Environmental EngineeringI Water Supply Engineering Prof Rajesh Bhagat

Environmental Engineering-I (Water Supply Engineering) Prof. Rajesh Bhagat Asst. Professor, CED, YCCE, Nagpur B. E. (Civil Engg. ) GCOE, Amravati M. Tech. (Enviro. Engg. ) VNIT, Nagpur Experience & Achievement: Selected Scientist, NEERI-CSIR, Govt. of India. GATE Qualified Three Times. Selected Junior Engineer, ZP Washim. Three Times Selected as UGC Approved Assistant Professor, PCE, Nagpur. Assistant Professor, Cummins College of Engg. For Women (MKSSS, Nagpur) Topper of Pre-Ph. D Course Work at UGC-HRDC, RTMNU Nagpur Mobile No. : - 8483002277 / 8483003474 Email ID : - rajeysh 7 bhagat@gmail. com Website: - www. rajeysh 7 bhagat. wordpress. com

UNIT – III Water Quality : Generalidea of water borne diseases, Physical, Chemical and biological characteristics of water, Standards of drinking water. Water Treatment : Objective of treatment, unit operations and processes. Treatment Flow sheet of conventional water treatment plant. Aeration: Purpose, types of aerators. Coagulation & Flocculation: Definition, Principals, types of coagulants Reactions, coagulant doses, types of mixing and flocculation devices. and

Types of Water: Pure Water: - 1. It is chemical compound (H 20), which contains two parts of hydrogen and one part of oxygen. 2. It is not good for health. 3. It is not possible to find absolutely pure water in nature water drops just emitting out of the clouds may be considered as chemically pure water. 4. Distilled water is the pure water. Potable Water: 1. The water have both the characteristics of ‘wholesome water’ and ‘palatable water’ is known as Potable water.

Wholesome Water: - 1. It is that water which is not chemically pure but does not contain anything harmful to human health. 2. It also have sufficient amount of necessary elements or minerals. Palatable Water: 1. Water which is aesthetically looking good and tasteful for drinking. Potable Water: 1. The water one which have sufficient amount of mineral, tasteful for drinking, aesthetically looking good and doesn’t contain anything harmful to human health is known as Potable water.

Contaminated Water: 1. It contains pathogenic bacteria and unfit for drinking. Polluted Water: - 1. It contains undesirable substances making it unfit for drinking and other domestic use. 2. The contaminated water is always polluted water but polluted water is not always contaminated water because contaminated water contents pathogens & Polluted water may or may not be contents pathogens.

Water Quality & Water Quality Standards Water quality is determined by assessing three attributes or parameter: Physical, Chemical & Biological. Water Quality Standards are the criteria which describes the parameter for drinking water. Standards are any definite measure or limited level of concentration established by regulatory authority for the purpose of protecting or preparing a water resources for one more uses. 1) United States (Federal government's) Environmental Protection Agency (EPA). 2) World Health Organization's Guideline for Drinking Water. (WHO) 3) Bureau of Indian Standards gives the specification for drinking water ( IS-10500) 4) American Water Work Association. 5) Central Pollution Control Board. 6) State Pollution Control Board. 7) Ministry of Urban Development. 8) Central Public Health & Environmental Engineering Organization (CPHEEO).

Water Analysis 1) Water analysis is the assessment or determination of quality of water, whether water is good for various domestic purpose or not. 2) Water analysis is the determination of various physical, chemical & biological characteristics of water. 3) Various types of impurities present in water & it can be determined by water analysis.

Water Borne Diseases: 1)Amoebiasis (hand-to-mouth) Protozoal infections 2) Cryptosporidiosis (oral) Protozoal infections 3) Cyclosporiasis 4) Giardiasis (fecal-oral) (hand-to-mouth) Parasite 5) Dracunculiasis (Guinea Worm Disease) Parasite 6) Cholera (bacteria) 7) Dysentery (bacteria) 8) Typhoid fever (bacteria) 9) Hepatitis A ( Virus infection) 10) Other: Polio, Fluorosis, Trachoma, Hepatitis, Diarrhea, Giardiasis, Ascariasis, Trichuriasis, Arsenicosis, Malnutrition, Legionellosis, Leptospirosis, Schistomiasis, Dracunculiasis, Onchocerciasis, Lead poisoning, Cryptosporiodiosis, Campylobacteriosis, Lymphatic filariasis, Hookworm infection, Ring Worm or Tinea, Methaemoglobinemia, Cyanobacterial toxins, Japanese encephalitis, etc.

Purpose of Water Analysis: 1) To classify the water with respect to general level of mineral constituents. 2) To determine the degree of clarity and ascertain the nature of matter in suspension. 3) To determine the chemical & bacteriological pollution of water. 4) To determine the presence constituents. or absence of an excess of any particular 5) To determine the level of organic impurities. 6) To ascertain whether purification of water has reached the required standards or not. 7) To decide treatment is necessary or not and if it is necessary then what extent of treatment needed.

Characteristics of Water: A. Physical Characteristics of water: 1) 2) 3) 4) 5) Color Taste Odor Temperature Turbidity B. Chemical Characteristics of water: 1) 2) 3) 4) 5) 6) 7) 8) 9) Total Solids Chloride p. H Conductivity Hardness Alkalinity Nitrogen Contents Metal & Other Chemical Substances( Iron, Manganese, Sulphate, Copper, etc. ) Dissolved Gasses (DO, BOD & COD) C. Biological Characteristics of water: 1) Micro-organism like aquatic plants, aquatic animals, bacteria, viruses, etc.
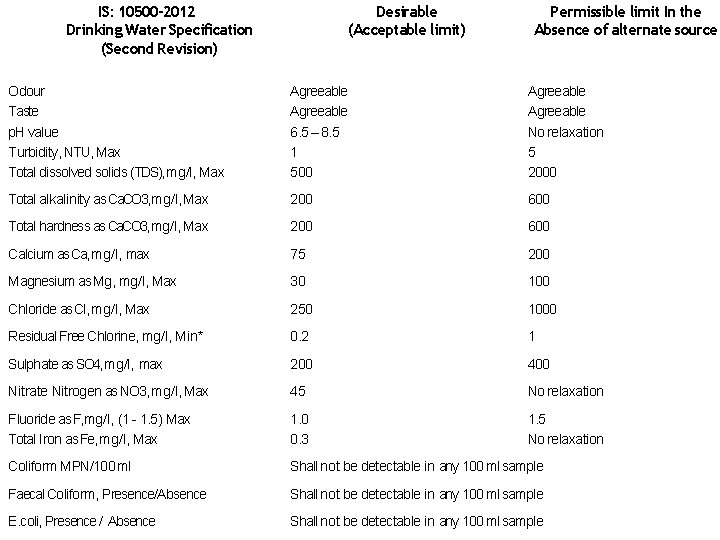
IS: 10500 -2012 Drinking Water Specification (Second Revision) Desirable (Acceptable limit) Permissible limit In the Absence of alternate source Odour Taste p. H value Turbidity, NTU, Max Total dissolved solids (TDS), mg/l, Max Agreeable 6. 5 – 8. 5 1 500 Agreeable No relaxation 5 2000 Total alkalinity as Ca. CO 3, mg/l, Max 200 600 Total hardness as Ca. CO 3, mg/l, Max 200 600 Calcium as Ca, mg/l, max 75 200 Magnesium as Mg, mg/l, Max 30 100 Chloride as Cl, mg/l, Max 250 1000 Residual Free Chlorine, mg/l, Min* 0. 2 1 Sulphate as SO 4, mg/l, max 200 400 Nitrate Nitrogen as NO 3, mg/l, Max 45 No relaxation Fluoride as F, mg/l, (1 - 1. 5) Max Total Iron as Fe, mg/l, Max 1. 0 0. 3 1. 5 No relaxation Coliform MPN/100 ml Shall not be detectable in any 100 ml sample Faecal Coliform, Presence/Absence Shall not be detectable in any 100 ml sample E. coli, Presence / Absence Shall not be detectable in any 100 ml sample

Turbidity: 1) Turbidity is a measure of how clear the water is. 2) It is the property of water which measures the resistance to passage of light through the water. 3) It is a measure of the degree to which the water looses its transparency due to the presence of suspended particulates. 4) It is imparted by the suspended, colloidal & dissolved matter present in water. 5) More turbidity more polluted water. 6) NTU: Nephelometric Turbidity Units. 7) Nephelometer, colorimeter or turbidimeter, which measures the intensity of light scattered at 90 degrees as a beam of light passes through a water sample.

Turbidity: 8) On olden days turbidity measured by Baylis Turbidimeter, Jackson Turbidimeter, Turbidity Rod, etc. 9) Jackson’s turbidimeter measures the influence to light passage in a straight line while nephelometer measure the scattering of light from particles 10) The more intensity of light scattered, more will be turbidity. 11) 1 NTU = 1 FTU (Formazin Turbidity Unit) 1 mg of formazin polymer can produce same 1 NTU turbidity if added in 1 liter distilled water. 12) Desirable 1 NTU and Permissible 5 NTU, according to IS-10500.

Color: 1) Color due to presence of organic matter, suspended matter, colloidal matter, etc. 2) It can be measured against various standard or scale such as Hazen, Platinum, Cobalt, Platinum Chloride scale using tintometer. 3) Color on platinum cobalt scale should not exceed 20 & preferably be less than 10 ppm color on platinum cobalt scale means 10 mg of platinum cobalt powder in 1 liter distilled water gives same kind of color. 4) Color, Hazen Units, IS 10500 -2012, Desirable : 5 Hz. , Permissible : 15 Hz. 5) If color is more, acceptance decreases. 6) Treatment: Filtration, Reverse osmosis, etc.

Taste & Odor: 1) It is due to dissolved organic matter, inorganic matter, dissolved gases, minerals, salts, microscopic organism chemicals compounds, etc. 2) Threshold Number is the standard unit for measuring taste and odor. 3) Taste is expressed as FTN ( Flavor Threshold Number). 4) Odor is expressed as TON ( Threshold Odor Number). 5) 1 to 3 is permissible. (pleasant) 6) As per IS-10500, it should be agreeable. Temperature: 1) The temperature of water should be between 100 c to 200 c. 2) Temperature higher than 250 c is considered objectionable.

p. H: 1) p. H value of water indicates the logarithm of reciprocal of hydrogen ion conc. Present in water. Lower H+ ions means high p. H. 2) It can also measure of degree of acidity or alkalinity of water. 3) p. H = -log 10 (H+) 4) Measured by potentiometer or colorimetric method.

Conductivity: - 1) It depends on dissolved salt content. 2) μ mho/cm (Specific Conductivity) 3) Measuring the conductivity is an accurate way to determine salinity. Freshwater = micro Siemens (u. S) Saltwater = milli Siemens (m. S)

Chloride : 1) Generally it present in the form Na. Cl. 2) It imparts salty taste. 3) Desirable = 250 mg/L & Permissible = 1000 mg/L 4) Presence of high conc. indicates pollution from sewage. 5) It can be measured by titrating the water with standard silver nitrate solution using potassium chromate (1 ML) as indicator (100 ML water sample). 6) Mohr Method (p. H range 6. 5 to 9) Ag++ Cl-→ Ag. Cl (white precipitate) 2 Ag+(excess) + Cr. O 2 - → Ag Cr. O 4 2 (brownish or red precipitate is endpoint) 4

Total Solids : 1) All matter except water containing liquid materials classified as solid matter. 2) Total solids as that matter which remains as a residue after drying & evaporation at 1050 C (+ - 50 C). 3) Suspended Solids can be found by filtering water sample through Whatman filter paper no. 44. 4) Material retained on the filter is dried & weighed.


Hardness : 1) It prevents the formation of sufficient lather or foam with soap. 2) Expressed in terms of mg/L of Ca. CO 3. 3) Temporary & Permanent hardness. 4) Temporary or carbonate hardness due to HCO 3 & CO 3 of Mg & Ca. (removed by boiling & lime) 5) Permanent or non-carbonate hardness by SO 4, Cl, NO 3, of Ca, Mg. (water softening methods like lime soda process, zeolite process, etc. ) 6) Versenate Method is used to determine the hardness by titrating against EDTA using EBT indicator. (Buffer + EBT + EDTA + wine red to sky blue) 7) Soft = 0 to 100, Moderate = 100 to 150, Hard = 150 to 300 & Very Hard = > 300

Alkalinity : 1) It is the acid neutralizing capacity of water. 2) It is caused by bicarbonate (HCO 3), hydroxide (OH), carbonate (CO 3) and very little -ve alkalinity is also caused by H+ 3) Total Alkalinity = HCO 3 + OH + CO 3 - H+ 4) 100 ML Water + 2 Drops Phenolphthalein Indicator + H 2 S 04 Till Colorless. 5) 2 Drops Mixed Indicator + H 2 S 04 Blue to Red. 6) Highly alkaline water is unpalatable. 7) Desirable = 200 mg/L & permissible = 600 mg/L in terms of Ca. CO 3.

Nitrogen Content : 1) The presence of nitrogen in water indicates presence of organic water. 2) The presence of the organic may occur in following form: 1) Free ammonia 2) Albuminoid or Organic nitrogen 3) Nitrites 4) Nitrates 3) Free ammonia indicates presence of undecomposed organic matter & represents first stage decomposition. (permissible limit < 0. 15 mg/L) 4) Albuminoid nitrogen indicates decomposition of organic matter has started, limit shall not exceed 0. 3 mg/L. 5) Nitrites indicates partly decomposed organic matter, permissible limit is NIL. 6) Nitrates indicates fully oxidised organic matter, permissible limit < 45 mg/L 7) Excessive conc. of nitrates can cause methaemoglobinaemia. 8) Sum of ammonia nitrogen & organic nitrogen is called kjedahl nitrogen. 9) Nitrates or nitrites present in water can be measured by color matching methods.

Metal & other chemical substances : 1) Iron (0. 3) & Manganese (0. 1 & 0. 3): High concentration is undesirable, as it may cause discoloration of cloth washed in such waters. They also cause incrustation in water mains due to deposition of ferric hydroxide and manganese oxide. 2) Fluoride: concentration less than 1 mg/L may cause dental caries like tooth decay. Concentration greater than 1. 5 mg/L may cause spotting & discoloration of teeth or fluorosis & deformation of bones. 3) Lead (0. 01), Copper (0. 05 & 1. 5) , Arsenic(0. 01), Selenium (0. 01)& barium are toxic therefore very low concentration is provided. 4) High conc. of Copper affect the human lungs adversely. 5) The most of the metals and other chemical substances present in water can be estimated by using color matching method by using different indicators.

Dissolved Gases : 1) The various gases may get dissolved in water due to its contact with the atmosphere or the ground surface. (nitrogen, methane, hydrogen sulphide, carbon dioxide & oxygen) 2) H 2 S even at small amounts may give bad taste and odor to water. 3) Presence of CO 2 indicates biological activity & imparts bad taste, odor & cause corrosion. 4) Dissolved Oxygen. 5) Biochemical Oxygen Demand.
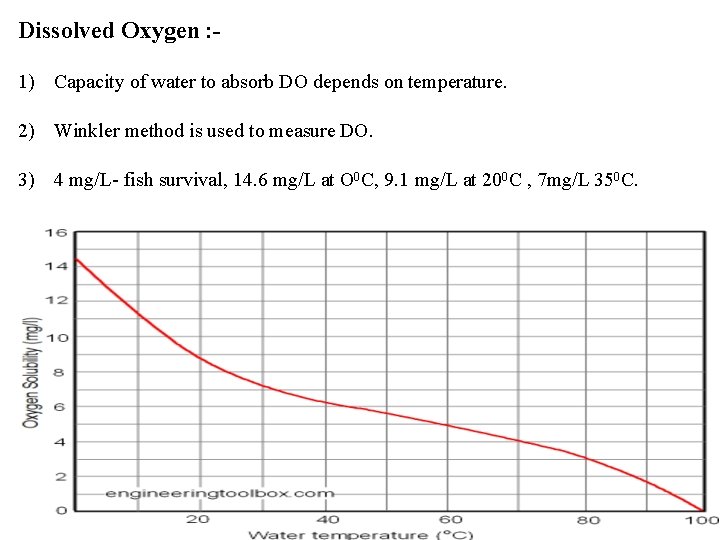
Dissolved Oxygen : 1) Capacity of water to absorb DO depends on temperature. 2) Winkler method is used to measure DO. 3) 4 mg/L- fish survival, 14. 6 mg/L at O 0 C, 9. 1 mg/L at 200 C , 7 mg/L 350 C.

BOD : 1) Amount of free oxygen required by micro-organism for stabilization of organic matter under aerobic condition at a specified temp. & time (200 C & 5 Days). 2) Theoretically infinite time required to complete biological oxidation, but all practical purpose, the reaction may be complete in 20 days. 3) However 20 days is to long period to wait for result & a large % of BOD is exerted in 5 days.

Biological Characteristics : 1) Pathogen is a bacteria, virus, or other microorganism that cancause disease. 2) It can be living things or non-living things. 3) Non-pathogenic bacteria are harmless ex E-coli. 4) E-coli is used as indicator & it should be Nil / 100 ml. 5) Measurement of E-coli by Total count Test, E-coli Test & Membrane filter. 6) The various micro-organism found in water are: 1) Aquatic plants ( algae) 2) Bacteria 3) Viruses 4) Protozoa

Water Treatment : 1) It is the removal impurities from the water, which are harmful the human health (Environment). 2) It is the process for enhancing the quality of water so that it meets water quality criteria for its fitness for particular purpose. 3) Water treatment is processes that makes water more acceptable for an end-use, which may be used for drinking, industry, or medicine.

Objectives of Water Treatment : 1) To make water fit for various industrial & domestic uses such as drinking, cooking, washing, etc. 2) To remove the unpleasant & objectionable color, odor & taste from the water. 3) To remove obnoxious gases from the water. 4) To kill all the pathogenic germs, which are harmful to human health. 5) To eliminate the tuberculating & corrosive properties of water which affects the conduits and pipes. 6) To achieve technical feasibility & economy. 7) To provide required quantity and quality of water to consumer. 8) To maintain the water quality as per requirements.

Water Treatments : Water treatment is a combination of physical, chemical & biological processes. These are three types of unit operation and unit processes. 1) Physical Unit Operation: in which physical forces are predominate for removal of impurities. (screening, equalization, mixing, flocculation, sedimentation, floatation, etc. ) 2) Chemical Unit Processes: in which removal of contamination are brought about by chemical activity. (chemical precipitation, disinfection, gas transfer, adsorption, etc. ) 3) Biological Unit Processes: : in which removal of contamination are brought about by biological activity. (Aerobic, Anaerobic & Aerobic-anaerobic Biological Unit processes)

Biological Unit Processes: in which removal of contamination are brought about by biological activity. (Aerobic , Anaerobic & Aerobic-anaerobic Biological Unit processes) 1) Aerobic Biological Unit Processes: removal of impurities or decomposition of organic matter in presence of oxygen (RSF, Trickling filter, aerated lagoons, etc) 2) Anaerobic Biological Unit Processes: removal impurities or decomposition of organic matter in absence of oxygen (anaerobic lagoons, etc) 3) Aerobic-Anaeerobic Biological Unit Processes: combination of above two (stabilization pond etc)

Methods of Water Treatment : 1) Screening 2) Aeration 3) Plain Sedimentation or Primary Sedimentation. 4) Coagulation & Flocculation 5) Secondary Sedimentation or Sedimentation with Coagulant (Clarifier, Clariflocculator) 6) Filtration 7) Disinfection 8) Softening etc.
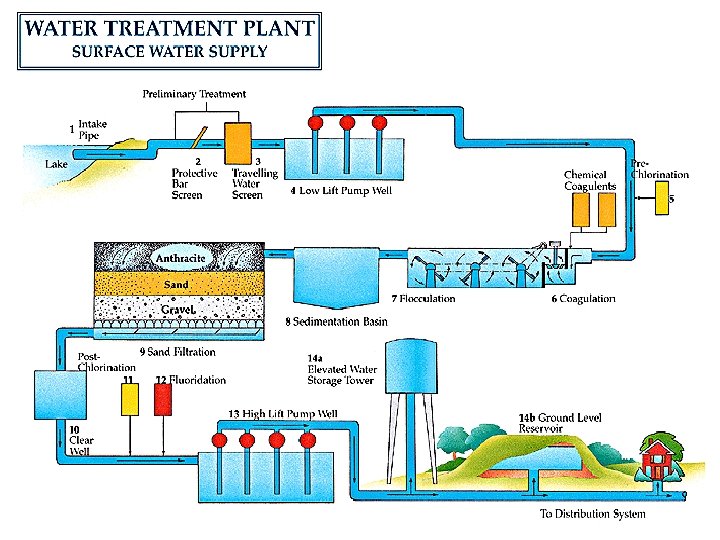

Treatment Units & Function : 1) Screens : To remove large size floating matter like debris, fish, vegetation, garbage, etc. from the raw water by protective bar screen. 2) Aeration: To add dissolved oxygen in water. 3) Plain sedimentation: To remove settleable suspended solids & other matter by passing water through large volume tanks where the flow speed slows down. 4) Coagulation: To Add coagulant in water. 5) Flocculation: To form floc in water. 6) Secondary sedimentation (Clarifier, Clariflocculator): water is applied to large volume tanks where the flow speed slows down and the suspended solids or floc are removed. 7) Filtration: To trap remaining impurities by physical straining. 8) Disinfection: To kill pathogens. 9) Softening: To remove hardness of water.

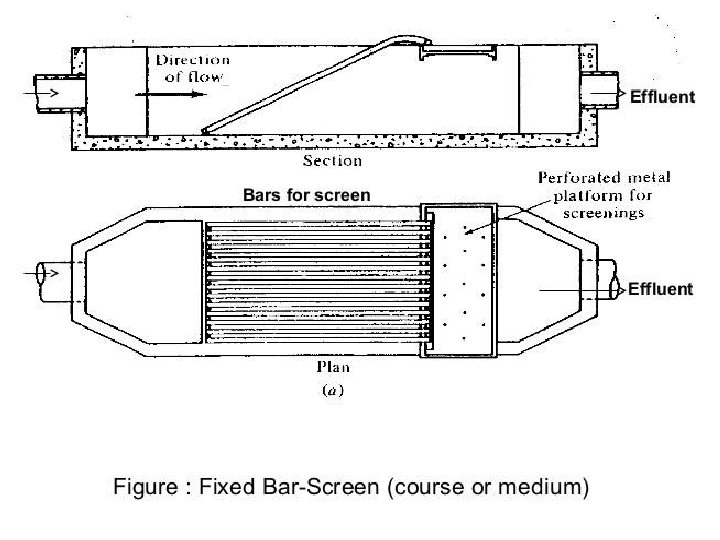
Screens : 1) Screening devices are the 1 st unit operation in treatment plant 2) To remove large size floating particles such as debris, animals, bushes, pieces of woods, plastics, leaves, aquatic plants, rags, solids, lumber, tree, branches, roots, etc. 3) It consists of parallel bars, rods or wire, grating, wire mesh or perforated plate, having opening of any shape but circular & rectangular are common. 4) The materials removed by these devices are termed as screening. 5) The screenings are usually no biodegradable materials & can be used for land filling purposes. 6) To prevent the pump, pipe, other equipment from getting damage or clogging. 7) To increase the efficiency of other treatment process.

Screens : 1) To prevent the pump, pipe, other equipment from getting damage or clogging. 2) To increase the efficiency of other treatment process. Types of Screens: 1) Fine Screens (Opening < 25 mm) 2) Medium Screens (Opening = 25 to 50 mm) 3) Coarse Screens (Opening = 50 to 150 mm)

Coarse Screens : 1) Are classified as either bar racks (trash racks) or bar screen. 2) It consists of parallel bars or rods of steel or stainless steel. 3) It can be used to entrap the coarse materials to protect the damaging of pumping units. 4) Bar racks are used as pretreatment applications. These bar racks may be hand cleaned if small in size or mechanical cleaned if large sizes. 5) To prevent the settling of coarse materials in the channel, the velocity in the approach channel must be in range of 0. 3 m/sec to 0. 6 m/sec. 6) Width of the bar 5 mm to 15 mm. 7) Allowable headloss 150 mm.


Aeration : 1) Aeration is the process of exchange of gases by creating good interface between liquid phase and gas phase. 2) It is the process of bringing water in intimate contact with air, while doing so the water absorbs oxygen from the air. 3) CO 2 can remove upto 60%. 4) Iron, Manganese. 5) H 2 S & other gases are also removed up-to certain extent from the water.

Objectives of Aeration : 1) To add the oxygen, to impart freshness to water. 2) To remove or decrease the CO 2 content of water & thereby raises its p. H value. 3) To remove H 2 S content of water & thereby removal of odor. 4) To remove CH 4 & taste caused due to organic decomposition. 5) To convert iron & manganese from their soluble states to their insoluble states, so that these can be precipitated & removed and thereby we can remove color, taste & odor. 6) To add oxygen, to oxidize organic matter & thereby removal of volatile substances.

Factors Affecting Aeration Process : 1) If the partial pressure is more, more will be the solubility & rate of exchange of gas & thereby more will be aeration. 2) If the temperature of water is less, less will be the solubility & less will be aeration. 3) If the concentration of impurities is more then solubility will be less & thereby aeration will be less. 4) If the surface area of interface is more, more will be the rate of exchange of gases & thereby aeration will be more. 5) If the thickness of interface is more, less will be the rate of exchange of gas & thereby aeration will be less. 6) If time of contact is more then more will be exchange of gas & thereby aeration will be more. 7) If degree of under saturation is more then fast will be rate of exchange & thereby aeration will be more.

Limitation of Aerations : 1) It is not an efficient process for removal of tastes & odors caused by relatively nonvolatile substances such as oils, algae & industrial wastewater. 2) Odor removal is 50% when symura bacteria is present. 3) Aeration may add more oxygen in water & making it more corrosive. 4) Fe & Mn can not be precipitated by aeration when organic matter is present. 5) Possibility of air borne contamination is there. 6) Additional lime may be required to neutralize the CO 2 that would be removed by aeration 7) Aeration is not economical in colder months or state or area.

Types of Aerators : 1) Spray Aerators. 2) Diffused Air Aerators. 3) Gravity Aerators: a) Cascade Aerator b) Multiple Tray Aerator c) Inclined Apron Aerator d) Slopping Tray Aerator.

Cascade Aerators : 1) Simplest free fall aerator. 2) Waterfalls & weirs of any kind are cascade aerator. 3) Water allowed to fall through a certain height of 1 to 3 m due to this water comes into close contact with air. 4) It consist of 3 to 9 steps of concrete or metal. 5) Rise of steps should be 20 to 50 cm.

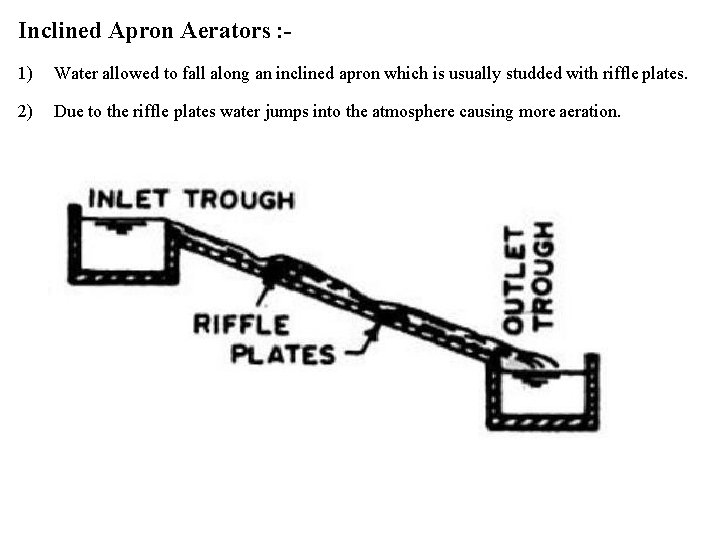
Inclined Apron Aerators : 1) Water allowed to fall along an inclined apron which is usually studded with riffle plates. 2) Due to the riffle plates water jumps into the atmosphere causing more aeration.
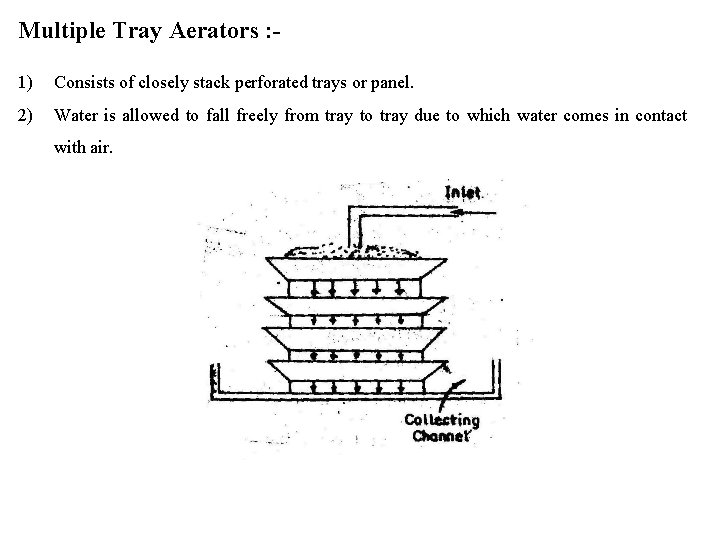
Multiple Tray Aerators : 1) Consists of closely stack perforated trays or panel. 2) Water is allowed to fall freely from tray to tray due to which water comes in contact with air.

Spray Aerators : 1) Spray aerator divide the water flow into fine streams & small droplets which come into intimates contact with the air in their trajectory. 2) Water is sprinkled into atmosphere in the form of fine spray or jets or droplets through nozzles. 3) It requires considerable head (0. 75 to 1. 5 Kg/cm 2) 2 to 9 m. 4) Reduces CO 2 by 70 % or more & H 2 S by 90% or more.
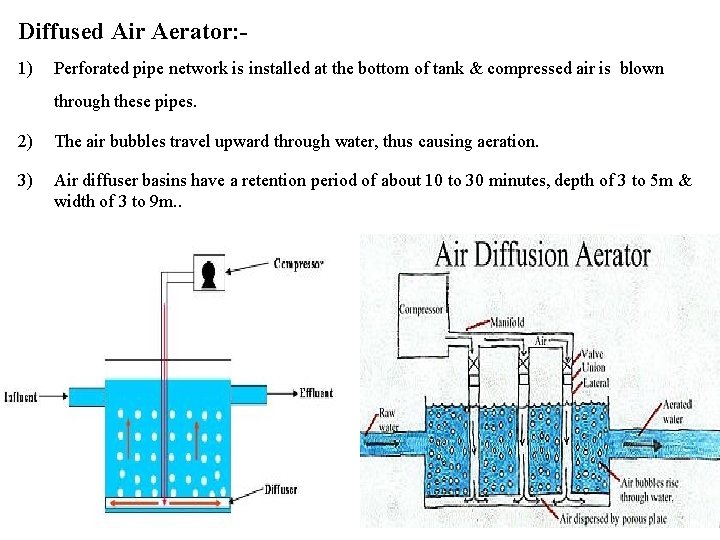
Diffused Air Aerator: 1) Perforated pipe network is installed at the bottom of tank & compressed air is blown through these pipes. 2) The air bubbles travel upward through water, thus causing aeration. 3) Air diffuser basins have a retention period of about 10 to 30 minutes, depth of 3 to 5 m & width of 3 to 9 m. .

Design Criteria for Design of Cascade Aerator: Area required for Design of Aerator = 0. 015 – 0. 045 m 2/m 3/hr. No. of cascade = 3 to 9. Height of aerator = 1 to 3 m. Rise of each step = 20 to 50 cm. Velocity of inlet pipe = 0. 3 to 0. 9 m/s

Design a Cascade Aerator for inflow 12 MLD. Assume other data suitably. A. Design of Inlet pipe: Flow = Q = 12 MLD = (12 x 106 x 10 -3)/ (24 x 60) = 0. 138 m 3/s Assuming velocity through pipe = 0. 6 m/s. Area of inlet pipe = (Q/v) = 0. 138 / 0. 6 = 0. 23 = (Π/4) dp 2 dp = Diameter of Inlet Pipe =0. 55 m B. Design of Aerator: Assuming Area required for Design of Aerator = 0. 030 m 2/m 3/hr. Q = 12 MLD = 500 m 3/hr. Area of Bottom Cascade aerator = 0. 03 x 500 = 15 m 2. Total area of bottom cascade aerator = Area of bottom aerator + Area of inlet pipe = 15 + ((Π/4)0. 552) = 15. 24 m 2.

B. Design of Aerator: Total area of bottom cascade aerator =15. 24 m 2 Assuming no. of steps = 5 Diameter of bottom Cascade: 2 2 (Π/4)d 5 = 15. 24 m d 5 = 4. 4 m Assuming Height of Aerator = 2 m Rise of each step 2/5 = 0. 4 m Diameter of 4 th Cascade Aerator : (4. 4 / 2) = d 4 / (2 - 0. 4) d 4 = 3. 52 m Diameter of 3 rd Cascade Aerator : (4. 4 / 2) = d 3 / (2 - 0. 8) d 3 = 2. 64 m

B. Design of Aerator: Diameter of 2 nd Cascade Aerator : (4. 4 / 2) = d 4 / (2 – 1. 2) d 2 = 1. 76 m Diameter of 1 st Cascade Aerator : (4. 4 / 2) = d 3 / (2 – 1. 6) d 1 = 0. 88 m C. Design of Collecting Channel: Diameter of Collecting Channel = 4. 4 + 1 =6. 4 m q = Q / 2 = 1. 375 b x h (3/2) q = discharge through channel in m 3/s b = width of channel in m h = height of channel 0. 138 / 2 = 1. 375 x 1 x h (3/2) h = 0. 14 m

Coagulation: 1) Process of mixing or adding certain chemicals to form insoluble precipitate for absorbing suspended & colloidal matter 2) Certain chemicals is called Coagulant. Ex. Alum 3) Finely divided impurities not removed in plain sedimentation without longer detention periods 4) To Remove turbidity up to 20 NTU & bacterial load by 70% 5) Adopted when turbidity more than 40 NTU 6) Coagulant produces thick gelatinous precipitate & has the property of arresting & attracting the suspended matter 7) Floc posses +ve charge & impurities has – ve charge

Why Coagulation : 1) Even after sedimentation, some finely divided particles such as clay and coloring matter are held in suspension. 2) These are removed only if their size is increased by using some chemical compounds. 3) In coagulation, coagulants are added and particles are made to settle down by neutralization.

Factors Affecting Coagulation: 1) Dose of coagulant 2) Type of coagulant 3) Form of coagulant 4) Type of mixing 5) Type of feeding 6) p. H value 7) Temperature 8) Impurities 9) Time

Types of coagulant: 1) Alum, Al 2(SO 4)3. 18 H 2 O, Aluminium Sulphate 2) Copperas Fe. SO 47 H 2 O, Ferrous Sulphate 3) Chlorinated Copperas, Fe(SO 4 ) 3 + Fe. Cl 3 4) Sodium Aluminate, Na 2 Al 2 O 4 5) Magnesium Carbonate, Mg. CO 3

Alum: 1) Widely used due to easy availability and minimum cost 2) Recovery is possible and recovery cost is just ¼ th of cost of alum 3) Dose of alum = 10 to 30 mg/l 4) Effective when p. H = 6. 5 to 8. 5 5) Requires presence of alkalinity in water 6) Many water have bicarbonate alkalinity naturally Al 2(SO 4)3. 18 H 2 O + 3 Ca(HCO 3)2 = 2 Al(OH)3 + 3 Ca. SO 4 + 6 CO 2 + 18 H 2 O Above reaction indicate that 1 mg of Al 3+, reacting with 5. 56 mg of alkalinity as Ca. CO 3, Form 2. 89 mg of aluminium hydroxide flocs. If insufficient alkalinity in water then lime will be added in water then reaction will be Al 2(SO 4)3. 18 H 2 O + 3 Ca(OH)2 = 2 Al(OH)3 + Ca. SO 4 + 18 H 2 O Sometimes soda also added into water Al 2(SO 4)3. 18 H 2 O + 3 Na. CO 2 = 2 Al(OH)3 + 3 Na. SO 4 + 3 CO 2 + 18 H 2 O Soda ash doesn’t cause hardness but expansive than lime

Advantages of Alum: 1) Reduces taste and odor in addition to turbidity 2) Cheap and easily available 3) Simple in working 4) Doesn’t required skilled labor 5) Crystal clear water 6) Heavy and better floc 7) Quite tough to broken down

Disadvantages of Alum: 1) Difficult to dewater the sludge 2) Sludge is not easy to dispose off 3) Imparts permanent hardness Ca. SO 4 4) Produces CO 2 and can cause corrosion 5) Effective p. H range is very small (6. 5 to 8. 5)

Iron compounds as a coagulant: 1) Ferrous Sulphate, Ferric Chloride and Ferric Sulphate 2) Ferrous sulphate is known as Copperas used as coagulant in conjunction with lime. Fe. SO 4. 7 H 2 O + Ca (HCO 3)2 Fe (HCO 3)2 + 2 Ca (OH)2 = = Fe (HCO 3)2 + Ca. SO 4 + 7 H 2 O Fe (OH)2 + 2 Ca. CO 3 + 2 H 2 O 1) Further Fe (OH)2 Ferrous Hydroxide is converted to Fe (OH)3 Ferric Hydroxide. 2) Dose of Iron salt depend upon on turbidity, natural alkalinity & CO 2 in water. 3) Because of addition of lime, color of water may be permanently set. 4) Moreover iron salt are less expensive than alum but skilled supervision is required.

Advantages of Iron compounds as a coagulant: 1) Floc produced is heavy and readily settles under favorable conditions. 2) They are more efficient & requires less time for reaction. 3) Active over a p. H range of 3. 8 to 10. 4) Cost of treatment is usually low. 5) Widely used for industrial purpose but less often for potable. 6) Removes H 2 S, taste, odor etc

Disadvantages of Iron salt as coagulant: 1) Iron salt causes staining. 2) Iron salt are difficult to store and handle. 3) Lime is required to be added which increases cost of treatment. 4) Promotes growth of iron bacteria & adversely affect the RSF process. 5) Imparts more corrosiveness than alum. 6) To maintain proper dosage skilled labor is required. 7) Ferrous fluoride introduces fluorine in supply which can cause ‘mottled teeth. 8) By Addition of lime, the color of water may be permanently set.

Coagulants Aids: Certain chemicals added to increase rate of reaction, to reduce the coagulant dose, to extend the optimum p. H range, to produce faster and tougher flocs. 1) Activated silica. 2) Bentonite clay. 3) Lime. 4) Soda ash. 5) Polyelectrolyte.

Jar Test : Consists of jar test assembly. Used to determine optimum coagulant dose. Sample (1 L) has to placed into number of beaker. Various amounts of dose in each beaker is added. Paddle speed = 100 rpm for 1 minute then make it slow for next 10 to 25 minutes with 40 rpm. After fast & slow mixing allow it to settle down for 30 minutes. Smallest dose that produces a more floc is optimum coagulant dose. This is trial and error method. One factor is varying and others are constant.

Jar test

Feeding Methods for Coagulant: 1) There are two methods: Dry feeding and Wet feeding. 2) Choice between dry & wet feeding depends on characterization of coagulants, convenience of its application, dosage of coagulant and size of plant. 3) Alum being uniform in grain size and not affected by atmospheric moisture can be fed by dry feeding. 4) Iron salt can not be fed by dry feeding due to hygroscopic property. 5) If dose of coagulant is small and accuracy is needed then coagulant must be fed in a solution form. 6) Generally for large treatment plant wet feeding is adopted and for small treatment plant dry feeding is adopted because of more cost of wet feeding device.
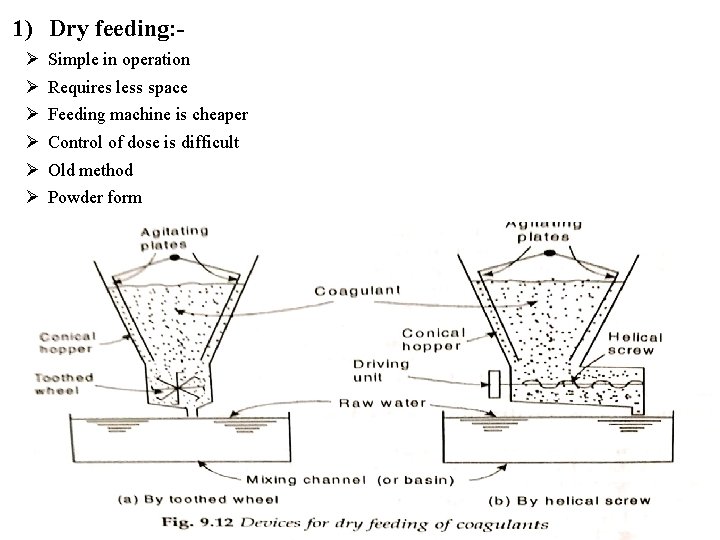
1) Dry feeding: Simple in operation Requires less space Feeding machine is cheaper Control of dose is difficult Old method Powder form
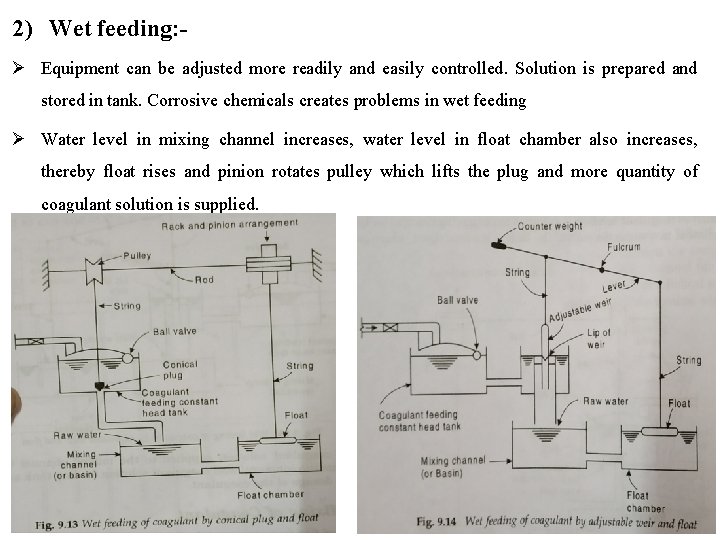
2) Wet feeding: Equipment can be adjusted more readily and easily controlled. Solution is prepared and stored in tank. Corrosive chemicals creates problems in wet feeding Water level in mixing channel increases, water level in float chamber also increases, thereby float rises and pinion rotates pulley which lifts the plug and more quantity of coagulant solution is supplied.

Mixing Devices: Floc formation depends on the mixing of coagulant. Point of turbulence. Methods of mixing: 1. Centrifugal pump 2. Compressed air agitation 3. Narrow mixing channel with flume 4. Mixing basin with baffle wall 5. Mixing with mechanical devices

1) Centrifugal pump Used to raise the water to settling tank Dose can be added to the suction pipe Water with coagulant passes through the impeller of the pump, mixing by agitation 2) Compressed air agitation: Water with coagulant is agitated by compressed air from bottom

3) Narrow mixing channel with flume (Hydraulic jump) Coagulant fed from feeding tank. Turbulence caused by vertical baffle mixes the chemical. Flume provided to develop the hydraulic jump for turbulence.
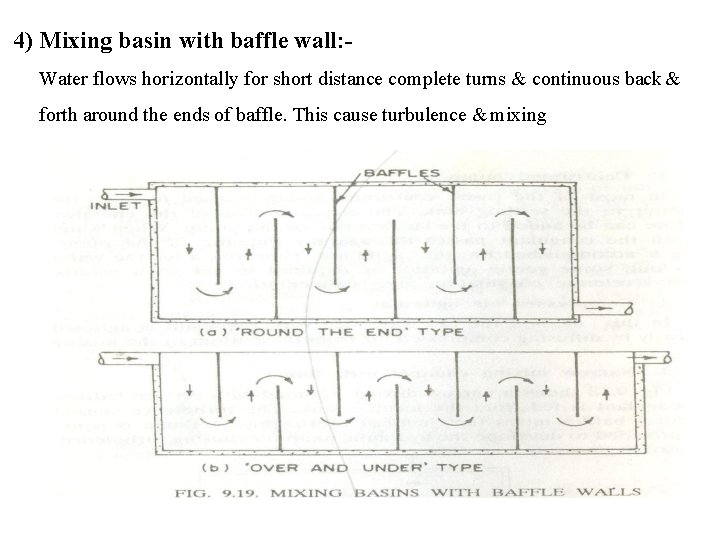
4) Mixing basin with baffle wall: Water flows horizontally for short distance complete turns & continuous back & forth around the ends of baffle. This cause turbulence & mixing

5) Mixing with mechanical device: 1) Most of WTP have mechanical devices ie Flash Mixer 2) Coagulant agitated vigorously by paddle 3) Propeller type impeller in flash mixer with speed of 100 to 1400 rpm 4) Detention time , t = 30 to 60 sec. 5) Velocity gradient, G = 300 to 600 sec-1 6) Power requirements = 1 to 3 watts per m 3/hr
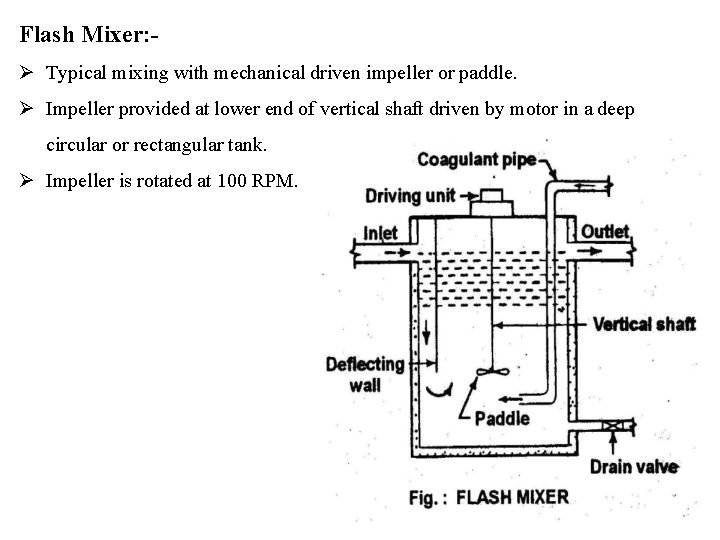
Flash Mixer: Typical mixing with mechanical driven impeller or paddle. Impeller provided at lower end of vertical shaft driven by motor in a deep circular or rectangular tank. Impeller is rotated at 100 RPM.

Flocculation: The process of floc formation After mixing, slow and gentle stirring is permitted to agglomerate the floc.

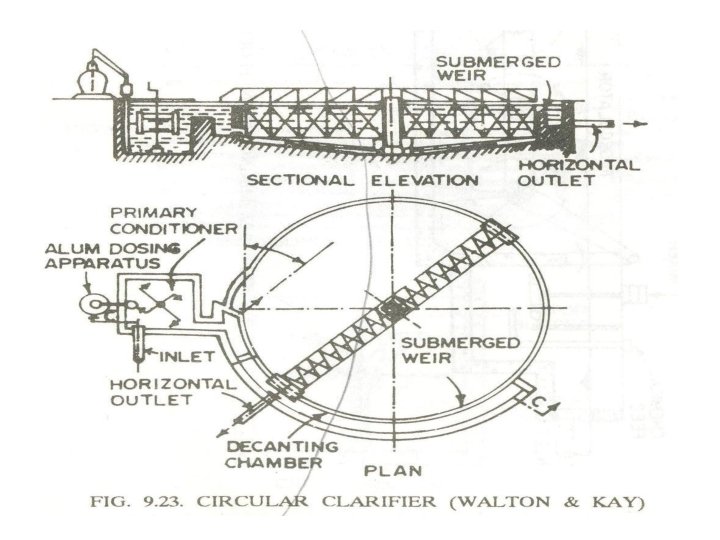
Clariflocculator or clarifier: 1) Consists of feeding devices, mixing devices, flocculator & settling basin. 2) Coagulant fed through feeding devices 3) Mixed and agitated in the mixing basin 4) Allowed to agglomerate in flocculation tank 5) Flocculated water passed into sedimentation tank 6) Clarified water taken out through outlet to RSF 7) Removes turbidity up to 20 NTU and 70 % of bacterial load




Design criteria for flash mixer: 1) Temporal mean velocity gradient, G = 300 to 600 sec. -1 2) Power requirements = 1 to 3 watts per m 3 per hour 3) RPM of Impeller = 100 to 1400 rpm 4) Ratio of tank dia. To impeller dia. = 5: 1 to 3: 1 5) Tangential velocity of impeller = 3 m/s at blade tip 6) Ratio of tank ht to tank dia. = 1: 1 to 3: 1 7) Product, G. t =18000 8) G = (P/u. V)1/2

Design Criteria For Flocculator : 1) Temporal mean velocity gradient, G = 10 to 75 sec. -1 2) Power requirements = 10 to 36 Killo watts per MLD 3) RPM of Impeller = 4. 5 rpm 4) Velocity of blades = 0. 2 to 0. 6 m/s 5) Velocity of flow = 0. 2 to 0. 8 m/s 6) Outlet flow velocity = 0. 15 to 0. 25 m/s 7) Ratio of tank ht to tank dia. = 1: 1 to 3: 1 8) Product, G. t = 10000 to 100000 9) G = (P/u. V)1/2 10) Area of paddles = 10 to 25 % of c/s area of tank 11) Velocity ratio, k = 0. 25 12) Kinematic viscosity, u= 1. 0087 x 10 -3 at 200 c 13) Density, ρ = 998 kg/m 3 at 200 c 14) Dept of tank = 3 to 4. 5 m
- Slides: 86