Environmental Pollution Environmental pollution can be defined as

Environmental Pollution Environmental pollution can be defined as any undesirable change in the physical, chemical or biological characteristics of any component of the environment ( air, water, soil), which can cause harmful effects on various form of life or property. Pollutant are defined as matter or energy which leads to undesirable changes in the environment. Pollutants include solid, liquid or gaseous substances present in greater than natural abundance produced due to human activity, which have a detrimental effect on our environment. From an ecological perspective pollutants can be classified as follows: 1) Degradable or non-persistent pollutants: These can be rapidly broken down by natural processes. Eg: domestic sewage, discarded vegetables etc. 2) Slowly degradable or persistent pollutants: Pollutants that remain in the environment for many years in an unchanged condition and take decades or longer to degrade. Eg: DDT and most plastics. 3) Non-degradable pollutants: These cannot be degraded by natural processes. Once they are released into the environment they are difficult to eradicate and continue to accumulate. Eg: toxic elements like lead or mercury.

Environmental Pollution could be of various types: 1) Air Pollution: Air pollution occurs due to the presence of certain substances (including the normal constituent in excess) in concentration which can cause undesirable effects on human health, property and structure. These substance include gases, particulate matter, radioactive substances etc. Classification of Air Pollutants: Based upon their origin air pollutant are classified into two type: a) Primary Pollutants: Pollutants that are emitted directly from identifiable sources are produced both by natural events (for example, dust storms and volcanic eruptions) and human activities (emission from vehicles, industries, etc. ). There are five primary pollutants that together contribute about 90 percent of the global air pollution. These are carbon oxides (CO and CO 2), nitrogen oxides, sulfur oxides, volatile organic compounds (mostly hydrocarbons) and suspended particulate matter.

Major primary pollutant: 1) Particulate matter: Particulates are small pieces of solid material (for example, smoke particles from fires, bits of asbestos, dust particles and ash from industries) dispersed into the atmosphere. The effects of particulates range from soot to the carcinogenic (cancer causing) effects of asbestos, dust particles and ash from industrial plants that are dispersed into the atmosphere. Some definition: a) Aerosol : General term for particles suspended in air. Example: Sprays from pressurized cans b) Mist : Aerosol consisting of liquid droplets. Example: Sulfuric acid mist c) Dust : Aerosol consisting of solid particles that are blown into the air or are produced from larger particles by grinding them down. Example : Dust storm

d ) Smoke: Aerosol consisting of solid particles or a mixture of solid and liquid particles produced by chemical reaction such as fires. Example: Cigarette smoke, smoke from burning garbage. e) Fume : It applies specifically to aerosols produced by condensation of hot vapors of metals. Example: Zinc/lead fumes f) Plume : Geometrical shape or form of the smoke coming out of a chimney. g) Fog : Aerosol consisting of water droplets. h) Smog : Term used to describe a mixture of smoke and fog. 2) Hydrocarbon: Hydrocarbons are a group of compounds consisting of carbon and hydrogen atoms. They either evaporate from fuel supplies or are remnants of fuel that did not burn completely. They act as precursor for the production of secondary pollutant. 3) Nitrogen oxides: Nitrogen oxides are found in vehicular exhausts. Nitrogen oxides are significant, as they are involved in the production of secondary air pollutants such as ozone. They are also important component of acid rain.

4) Sulfur oxides: are produced when sulfur containing fossil fuels are burnt. They are major source of increase acidity in the atmospheric rain, a phenomenon known as acid rain. 5) Carbon monoxide: is a colorless, odorless and toxic gas produced when organic materials such as natural gas, coal or wood are incompletely burnt. Vehicular exhausts are the single largest source of carbon monoxide. Carbon monoxide is however not a persistent pollutant. Natural processes can convert carbon monoxide to other compounds that are not harmful. 6) Carbon dioxide: It is a component of atmospheric system and currently make 0. 038 per cent of the atmosphere. It is important for maintaining the biotic system as source of carbon. However during past decade its concentration is increasing in atmosphere due to burning of fossils fuel and land use changes. It shows property of green house gas, thereby results into a phenomenon known as global warming. Beside these primary pollutant there are certain pollutant which are not a natural component of atmosphere, however it is introduced by human activities, such as CFC, MIC (methyl isocyanate).
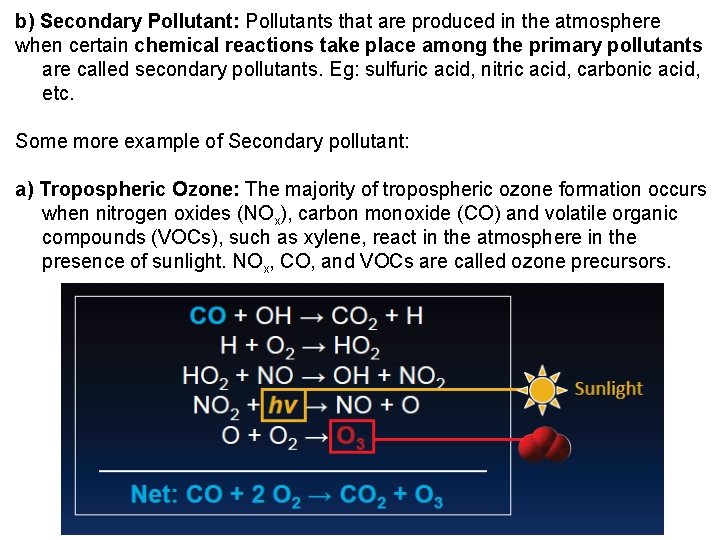
b) Secondary Pollutant: Pollutants that are produced in the atmosphere when certain chemical reactions take place among the primary pollutants are called secondary pollutants. Eg: sulfuric acid, nitric acid, carbonic acid, etc. Some more example of Secondary pollutant: a) Tropospheric Ozone: The majority of tropospheric ozone formation occurs when nitrogen oxides (NOx), carbon monoxide (CO) and volatile organic compounds (VOCs), such as xylene, react in the atmosphere in the presence of sunlight. NOx, CO, and VOCs are called ozone precursors.

b) Peroxyacetyl nitrate (PAN): Acyl peroxy nitrates, or Peroxyacyl nitrates (also known as APNs, or PANs), are powerful respiratory and eye irritants present in photochemical smog. CH 3 C(O)OO· + NO 2 → CH 3 C(O)OONO 2 General equation: Hydrocarbons + O 2 + light → Cx. Hy. C(O)OO· + NO 2 → Cx. Hy. C(O)OONO 2 Sources of Air Pollutants: It is mainly divided into two types: a) Natural sources: The natural sources of air pollution are volcanic eruption, forest fire, sea salt sprays, biological decay, pollen grains of flowers, spore etc. Radioactive material present in earth crust are the sources of radioactivity in the atmosphere. b) Man-made (Anthropogenic sources): it include thermal power plant, industrial units, vehicular emissions, fossil fuel burning, agricultural activities etc.

Human can be affected by both indoor or outdoor air pollution. The most important indoor air pollutant is radon gas ( causing lung cancer) a radioactive atomic gas that results from the radioactive decay of radium, which may be found in rock formations beneath buildings or in certain building materials (bricks, concrete, tiles etc) themselves. Beside radon carbon monoxide, sulphur dioxide and hydrocarbon released due to incomplete combustion of fossil fuel, wood for cooking purpose are other major air pollutant. Fate of pollutant in the atmosphere: • Once pollutants enter the troposphere they are transported downwind, diluted by the large volume of air, transformed through either physical or chemical changes or are removed from the atmosphere by rain during which they are attached to water vapor that subsequently forms rain or snow that falls to the earth’s surface. • However this transportation and dilution of air pollutant depend strongly upon the metrological condition prevailing in that area. In an unstable atmosphere with high wind speed dispersion and dilution rate of pollutant are much higher compare to a stable atmosphere.
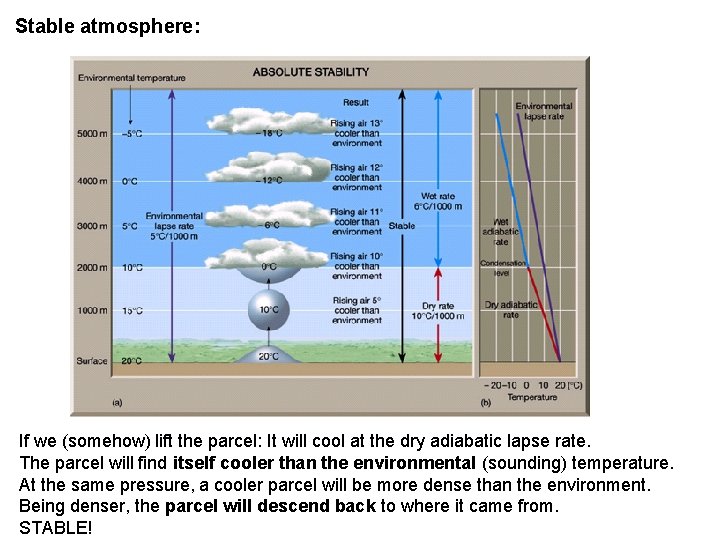
Stable atmosphere: If we (somehow) lift the parcel: It will cool at the dry adiabatic lapse rate. The parcel will find itself cooler than the environmental (sounding) temperature. At the same pressure, a cooler parcel will be more dense than the environment. Being denser, the parcel will descend back to where it came from. STABLE!
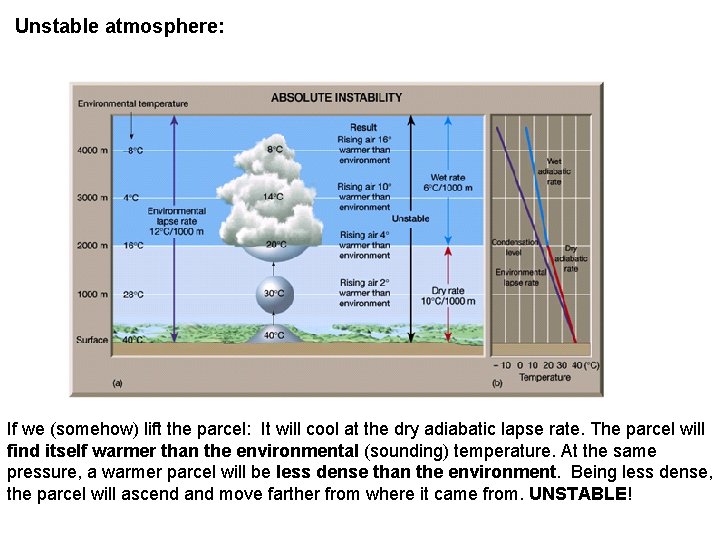
Unstable atmosphere: If we (somehow) lift the parcel: It will cool at the dry adiabatic lapse rate. The parcel will find itself warmer than the environmental (sounding) temperature. At the same pressure, a warmer parcel will be less dense than the environment. Being less dense, the parcel will ascend and move farther from where it came from. UNSTABLE!

Temperature Inversion: • Although the temperature of air relatively near the earth’s surface normally decreases with increasing altitude, certain atmospheric conditions can result in the opposite condition—increasing temperature with increasing altitude. Such conditions are characterized by high atmospheric stability and are known as temperature inversions. Because they limit the vertical circulation of air, temperature inversions result in air stagnation and the trapping of air pollutants in localized areas. Frontal inversion: An inversion can form from the collision of a warm air mass (warm front) with a cold air mass (cold front). The warm air mass overrides the cold air mass in the frontal area, producing the inversion. Radiation inversions: It is the most common form of surface inversion and occurs when the earth's surface cools rapidly. As the earth cools, so does the layer of air close to the surface. If this air cools to a temperature below that of the air above, it becomes very stable, and the layer of warmer air impedes any vertical motion. Radiation inversions usually occur in the late evening through the early morning under clear skies with calm winds, when the cooling effect is greatest.
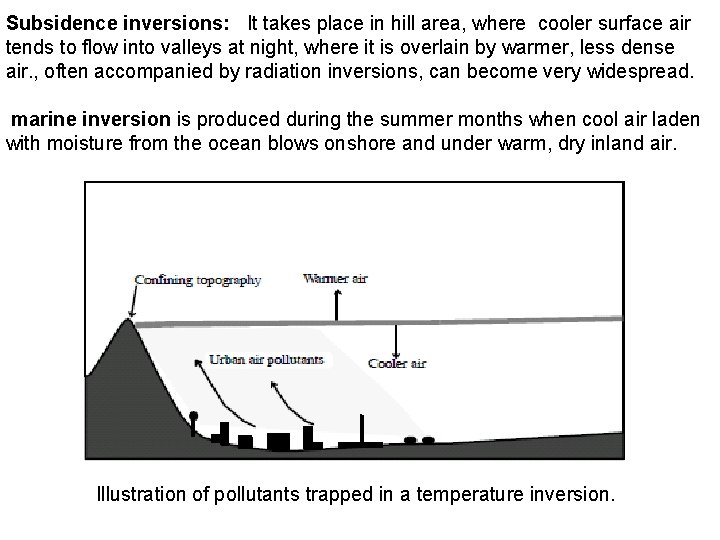
Subsidence inversions: It takes place in hill area, where cooler surface air tends to flow into valleys at night, where it is overlain by warmer, less dense air. , often accompanied by radiation inversions, can become very widespread. marine inversion is produced during the summer months when cool air laden with moisture from the ocean blows onshore and under warm, dry inland air. Illustration of pollutants trapped in a temperature inversion.

Impact of Temperature Inversion: Due to temperature inversion condition, environment become more stable thereby prevent dispersal and dilution of air pollutant from localized areas. One major air pollution scenario due to temperature inversion condition is London Smog (winter of 1952). The weather in Greater London had been unusually cold for several weeks leading up to the event. Because of the cold weather, households were burning more coal than usual to keep warm. The smoke from approximately one million coal-fired stoves, in addition to the emissions from local industry, was released into the atmosphere. Thousands of tons of black soot, tar particles, and sulfur dioxide had accumulated in the air from the heavy coal combustion. Estimates of PM 10 concentrations during December, 1952, range between 3, 000 and 14, 000 μg/m³ with the high range being approximately 50 times higher than normal levels at the time. PM 10 is particulate matter less than 10 micrometers in diameter. Due to temperature inversion condition the thick, smoke-polluted air to be trapped under the inversion. The following 114 hours in London experienced visibility less than 500 meters with 48 hours below 50 meters visibility. The smog-related deaths were primarily attributed to pneumonia, bronchitis, tuberculosis, and heart failure.

Effect of air pollution: Air pollution has adverse effects on living organisms and materials. 1) Effects on human health: • Air pollutants especially particulate matter( PM) is related to respiratory disease in human being. Particles come in a wide range of sizes. Those less than 10 micrometers in diameter (PM 10) are so small that they can get into the lungs, potentially causing serious health problems. Suspended particulates can cause damage to lung tissues and diseases like asthma, chronic bronchitis and emphysema( damage of air sacs leading to loss of lung elasticity and acute shortness of breath. Metal in form of PM like lead, Asbestos etc also cause cancer or affect vital organs like kidney, liver, spleen etc. • Carbon monoxides combine with hemoglobin of blood to form carboxyhaemoglobin, due to which oxygen carrying capacity to various part of the body decrease which may results into suffocation, dizziness, unconsciousness and even death. • Hydrocarbon emission from vehicle or industrial units ( benzene, formaldehyde etc. ) may results into mutation, reproductive problem or even cancer.

2) Effects on Plant: Air pollution affects plant by entering through stomata (leaf pores through which gases diffuse), destroy chlorophyll and affect photosynthesis. Gaseous pollutant like SO 2, ozone etc can leads to necrosis( dead areas of leaf), chlorosis( yellowing of leaf due to reduction of chlorophyll), abscission ( dropping of leaves). 3) Effects on aquatic life: Air pollutants (SOx and NOx) when mix up with rain can results into acid rain which reduces the p. H of fresh water lakes especially in the higher latitude. This reduction in p. H has adverse effect on aquatic biotic life. 4) Effects on materials: Metal parts of building, vehicle, bridges, wires and railway tracks are affected due to corrosion by particulate matter which further get accerlated in presence of SO 2 and moisture. Acid rain also affect the structure made up of marble and limestone. Example: Yellowing of Tajmahal in Agra. Ozone in the atmosphere can cause cracking of rubber tyres. Beside these effect, air pollutants also leads to stratospheric Ozone depletion which results into increase UV-rays exposure on earth surface and Global warming due to green house gases are major environmental problem.

Control of Air Pollution: Air pollution can be controlled by applying various measures such as: 1) Zonation in landuse pattern: Industries should be set up far away from the residential areas and Industries should be located in places so as to minimize the effects of pollution after considering the topography and the wind directions. 2) Dilution of emission: It can be done by increasing stack height, beyond inversion layer. Wind current will disperse the pollutant. 3) Low Sulphur coal in industries to reduce emission of SOx from coal burning. 4) Installment of pollutant control equipment such as cyclones, bag house filters etc to reduce pollutant emission from industry. 5) Establish vehicular emission norm to reduce emission from vehicles, regular tuning of engines, installing catalytic converters, engine modification to have fuel efficient (lean) mixture to reduce CO and hydrocarbon emission, slow and cooler burning of fuels to reduce NOx emission( Honda Technology). 6) Using mass transport system, bicycles etc.
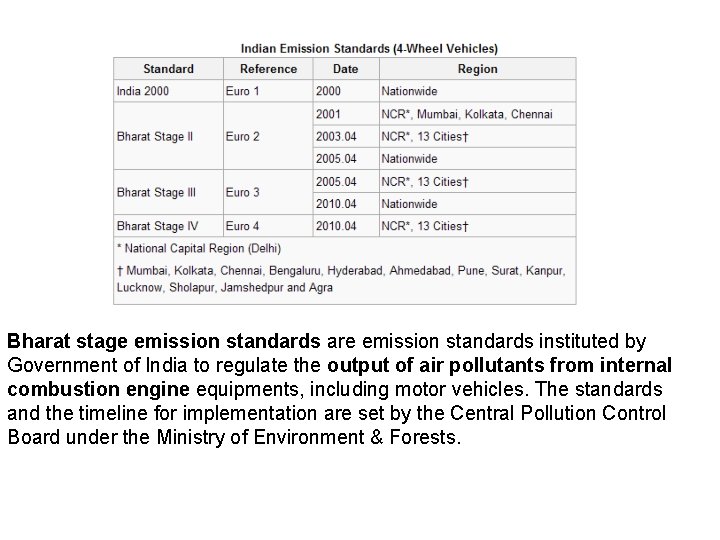
Bharat stage emission standards are emission standards instituted by Government of India to regulate the output of air pollutants from internal combustion engine equipments, including motor vehicles. The standards and the timeline for implementation are set by the Central Pollution Control Board under the Ministry of Environment & Forests.

Air Pollution control through pollution control equipment: 1) Particulate matter: Many devices are available for removal of particulate matter and choice of which depend upon characteristics of particulate matter, flow rate, collection efficiency, costs etc. a) Cyclone: These are employed for large size particle. The gas with particle in it enters tangentially at the top of cylinder and spin forming a vortex. Due to centrifugal force, particle strike the wall of cylinder.
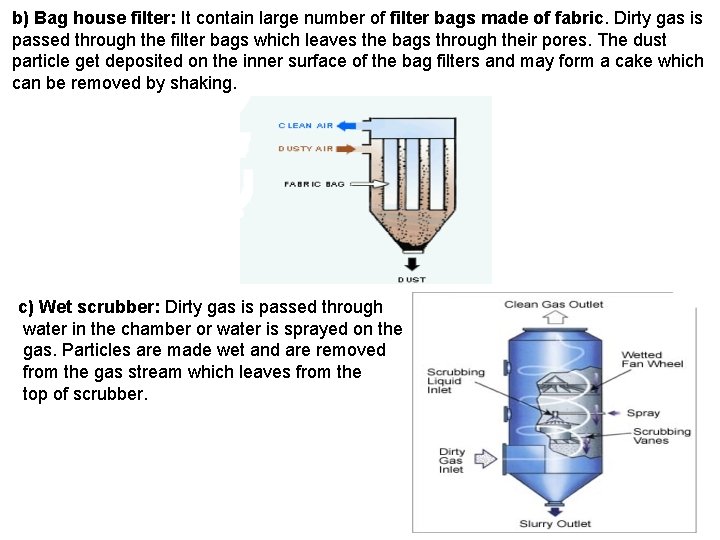
b) Bag house filter: It contain large number of filter bags made of fabric. Dirty gas is passed through the filter bags which leaves the bags through their pores. The dust particle get deposited on the inner surface of the bag filters and may form a cake which can be removed by shaking. c) Wet scrubber: Dirty gas is passed through water in the chamber or water is sprayed on the gas. Particles are made wet and are removed from the gas stream which leaves from the top of scrubber.
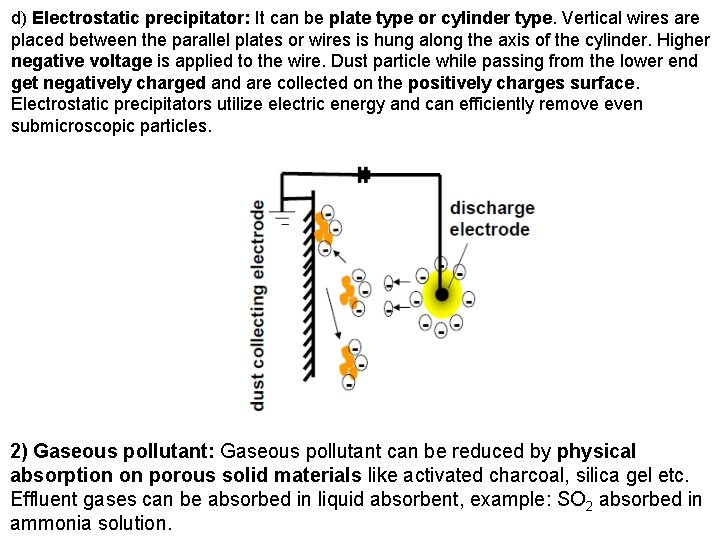
d) Electrostatic precipitator: It can be plate type or cylinder type. Vertical wires are placed between the parallel plates or wires is hung along the axis of the cylinder. Higher negative voltage is applied to the wire. Dust particle while passing from the lower end get negatively charged and are collected on the positively charges surface. Electrostatic precipitators utilize electric energy and can efficiently remove even submicroscopic particles. 2) Gaseous pollutant: Gaseous pollutant can be reduced by physical absorption on porous solid materials like activated charcoal, silica gel etc. Effluent gases can be absorbed in liquid absorbent, example: SO 2 absorbed in ammonia solution.
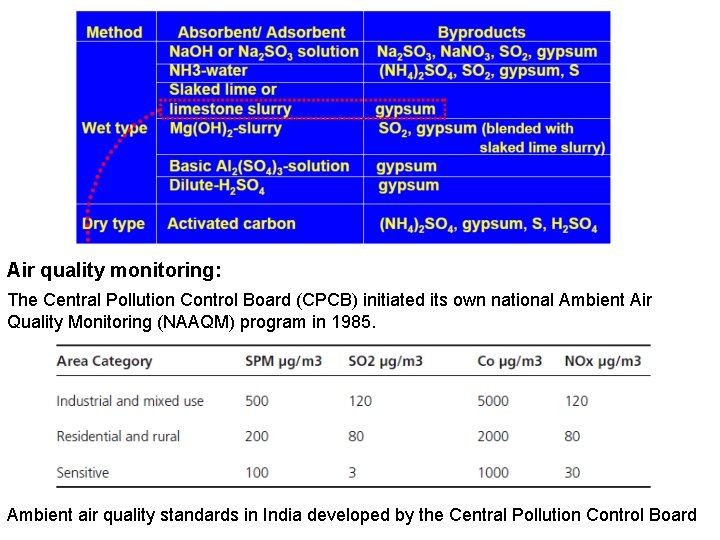
Air quality monitoring: The Central Pollution Control Board (CPCB) initiated its own national Ambient Air Quality Monitoring (NAAQM) program in 1985. Ambient air quality standards in India developed by the Central Pollution Control Board

Legal aspects of air pollution control in India: The Air (Prevention and Control of Pollution) Act was legislated in 1981. The Act provided for prevention, control and abatement of air pollution. In areas notified under this Act no industrial pollution causing activity could come up without the permission of the concerned State Pollution Control Board. To regulate vehicular pollution the Central Motor Vehicles Act of 1939 was amended in 1989. Following this amendment the exhaust emission rules for vehicle owners were notified in 1990 and the mass emission standards for vehicle manufacturers were enforced in 1991 for the first time. The mass emission norms have been further revised for 2000.
- Slides: 22