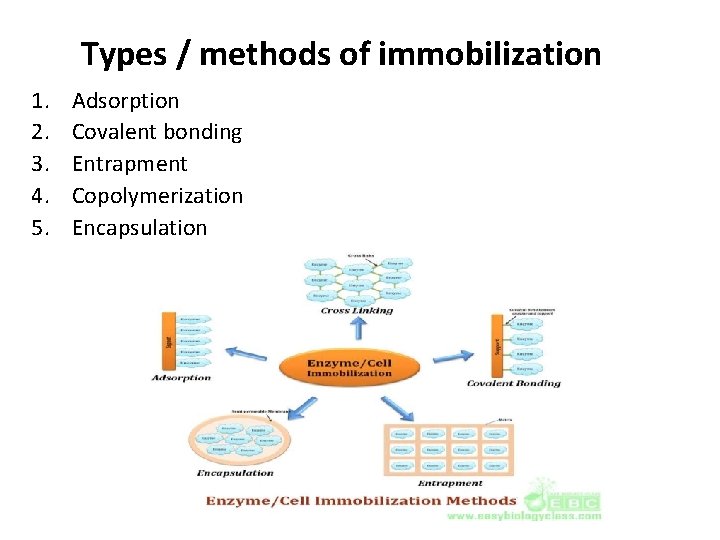
Types / methods of immobilization 1. 2. 3. 4. 5. Adsorption Covalent bonding Entrapment Copolymerization Encapsulation
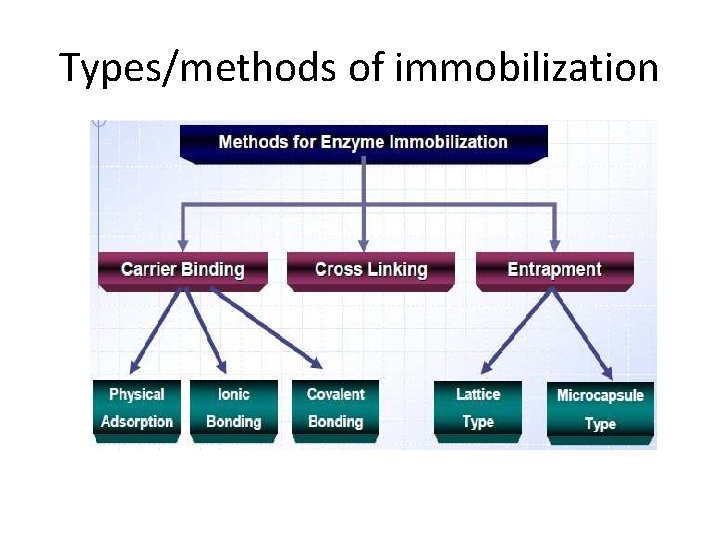
Types/methods of immobilization

1. Adsorption (Non Covalent Interactions) • Oldest and simplest method used for enzyme immobilization • Enzymes are adsorbed to external surface of support • Support /carrier may be: • Mineral support: aluminium oxide, clay • Organic support: starch • Ion exchange resins • Weak bonds are formed between the support and the enzyme (ionic interactions, hydrogen bonds, vand der waals forces) stabilize enzymes to the support • Carrier particle size must be small for appreciable surface bonding
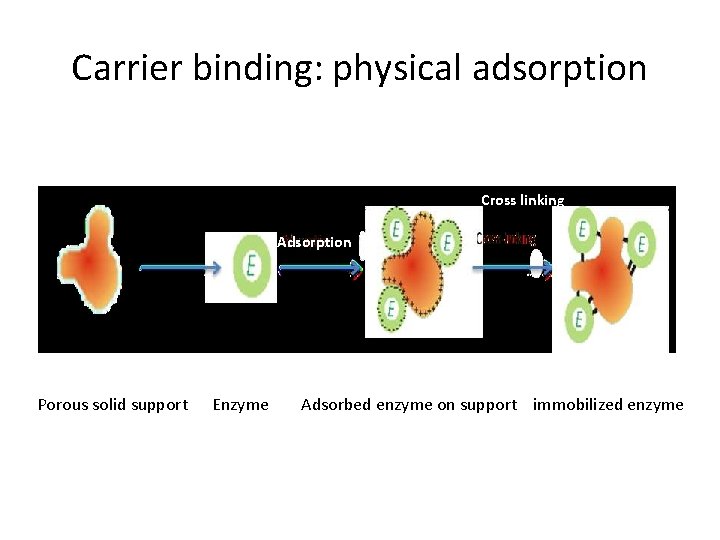
Carrier binding: physical adsorption Cross linking Adsorption Porous solid support Enzyme Adsorbed enzyme on support immobilized enzyme

Methods of adsorption • Static process: the solution containing enzyme is allowed to contact the carrier (no stirring) • Dynamic batch process: carrier is placed in the enzyme solution and mixed by stirring/agitation. • Reactor loading process: carrier is placed in the reactor and enzyme solution is transferred to reactor. • Electrode position process: carrier is placed proximal to an electrode in an enzyme bath and current is passed. The enzyme migrates to the carrier and deposited on the surface.

2. Covalent Bonding • Involves the formation of stable covalent bonds between enzyme and support. • Chemical groups in enzymes that form covalent bonds with support are amino groups (alpha amino group at N terminal), hydroxyl, carboxyl (alpha carboxyl group at C terminal), thiols and phenol rings. • The method is widely employed when there is a strict requirement for the absence of the enzyme in the product. • The enzyme is not released into the solution upon use. • The matrix has to be discarded together with the enzyme once the enzymatic activity decays. • The disadvantage of this type of immobilization is high cost in terms of generally low yield of immobilized activity and by the nonreversible character of this binding • Enzymes attached covalently by disulfide bonds to solid supports represent one way to avoid this problem.
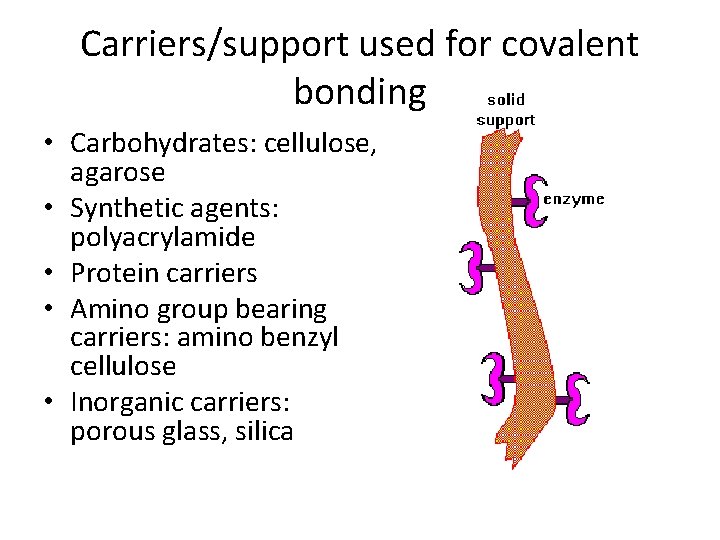
Carriers/support used for covalent bonding • Carbohydrates: cellulose, agarose • Synthetic agents: polyacrylamide • Protein carriers • Amino group bearing carriers: amino benzyl cellulose • Inorganic carriers: porous glass, silica

Covalent bonding (contd. ) • Advantages Strong linkage of enzyme to the support No leakage or desoprtion problem Comparatively simple method A variety of support materials with different functional groups available Wide applicability • Disadvantages Chemical modification of enzyme leading to functional conformation loss Enzyme inactivation by changes in conformation during reactions at active sites (can be overcome through immobilization in the presence of enzyme substrate or a competitive inhibitor)

3. Entrapment • The enzymes are physically entrapped inside a matrix. • The entrapment method refers to the occlusion of an enzyme within a polymeric network that allows the substrate and products to pass through but retains the enzyme. • Bonds involved may be covalent or non-covalent. • The enzyme is not bound to the matrix or membrane • There are different approaches to entrap enzymes such as gel or fiber entrapping and micro-encapsulation. • The practical use of these methods is limited by mass transfer limitations through membranes or gel
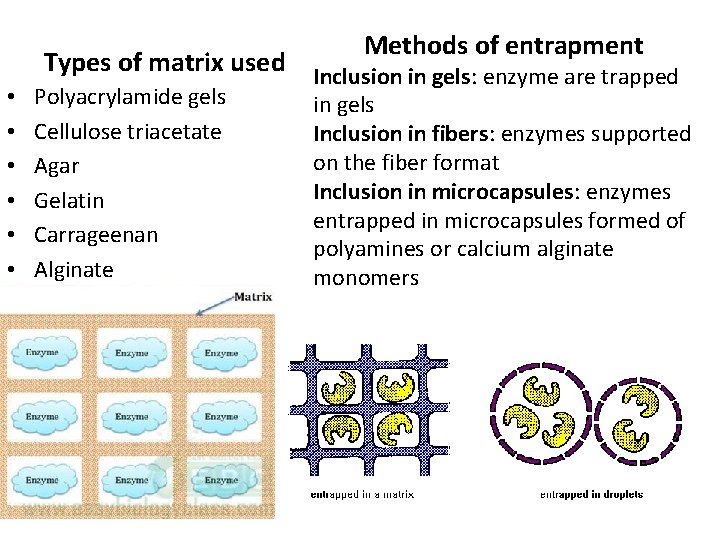
Methods of entrapment • • • Types of matrix used Inclusion in gels: enzyme are trapped Polyacrylamide gels Cellulose triacetate Agar Gelatin Carrageenan Alginate in gels Inclusion in fibers: enzymes supported on the fiber format Inclusion in microcapsules: enzymes entrapped in microcapsules formed of polyamines or calcium alginate monomers

Entrapment (contd. ) • Form and nature of matrix varies • Pore size of the matrix is adjusted to prevent enzyme loss • There might be a possibility of leakage of low MW enzymes • Agar and carrageenan have large pore size • Pore size can be adjusted by changing the concentration of polymer • The method is not commonly used in industrial process • Easy to practice at small scale

Entrapment (contd. ) Advantages Disadvantages • Loss of enzyme activity upon immobilization is minimized • Fast • Cheap due to availability of low cost matrix • Mild conditions are required • Less chances of conformational changes in enzymes • Leakage of enzyme in surrounding medium • Pore diffusion limitation • Substrate cannot diffuse deep into the gel matrix • Mass transfer resistance to substrates and products • Chance of microbial contamination

4. Cross linking (copolymerization) • Cross linking: covalent bonding between various groups of enzymes via polyfunctional reagents • No matrix or support are involved • Cheap and simple technique but not often used with pure proteins • Widely used in commercial preparations • However, polyfunctional reagents can denature the enzyme
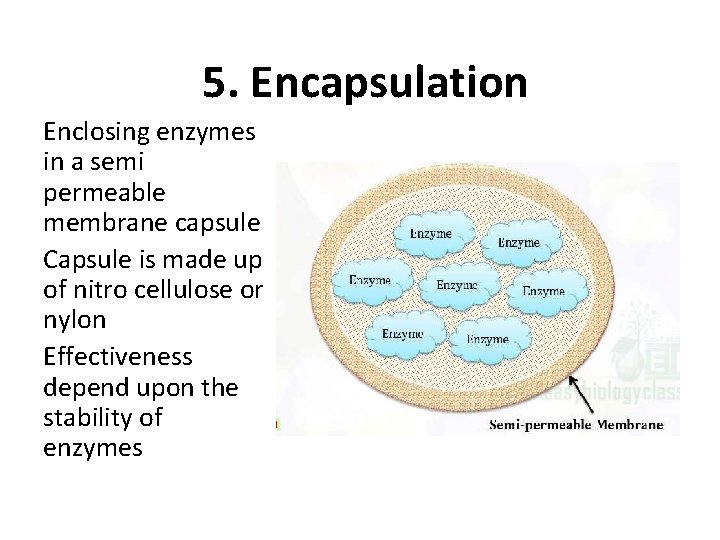
5. Encapsulation Enclosing enzymes in a semi permeable membrane capsule Capsule is made up of nitro cellulose or nylon Effectiveness depend upon the stability of enzymes

Encapsulation (contd. ) Advantages Disadvantage • Cheap and simple method • A large quantity of enzyme can be immobilized by this method • Pore size limitation • Only small substrate molecules are able to cross the membrane
Immobilization of cells • It is an alternative to enzyme immobilization • Well developed method for utilization of enzymes from microorganisms • The method is effectively used when: • Individual enzymes become inactive due to immobilization • Isolation and purification of enzyme is not cost effective • In this method enzymes remain stable and active for longer periods of time • Methods of cell immobilization are essentially the same as for enzyme immobilization
Immobilization of cells Advantages Disadvantages • Multiple enzymes can be introduced to a single step • Extraction and purification of enzymes are not required • Enzymes remain stable for a long period of time • Native conformation of enzyme is best maintained • Can be used to immobilize mitochondria and chloroplasts • Concentration of enzymes is less • Production of unwanted enzymes and products • Modification of end products by other enzymes

Methods of whole cell immobilization • Methods of cell immobilization are same as for enzyme immobilization and include • Adsorption • Covalent bonding • Cell to cell cross linking • Encapsulation • Entrapment
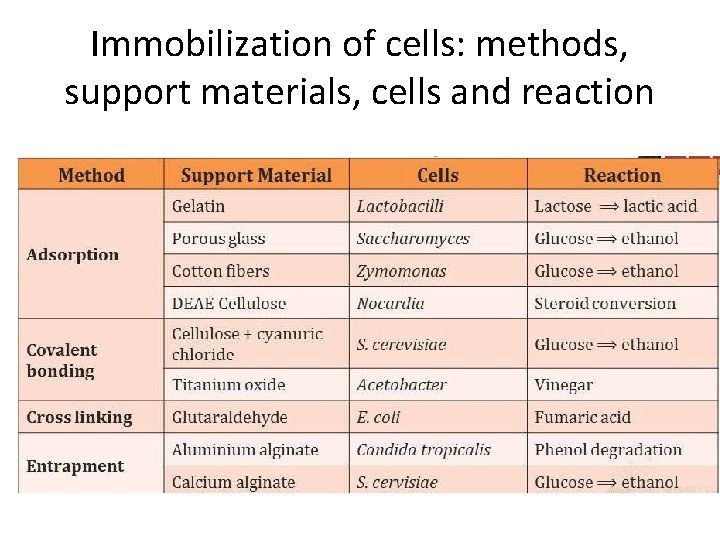
Immobilization of cells: methods, support materials, cells and reaction

REFERENCES • http: //enzymeimmobilization. blogspot. in/201 1/02/enzymeimmobilization. html • https: //en. wikipedia. org/wiki/Immobilized_en zyme • https: //www. google. co. in/search? q=enzyme+i mmobilization&espv=2&biw=1600&bih=766& source=lnms&tbm=isch&sa=X&ved=0 CAg. Q_A Uo. Amo. VCh. MIxuqan. KDOxw. IVE 3 GOCh 3 ul. Qif
- Slides: 27