Environmental Science Unit 2 Abiotic and Biotic Parts

Environmental Science Unit 2 Abiotic and Biotic Parts of Ecosystems Thanks to Dr. E – La Canada High School

Systems and Feedback n System: n n Open System: n n n A set of components or parts that function together to act as a whole. Not generally contained within boundaries Some energy or material moves into or out of the system Closed System: n No energy movement into or out of the system

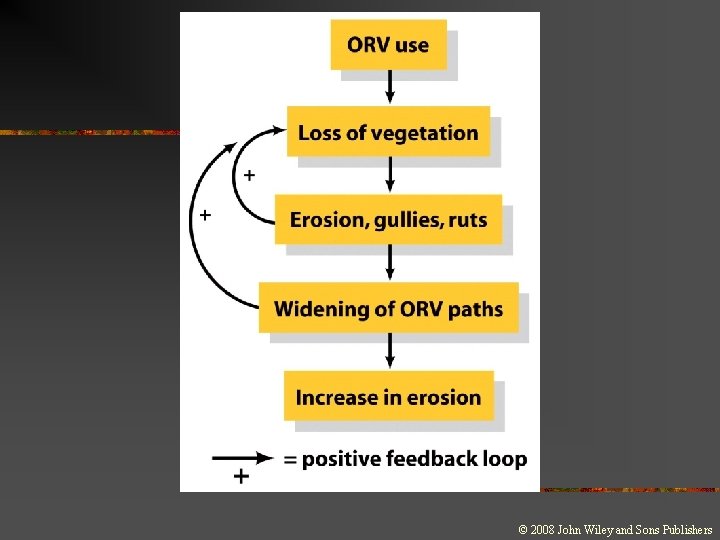
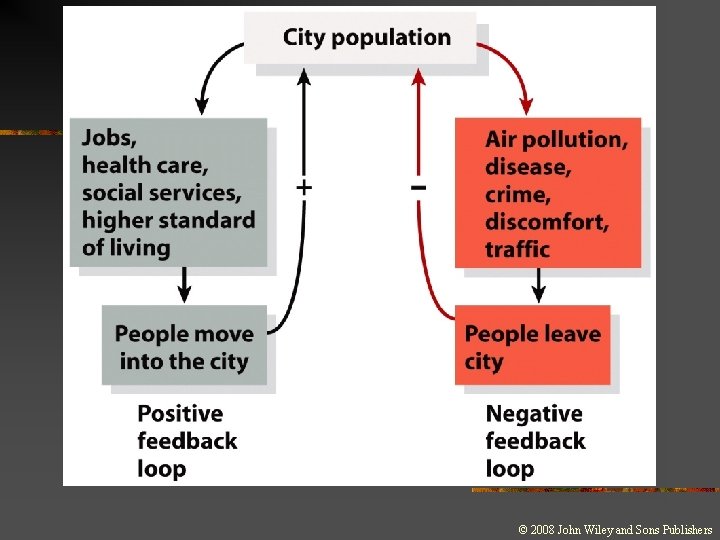
Systems and Feedback n n Negative Feedback n n n Occurs when the output of the system also serves as an input, leading to further changes in the system Occurs when the system’s response is in the opposite direction of the output Self-regulating Positive Feedback n Occurs when an increase in output leads to a further increase in output
© 2008 John Wiley and Sons Publishers
© 2008 John Wiley and Sons Publishers

Exponential Growth n Exponential growth: n n n Growth occurs at a constant rate per time period Equation to describe exponential growth is: Doubling time n n The time necessary for the quantity being measured to double. Approximately equal to 70 divided by the annual percentage growth rate

Environmental Unity n Environmental unity: n It is impossible to change only one thing; everything affects everything else.

Uniformitarianism n Uniformitarianism: n n The principle that processes that operate today operated in the past. Observations of processes today can explain events that occurred in the past and leave evidence “The present is the key to the past. ”

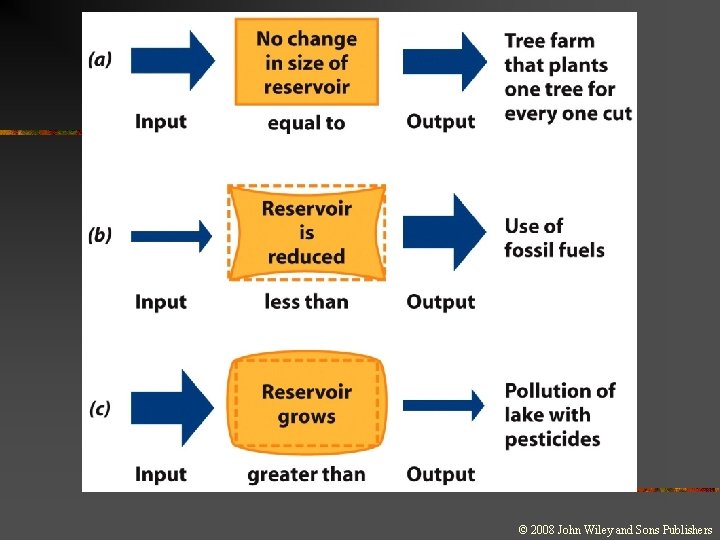
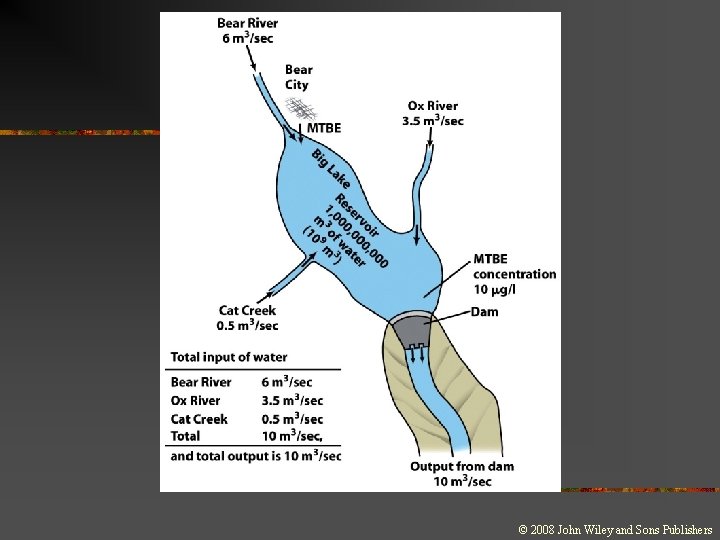
Changes and Equilibrium in Systems n Steady state: n n n A dynamic equilibrium Material or energy is entering and leaving the system in equal amounts Opposing processes occur at equal rates
© 2008 John Wiley and Sons Publishers

Changes and Equilibrium in Systems n Average residence time: n n The time it takes for a given part of the total reservoir of a particular material to be cycled through the sytem The equation for average residence time is: ART = S/F
© 2008 John Wiley and Sons Publishers

Earth as a Living System n Biota: n n All the organisms of all species living in an area or region up to and including the biosphere Biosphere: 1. 2. That part of a planet where life exists The planetary system that includes and sustains life

Ecosystem n Ecosystem: n n n A community of organisms and its local nonliving environment in which matter (chemical elements) cycles and energy flows. Sustained life on Earth is a characteristic of ecosystems Can be natural or artificial

Ecosystems n The Gaia Hypothesis: n n Named for Gaia, the Greek goddess Mother Earth States that the surface environment of the Earth, with respect to such factors as the n n atmospheric composition of gases acidity-alkalinity of waters Surface temperature actively regulated by the sensing, growth, metabolism and other activities of the biota. Or, life manipulates life the environment for the maintenance of life.

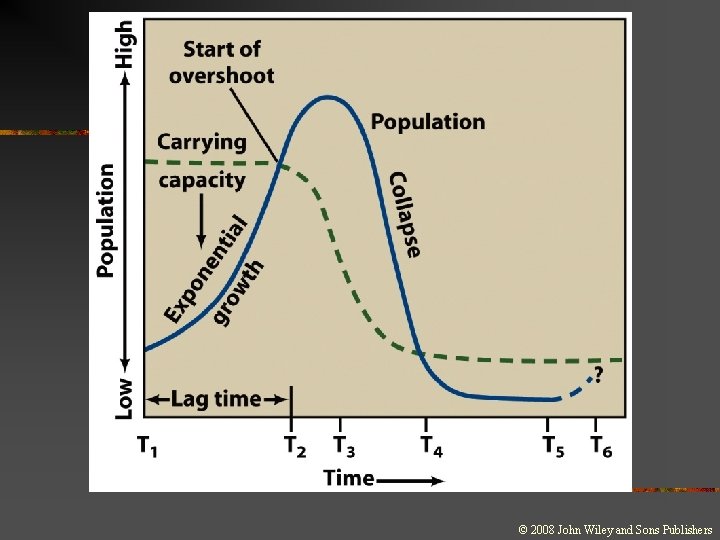
Why Solving Environmental Problems Is Often Difficult Exponential growth 1. • The consequences of exponential growth and its accompanying positive feedback can be dramatic Lag time 2. • • The time between a stimulus and the response of a system If there is a long delay between stimulus and response, then the resulting changes are much more difficult to recognize. Irreversible consequences 3. • Consequences that may not be easily rectified on a human scale of decades or a few hundred years.
© 2008 John Wiley and Sons Publishers

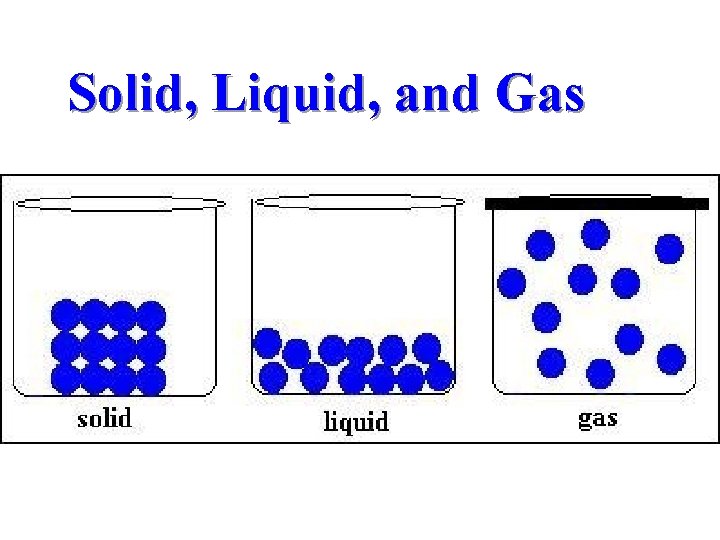
Matter Forms, Structure, and Quality n n n Matter is anything that has mass and takes up space. Matter is found in two chemical forms: elements and compounds. Various elements, compounds, or both can be found together in mixtures.
Solid, Liquid, and Gas

Atoms, Ions, and Molecules n n n Atoms: The smallest unit of matter that is unique to a particular element. Ions: Electrically charged atoms or combinations of atoms. Molecules: Combinations of two or more atoms of the same or different elements held together by chemical bonds.

What are Atoms? n n The main building blocks of an atom are positively charged PROTONS, uncharged NEUTRONS, and negatively charged ELECTRONS Each atom has an extremely small center, or nucleus, containing protons and neutrons.

http: //mediaserv. sus. mcgill. ca/content/2004 -Winter/180 -Winter/Nuclear/frame 0008. htm

Atomic Number and Mass Number. n Atomic number n The number of protons in the nucleus of each of its atoms. n Mass n The number total number of protons and neutrons in its nucleus.
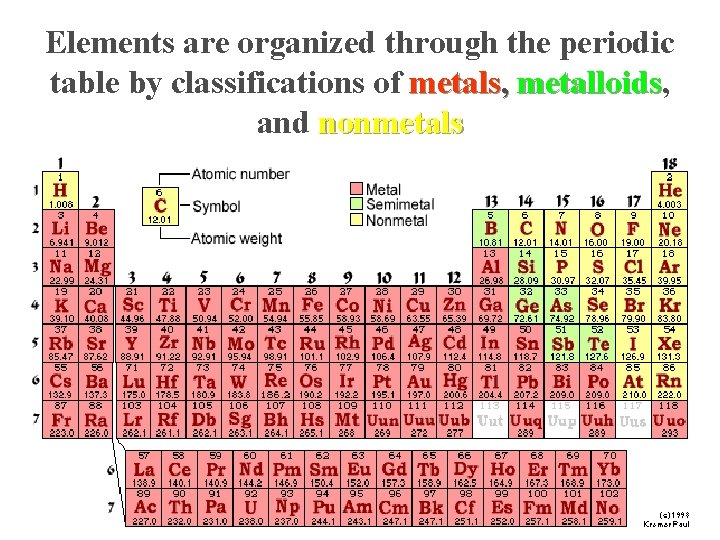
Elements are organized through the periodic table by classifications of metals, metalloids and nonmetals

Inorganic Compounds n n All compounds not Organic Ionic Compounds n n n sodium chloride (Na. Cl) sodium bicarbonate (Na. OH) Covalent compounds n n n hydrogen(H 2) carbon dioxide (CO 2) nitrogen dioxide (NO 2) sulfur dioxide (SO 2) Ammonia (NH 3)

Inorganic Compounds n n The earth’s crust is composed of mostly inorganic minerals and rock The crust is the source of all most nonrenewable resource Various combinations of only we use: fossil fuels, metallic minerals, etc. eight elements make up the bulk of most minerals.

Nonmetallic Elements. n Carbon (C), Oxygen (O), Nitrogen (N), Sulfur (S), Hydrogen (H), and Phosphorous (P). n Nonmetallic elements make up about 99% of the atoms of all living things.

Ionic Compounds Structure n n Composed of oppositely-charged ions Network of ions held together by attraction Ionic bonds n Forces of attraction between opposite charges

Formation of Ionic Compounds n Transfer of electrons between the atoms of these elements n n n Atom that is metal loses electrons (oxidation) to become positive Atom that is nonmetal gains electrons (reduction) to become negative Results in drastic changes to the elements involved
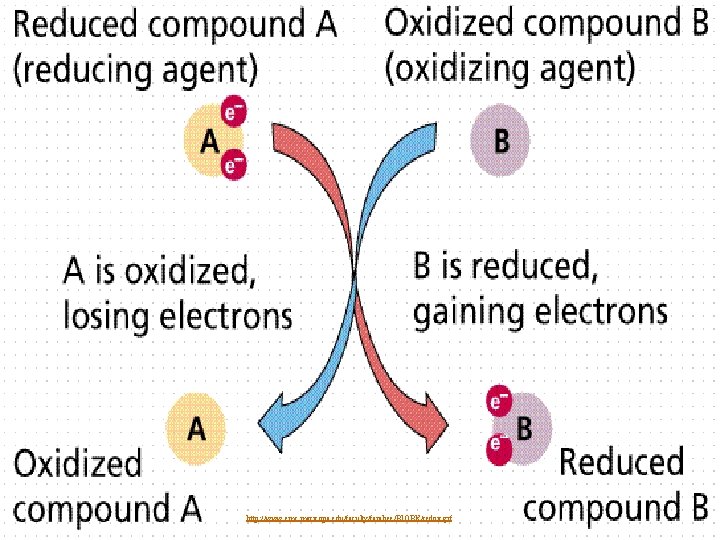
http: //www. emc. maricopa. edu/faculty/farabee/BIOBK/redox. gif

Sodium Chloride n n n Sodium is a rather "soft" metal solid, with a silver-grey color Chlorine is greenish colored gas When a single electron is transferred between these elements, their atoms are transformed via a violent reaction into a totally different substance called, sodium chloride, commonly called table salt -- a white, crystalline, and brittle solid

Covalent Bonds Formed by two non-metals n Similar electronegativities n Neither atom is "strong" enough to steal electrons from the other n Therefore, the atoms must share the electrons n

Covalent Bonds n n Chlorine atoms with valence electrons shown Chlorine atom has seven valence electrons, but wants eight When unpaired electron is shared, both atoms now have a full valence of eight electrons Individual atoms are independent, but once the bond is formed, energy is released, and the new chlorine molecule (Cl 2) behaves as a single particle

Organic Compounds n Compounds containing carbon atoms combined with each other with atoms of one or more other elements such as hydrogen, oxygen, nitrogen, sulfur, etc. n Hydrocarbons n n Chlorofluorocarbons n n Compounds of carbon and hydrogen Carbon, chlorine, and fluorine atoms Simple carbohydrates n carbon, hydrogen, oxygen combinations
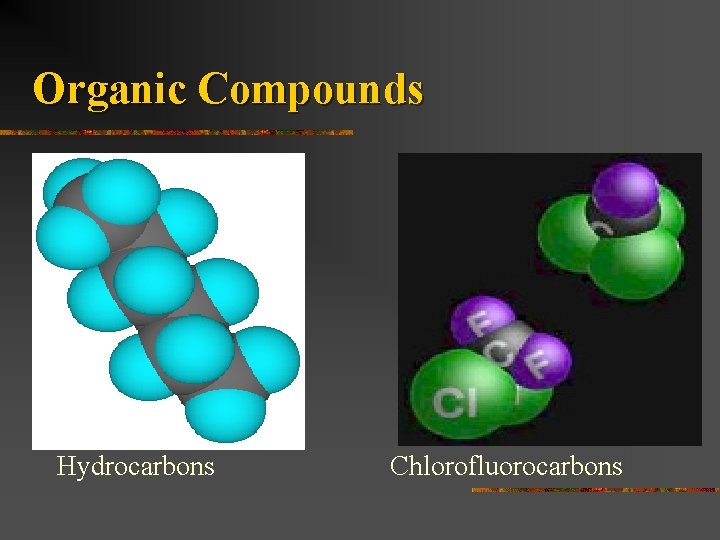
Organic Compounds Hydrocarbons Chlorofluorocarbons
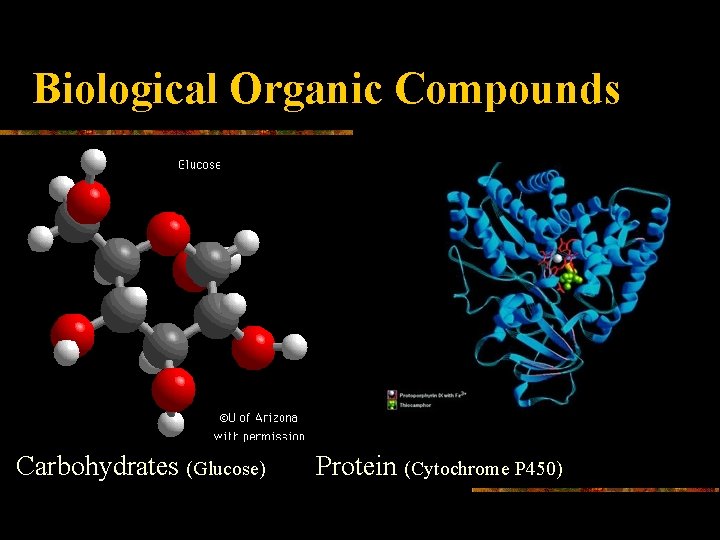
Biological Organic Compounds Carbohydrates (Glucose) Protein (Cytochrome P 450)
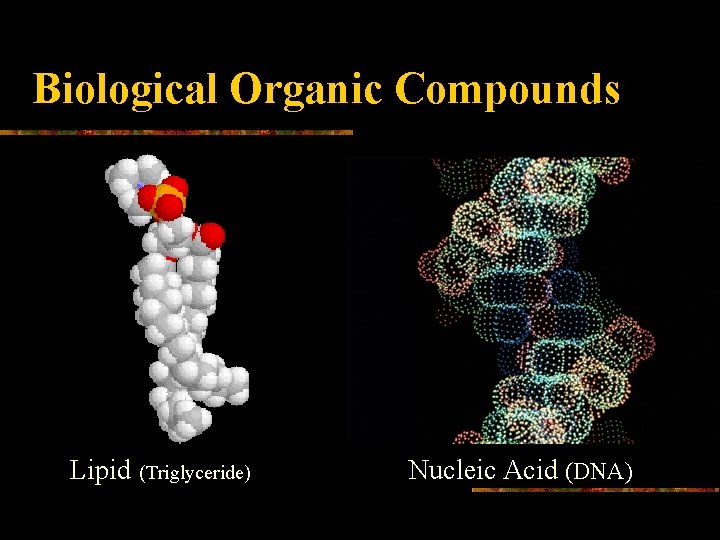
Biological Organic Compounds Lipid (Triglyceride) Nucleic Acid (DNA)
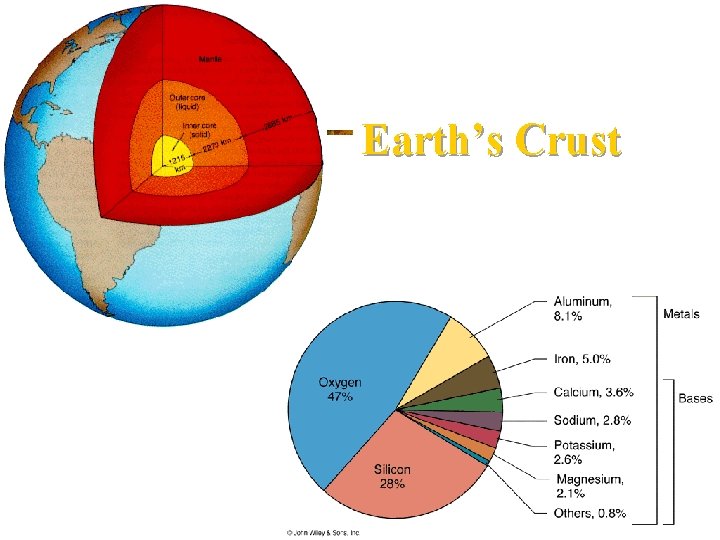
Earth’s Crust

Matter Quality n n n Matter quality is a measure of how useful a matter resource is, based in its availability and concentration. High quality matter is organized, concentrated, and usually found near the earth’s crust. Low quality is disorganized, dilute, and has little potential for use as a matter resource.

High quality & Low quality HIGH QUALITY LOW QUALITY

Energy n n n Energy is the capacity to do work and transfer heat. Energy comes in many forms: light, heat, and electricity. Kinetic energy is the energy that matter has because of its mass and its speed or velocity.
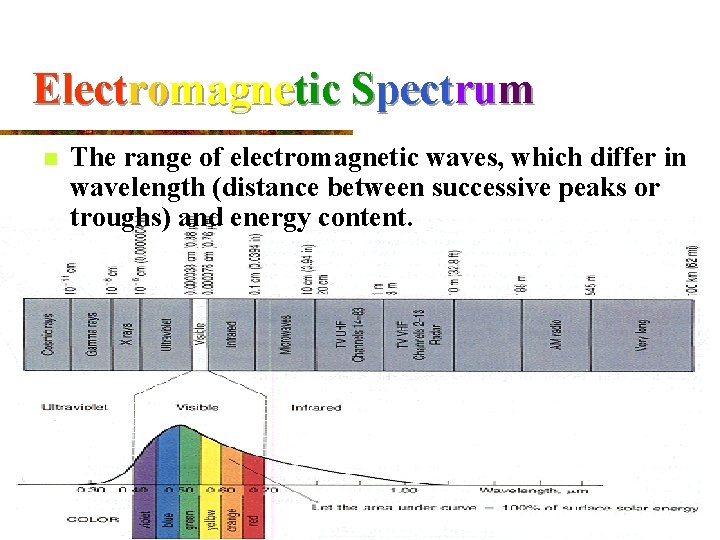
Electromagnetic Spectrum n The range of electromagnetic waves, which differ in wavelength (distance between successive peaks or troughs) and energy content.

Kinetic energy. n n n Kinetic energy is the energy that matter has because of its mass and its speed or velocity. It is energy in action or motion. Wind, flowing streams, falling rocks, electricity, moving car - all have kinetic energy.

Potential energy n n Potential energy is stored energy that is potential available for use. Potential energy can be changed to kinetic energy.

Energy Quality n n Very High: Electricity, Nuclear fission, and Concentrated sunlight. High: Hydrogen gas, Natural gas, and Coal. Moderate: Normal sunlight, and wood. Low: Low- temperature heat and dispersed geothermal energy.

The “Law of Conservation of Matter and Energy” n n In any nuclear change, the total amount of matter and energy involved remains the same. E = mc 2 n The energy created by the release of the strong nuclear forces for 1 kilogram of matter will produce enough energy to elevate the temperature of all the water used in the Los Angeles basin in one day by 10, 000 o. C

First Law of Thermodynamics n n n In all physical and chemical changes Energy is neither created nor destroyed But it may be converted from one form to another

Second Law of Thermodynamics n n n When energy is changed from one form to another Some of the useful energy is always degraded to lower-quality, more dispersed, less useful energy Also known as Law of Entropy

High Waste Societies n n People continue to use and waste more and more energy and matter resources at an increasing rate At some point, high-waste societies will become n UNSUSTAINABLE!

Goals of Matter Recycling Societies To allow economic growth to continue without depleting matter resources or producing excess pollution

Matter Recycling Societies Advantages n n Saves Energy Buys Time Disadvantages n n Requires high-quality energy which cannot be recycled Adds waste heat No infinite supply of affordable high-quality energy available Limit to number of times a material can be recycled

Low Waste Societies n Works with nature to reduce throughput n Based on energy flow and matter recycling

Low Waste Societies Function 1. Reuse/recycle most nonrenewable matter resources 2. Use potentially renewable resources no faster than they are replenished 3. Use matter and energy resources efficiently

Low Waste Societies Function 4. Reduce unnecessary consumption 5. Emphasize pollution prevention and waste reduction 6. Control population growth
www. sws. uiuc. edu/nitro/biggraph. asp
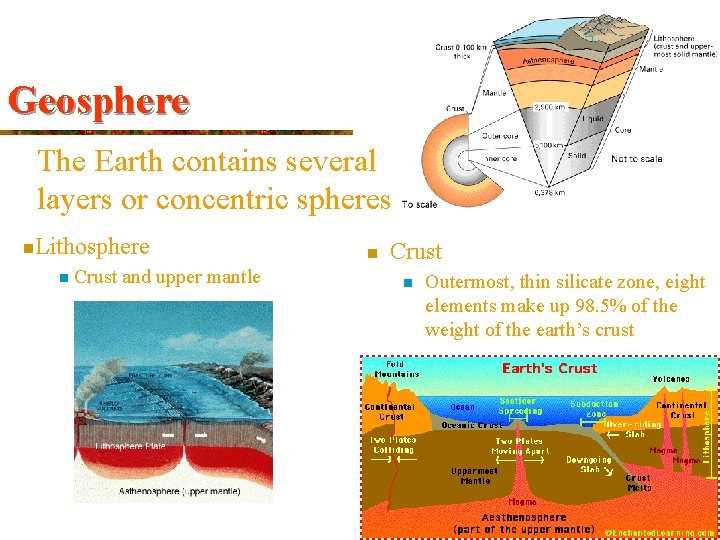
Geosphere The Earth contains several layers or concentric spheres n. Lithosphere n Crust and upper mantle n Crust n Outermost, thin silicate zone, eight elements make up 98. 5% of the weight of the earth’s crust
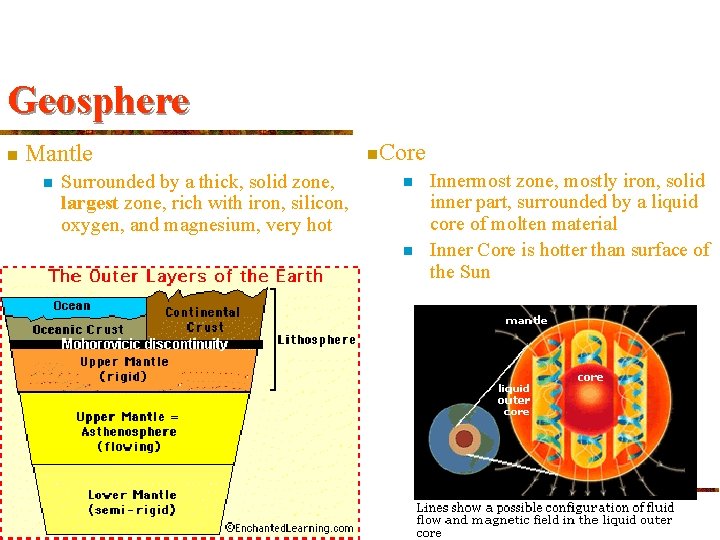
Geosphere n Mantle n Surrounded by a thick, solid zone, largest zone, rich with iron, silicon, oxygen, and magnesium, very hot n. Core n n Innermost zone, mostly iron, solid inner part, surrounded by a liquid core of molten material Inner Core is hotter than surface of the Sun
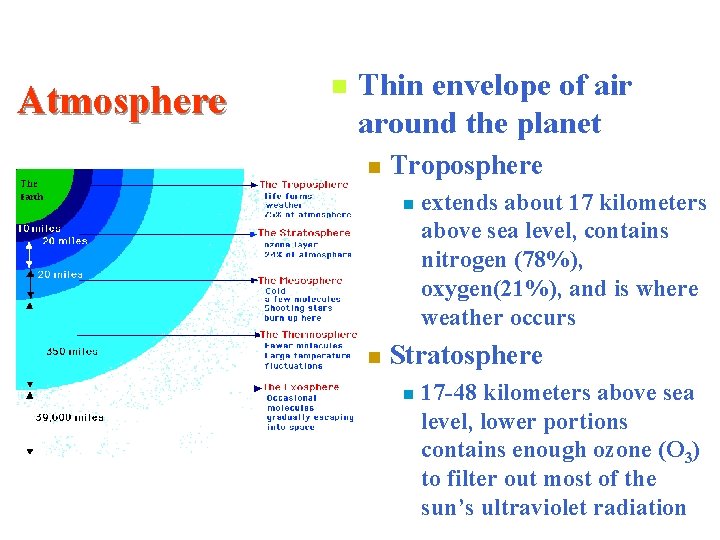
Atmosphere n Thin envelope of air around the planet n Troposphere n n extends about 17 kilometers above sea level, contains nitrogen (78%), oxygen(21%), and is where weather occurs Stratosphere n 17 -48 kilometers above sea level, lower portions contains enough ozone (O 3) to filter out most of the sun’s ultraviolet radiation

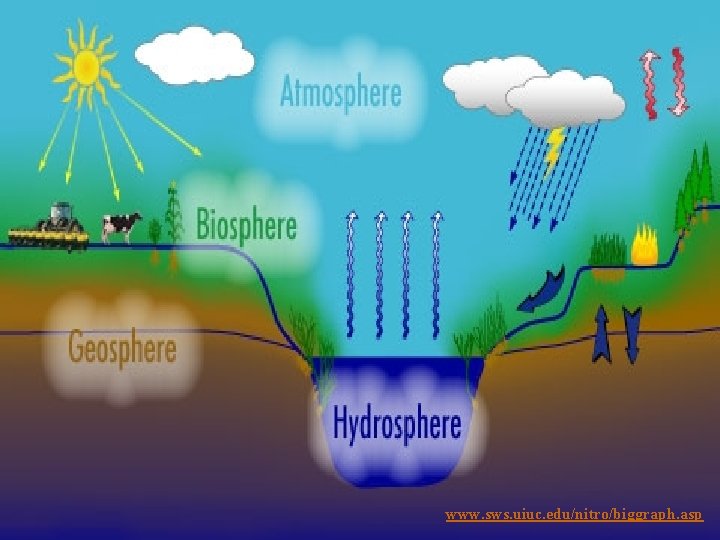
Hydrosphere Consists of the earth’s liquid water, ice, and water vapor in the atmosphere
What Sustains Life on Earth? n Life on the earth depends on three interconnected factors n n n One-way flow of high-quality energy from the sun Cycling of matter or nutrients (all atoms, ions, or molecules needed for survival by living organisms), through all parts of the ecosphere Gravity, which allows the planet to hold onto its atmosphere and causes the downward movement of chemicals in the matter cycles

Sun n n Fireball of hydrogen (72%) and helium (28%) Nuclear fusion Sun has existed for 6 billion years Sun will stay for another 6. 5 billion years Visible light that reaches troposphere is the ultraviolet ray which is not absorbed in ozone

Solar Energy 72% of solar energy warms the lands n 0. 023% of solar energy is captured by green plants and bacteria n Powers the cycling of matter and weather system n Distributes heat and fresh water n

www. bom. gov. au/lam/climate/levelthree/climch/clichgr 1. htm

Type of Nutrients n Nutrient n n n Macronutrient n n n Any atom, ion, or molecule an organism needs to live grow or reproduce Ex: carbon, oxygen, hydrogen, nitrogen… etc nutrient that organisms need in large amount Ex: phosphorus, sulfur, calcium, iron … etc Micronutrient n n nutrient that organism need in small amount Ex: zinc, sodium, copper… etc

Limiting Factor n More important than others in regulating population growth n n Ex: water light, and soil Lacking water in the desert can limit the growth of plants

Limiting Factor Principle n too much or too little of any abiotic factor can limit growth of population, even if all the other factors are at optimum (favorable) range of tolerance. n Ex: If a farmer plants corn in phosphorus-poor soil, even if water, nitrogen are in a optimum levels, corn will stop growing, after it uses up available phosphorus.

Living Organisms in Ecosystem Producers or autotrophs- makes their own food from compound obtained from environment. n Ex: sun plant gets energy or food from

Living Organisms in Ecosystem Photosynthesis- ability of producer to convert sunlight, abiotic nutrients to sugars and other complex organic compounds n Chlorophyll- traps solar energy and converts into chemical energy
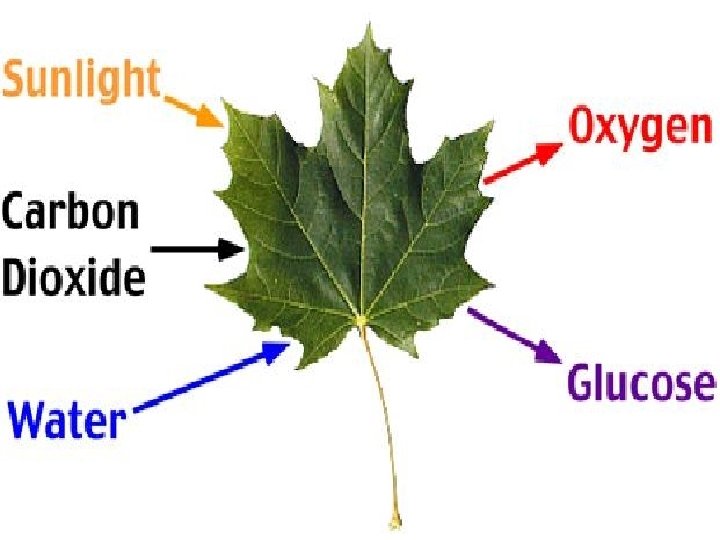

n Producer transmit 1 -5% of absorbed energy into chemical energy, which is stored in complex carbohydrates, lipids, proteins and nucleic acid in plant tissue

Chemosynthesisn Bacteria can convert simple compounds from their environment into more complex nutrient compound without sunlight n n Ex: becomes consumed by tubeworms, clams, crabs Bacteria can survive in great amount of heat

Respiration n Aerobic Respiration n Uses oxygen to convert organic nutrients back into carbon dioxide and water Glucose + oxygen Carbon dioxide + water + energy Anaerobic Respiration or Fermentation n Breakdown of glucose in absence of oxygen

Second Law of Energy n Organisms need high quality chemical energy to move, grow and reproduce, and this energy is converted into low-quality heat that flows into environment n n Trophic levels or feeding levels- Producer is a first trophic level, primary consumer is second trophic level, secondary consumer is third. Decomposers process detritus from all trophic levels.

Chapter 5 Nutrient Cycles and Soils

Matter Cycling in Ecosystems n Nutrient n Natural or Biogeochemical Cycles processes that recycle nutrients in various chemical forms in a cyclic manner from the nonliving environment to living organisms and back again

Nutrient Cycles (Closed System) Energy Flow (Open System) n Water n Sulfur n Carbon n Rock n Nitrogen n Soil n Phosphorus n Energy Flow

Biogeochemical Cycle Locations n Hydrosphere n n n Atmospheric n n n Water in the form of ice, liquid, and vapor Operates local, regional, and global levels Large portion of a given element (i. e. Nitrogen gas) exists in gaseous form in the atmosphere Operates local, regional, and global levels Sedimentary n n The element does not have a gaseous phase or its gaseous compounds don’t make up a significant portion of its supply Operates local and regional basis

Nutrient Cycling & Ecosystem Sustainability n Natural ecosystems tend to balance n n Humans are accelerating rates of flow of mater n n n Nutrients are recycled with reasonable efficiency Nutrient loss from soils Doubling of normal flow of nitrogen in the nitrogen cycle is a contributes to global warming, ozone depletion, air pollution, and loss of biodiversity Isolated ecosystems are being influenced by human activities
- Slides: 78