Environmental Engineering Lecture Note Week 4 Heterogeneous Transformation






















- Slides: 37

Environmental Engineering Lecture Note Week 4 (Heterogeneous Transformation) Joonhong Park Yonsei CEE Department 2016. 3. 24 CEE 3330 Y 2013 WEEK 3

Environ. Eng. Week 4 3. B. Phase Changes and Partitioning -Types of heterogeneous transformation -Vapor pressure -Dissolution of species in water -Sorption

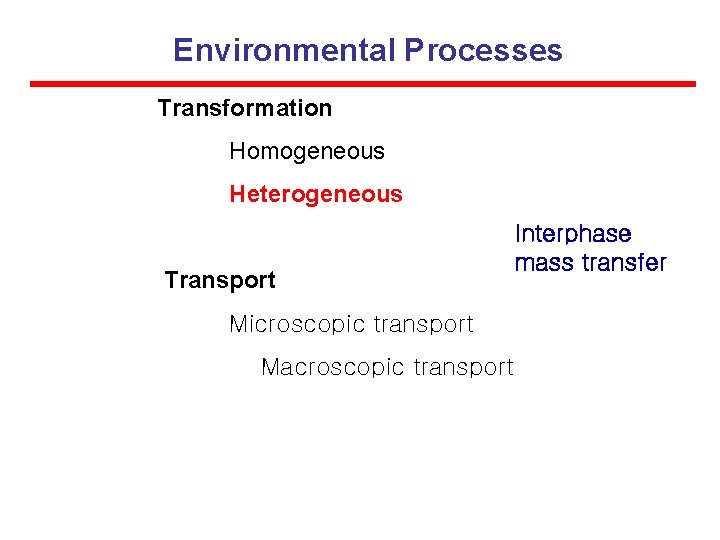
Environmental Processes Transformation Homogeneous Heterogeneous Transport Microscopic transport Macroscopic transport Interphase mass transfer

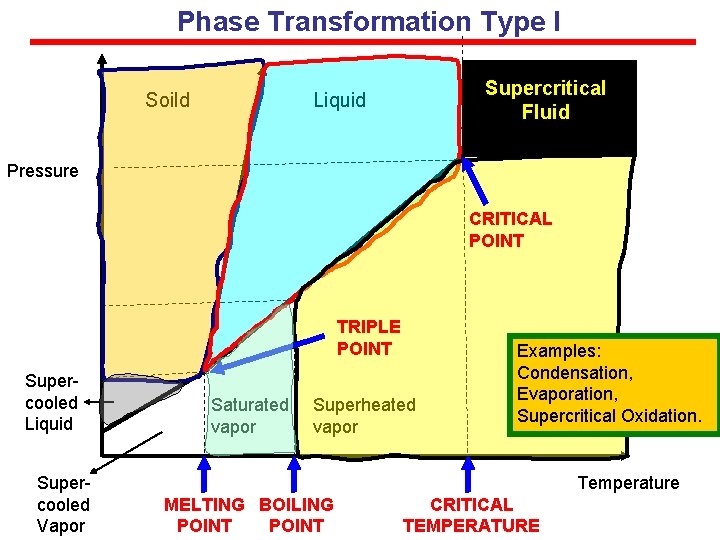
Phase Transformation Type I Soild Supercritical Fluid Liquid Pressure CRITICAL POINT TRIPLE POINT Supercooled Liquid Supercooled Vapor Saturated vapor Superheated vapor Examples: Condensation, Evaporation, Supercritical Oxidation. Temperature MELTING BOILING POINT CRITICAL TEMPERATURE

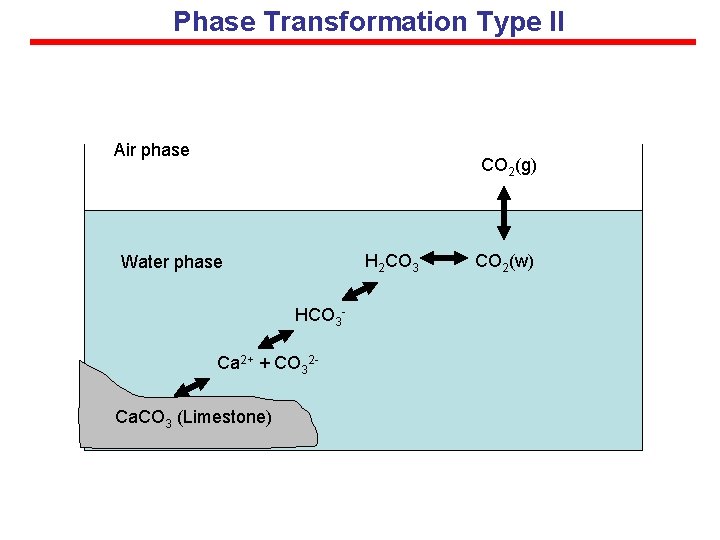
Phase Transformation Type II Air phase CO 2(g) H 2 CO 3 Water phase HCO 3 Ca 2+ + CO 32 Ca. CO 3 (Limestone) CO 2(w)

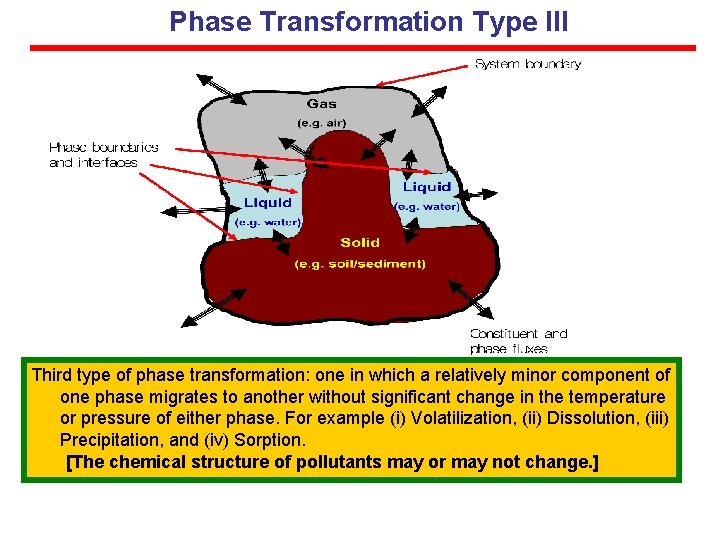
Phase Transformation Type III Third type of phase transformation: one in which a relatively minor component of one phase migrates to another without significant change in the temperature or pressure of either phase. For example (i) Volatilization, (ii) Dissolution, (iii) Precipitation, and (iv) Sorption. [The chemical structure of pollutants may or may not change. ]

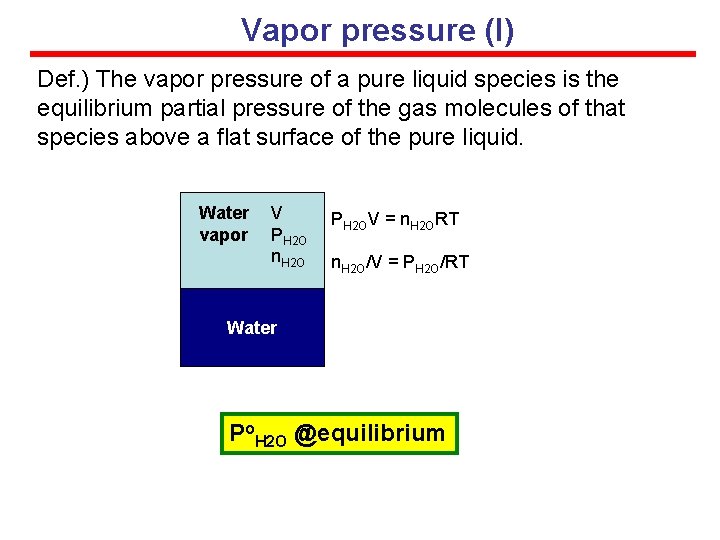
Vapor pressure (I) Def. ) The vapor pressure of a pure liquid species is the equilibrium partial pressure of the gas molecules of that species above a flat surface of the pure liquid. Water vapor V PH 2 O n. H 2 O PH 2 OV = n. H 2 ORT n. H 2 O/V = PH 2 O/RT Water Po. H 2 O @equilibrium

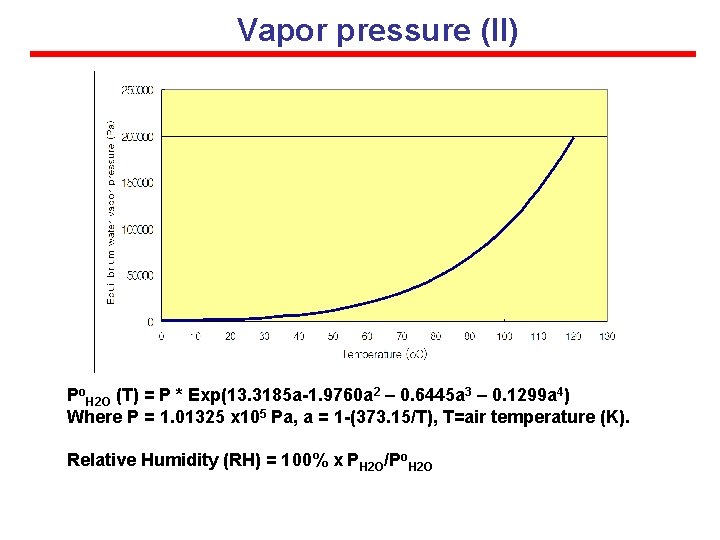
Vapor pressure (II) PH 2 OV = n. H 2 ORT n. H 2 O/V = PH 2 O/RT Po. H 2 O (T) = P * Exp(13. 3185 a-1. 9760 a 2 – 0. 6445 a 3 – 0. 1299 a 4) Where P = 1. 01325 x 105 Pa, a = 1 -(373. 15/T), T=air temperature (K). Relative Humidity (RH) = 100% x PH 2 O/Po. H 2 O

Vapor pressure (III) Poi (T) = function of chemical structure Poi : the equilibrium pressure of species i in its pure liquid or solid Poi, mix (T) = Xi Pomix (T) Where Xi: the mole fraction of the volatile component in the mixture. (or partial P) Poi, mix: equilibrium vapor pressure of species i in a mixture. Pomix: equilibrium vapor pressure of the mixture. Example: PO 2, air = 0. 21 * 1 atm = 0. 21 atm

Dissolution in liquid phases Species i (Pi, pi = Xi, Ci) Species i (Ci, Xi)) Air phase Water phase

Dissolution of Species in Water Partitioning between Gas Phase and Water: Henry’s Law Expression for Henry’s law Definition of terms Units for Henry’s Constant Pi =K’H, i Xi Pi: vapor pressure of species i in gas phase Xi: mole fraction of species i in water phase atm (per mole fraction) Pi =KH, i C Pi: vapor pressure of species i in gas phase Ci: molar concentration of species i in water phase atm-m 3/mol pi: partial pressure of species i in gas phase (Pi/Ptotal) Ci: mole fraction of species i in water phase Dimensionless KOH, i = K’H, I/Ptotal Ci, v: molar concentration of species i in gas phase Ci, a: mole concentration of species i in water phase Dimensionless KOH, i = KH, I/RT i, w pi =KO’H, i X i Ci, v =KOH, i C i, w

Dissolution of Species in Water Partitioning between Gas Phase and Water: Henry’s Law Example: Saturation concentration of oxygen in water Compute the equilibrium mass concentration of oxygen (O 2) in units of mg/L for water exposed to the atmosphere at sea level at a temperature of 15 o. C. (At 15 o. C, KH, O 2 = 654 atm/M, partial pressure O 2 in air = 0. 209 ) Solution: Since P = 1 atm at sea level, PO 2 =0. 209 * 1 atm PO 2 = KH, O 2 * CO 2, w = PO 2/KH, O 2 = 0. 209 (atm)/654 (atm/M) = 0. 000320 M Mass Concentration = Molarity * Oxygen MW = 0. 000320 (M) * 32(g/mol) * 1000(mg/g) = 10. 2 mg/L

Dissolution of Species in Water Partitioning between Gas Phase and Water: Henry’s Law Example: Partitioning of toluene in a closed system A 2 L glass jar is half filled with water and half filled with air at a temperature of 293 K. After 92 mg (=10 -3 mol) of liquid toluene is added, the jar is sealed. (1) What is the equilibrium concentration of toluene in the water? And (2) what is the equilibrium partial pressure of toluene in the gas phase? (KH for toluene, KH, tol, @ 20 o. C = 6. 7 atm/M) Solution: Henry’s law Ptol = KH, tol * Ctol, w => Ctol, w = Ptol/KH, tol Ideal gas law Ptol * Vg = n. RT => Ctol, g = (n/Vg) = Ptot/RT Material balance: Ctol, w * Vw + Ctol, g * Vg = Total amount of toluene (Ptot /KH, tol)* Vw + Ptol* Vg/(RT) = 0. 0010 mol Ptot = 0. 0052 atm => Ctot, g = 0. 0052/(0. 0821*293) =0. 00022 M C tot, w = 0. 00078 M

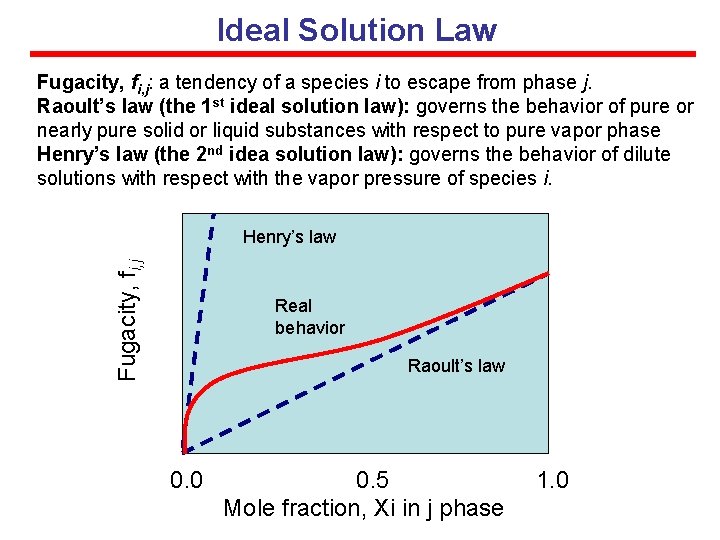
Ideal Solution Law Fugacity, fi, j: a tendency of a species i to escape from phase j. Raoult’s law (the 1 st ideal solution law): governs the behavior of pure or nearly pure solid or liquid substances with respect to pure vapor phase Henry’s law (the 2 nd idea solution law): governs the behavior of dilute solutions with respect with the vapor pressure of species i. Fugacity, fi, j Henry’s law Real behavior Raoult’s law 0. 0 0. 5 Mole fraction, Xi in j phase 1. 0

Dissolution of Species in Water Solubility of Nonaqueous-Phase Liquids (NAPLs) NAPL: liquids that do not mix with water (e. g. spills of petroleum products or organic solvents) Aqueous solubility @ a temperature, Cs. Xylene phase Xylene Distribution of Xylene Cs Water phase Raoult’s law

Dissolution of Species in Water When a small amount of xylene was spilled, no free phase exists. Xylene-air, Water-air Air phase Xylene-water, Air-water Water phase Fugacity of Xylene from air Henry’s law Fugacity of H 2 O from water Raoult’s law When a large amount of xylene was spilled, a free phase exists. Xylene-air = Po, Water-air Air-xylene, Water-xylene Air phase Xylene-water = Cs, Air-water Water phase Distribution of Xylene Raoult’s law (Non-Aqueous Phase Liquids [NAPLs])

Dissolution of Species in Water Solubility of Nonaqueous-Phase Liquids (NAPLs) Example: Dissolution of a NAPL (a) 1 L of liquid toluene (density =87 g/cm 3) is spilled into 1 m 3 of water. Assuming no loss of toluene or water from the system, what is the steadystate concentration of toluene dissolved in the water? (b) Repeat (a) assuming that the water volume is 10 m 3. [Hint: Cs of toluene = 530 mg/L] Solution: (a) [1000 cm 3 of toluene * 0. 87 g/cm 3]/[1000 L of water] = 870 mg/L > Cs The toluene concentration in water cannot be higher than Cs. Therefore, toluene concentration in water should be Cs. Residual NAPL remains. (b) 870 g/10, 000 L = 87 mg/L < Cs No NAPL is left.

Dissolution of Species in Water Dissolution and Precipitation of Solids Ax. By (solid) x. A + y. B Solubility product: Ksp = [A]x[B]y Ca 2+ + 2 FXylene Ca. F 2 (solid) Ksp =3 x 10 -11 Water phase M 3 Water phase At equilibrium, what is the conc. of F- in water? [Ca 2+] = 0. 5[F-] [Ca 2+]*[F-]2 = Ksp 0. 5[F-]3 = Ksp

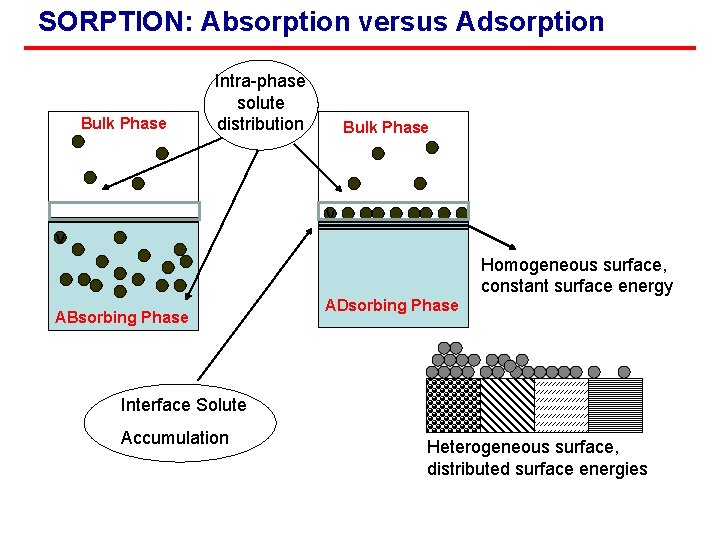
SORPTION: Absorption versus Adsorption Bulk Phase Intra-phase solute distribution Bulk Phase v v Homogeneous surface, constant surface energy ABsorbing Phase ADsorbing Phase Interface Solute Accumulation Heterogeneous surface, distributed surface energies

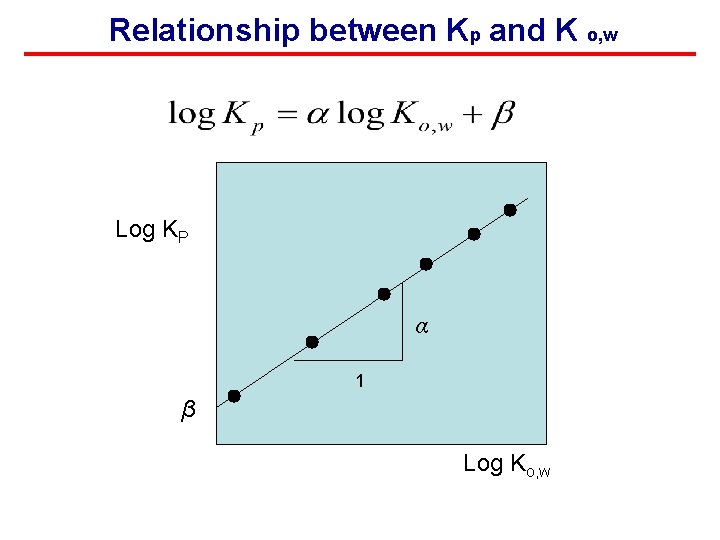
ABsorption: Partitioning Coefficient KP: partitioning coefficient i: compound i phase 1 phase 2 Octanol-water partition coefficient (often used as a measure of “hydrophobicity”) Empirical constants

Relationship between Kp and K o, w Log KP α 1 β Log Ko, w

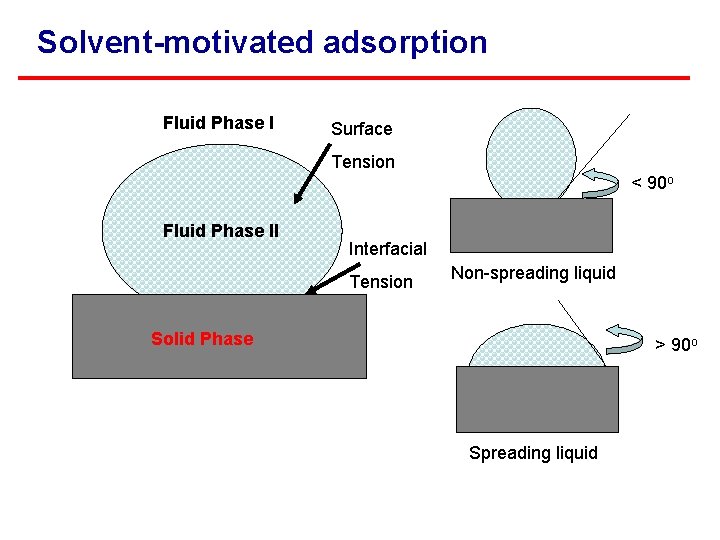
Adsorption Definition: the accumulation of dissolved substances at interfaces of and between phases. Solvent-motivated adsorption: surface tension and interfacial tension. Sorbent-motivated adsorption: - Chemical adsorption - Electrostatic adsorption - Physical adsorption

Solvent-motivated adsorption Fluid Phase I Surface Tension < 90 o Fluid Phase II Interfacial Tension Non-spreading liquid Solid Phase > 90 o Spreading liquid

Sorbent-motivated adsorption Chemical (100 -400 k. J/mol) -covalent -coordinate covalent Electrostatic (200 k. J/mol) - Ion-Ion-Dipole Physical (5 -10 k. J/mol): result from van der Waals forces -Dipole-Induced Dipole -Instantaneous Induced Dipoles (London dispersion)

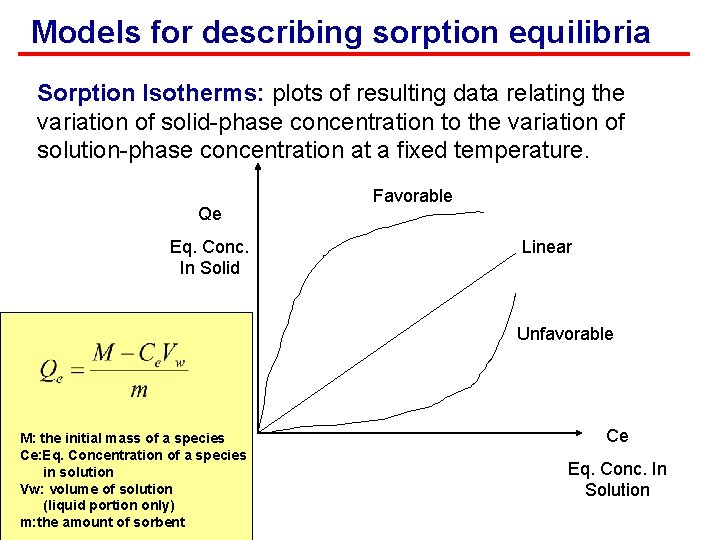
Models for describing sorption equilibria Sorption Isotherms: plots of resulting data relating the variation of solid-phase concentration to the variation of solution-phase concentration at a fixed temperature. Qe Eq. Conc. In Solid Favorable Linear Unfavorable M: the initial mass of a species Ce: Eq. Concentration of a species in solution Vw: volume of solution (liquid portion only) m: the amount of sorbent Ce Eq. Conc. In Solution

Models for describing sorption equilibria Linear Model Distribution coefficient. Qe Equil. Conc. Mass fraction of organic carbon. in Solid. Empirical coefficients. Ce Equil. Conc. in Water.

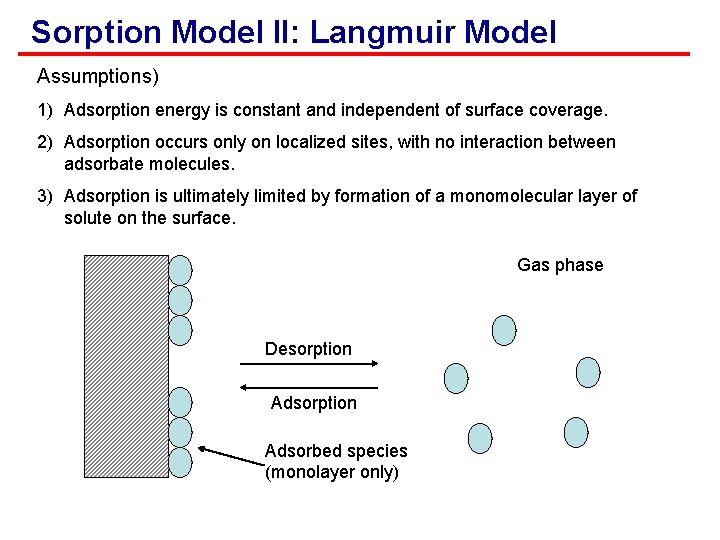
Sorption Model II: Langmuir Model Assumptions) 1) Adsorption energy is constant and independent of surface coverage. 2) Adsorption occurs only on localized sites, with no interaction between adsorbate molecules. 3) Adsorption is ultimately limited by formation of a monomolecular layer of solute on the surface. Gas phase Desorption Adsorbed species (monolayer only)

Sorption Model II: Langmuir Model (continued) Rate of desorption Rate of adsorption desorption rate constant fraction of complete monolayer coverage of the surface by adsorbed molecules at any time. adsorption rate constant partial pressure of the compound in gas

Sorption Model II: Langmuir Model (continued) At equilibrium Here

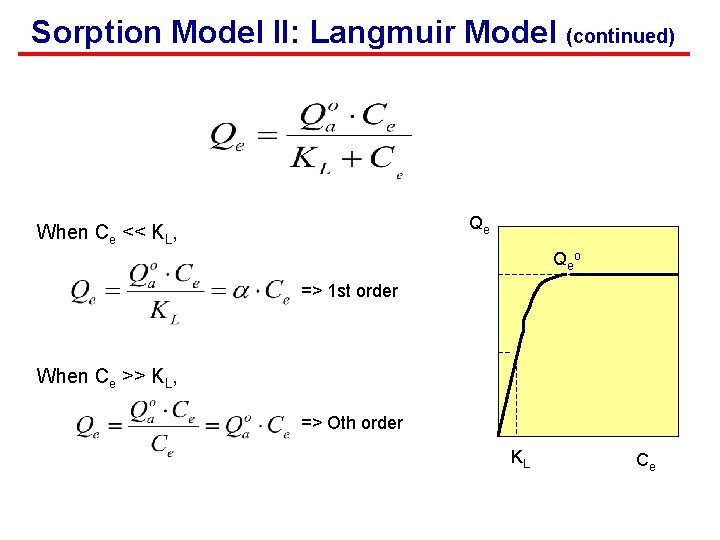
Sorption Model II: Langmuir Model (continued) For solid-water system Qe Qe o Max. sorption capacity 0. 5*Qeo Langmuir model (half saturation constant) KL Ce

Sorption Model II: Langmuir Model (continued) Qe When Ce << KL, Qe o => 1 st order When Ce >> KL, => Oth order KL Ce

Sorption Model III: Freundlich Model • Sorption of solute in water-soil system is not well described by theoretical models such as linear and Langmuir. • Heinrich Freundlich and other investigators suggested that data for solutions are frequently best described by a general exponential concentration-dependent relationship of the form 0< n <1 Qe n=1 n >1

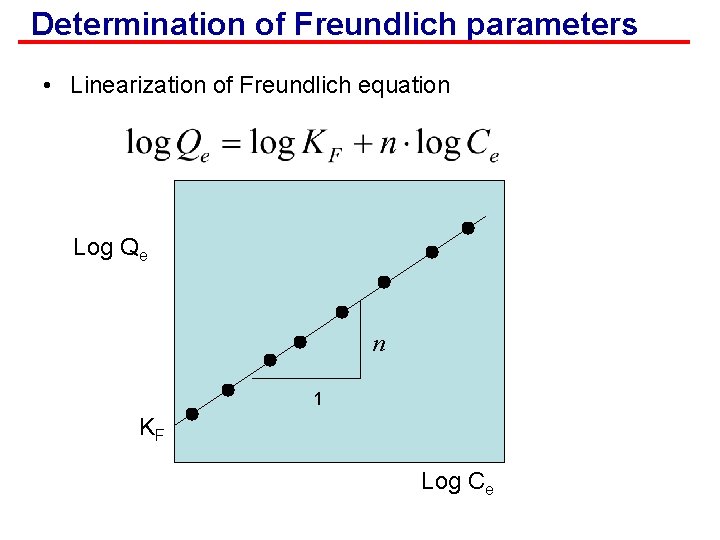
Determination of Freundlich parameters • Linearization of Freundlich equation Log Qe n 1 KF Log Ce

Multiple phase distribution of a species Example: 10 -3 mol of toluene was spilled in a 2 L sealed jar, half filled with water and half filled with air. The equilibrium partitioning was described by Henry’s law, so that the aqueous phase contained 0. 00078 M and 0. 00022 M. To this system, 200 mg of activated carbon was added. Toluene partitioning between the sorbed and aqueous phases was described by the Freundlich isotherm Qe =100 Cw where Ce is the aqueous concentration in mg/L and Qe is the sorbed mass concentration in mg-toluene per g-activated carbon. What is the new equilibrium concentration of toluene in the water? How much of activated carbon is needed to reduce Cw into 0. 000008 M? Air phase 1 L Water phase 1 L Total amt of toluene = Qe*(amt of a. c. ) + Cw*Vw +Cg*Vg = KF * Cw * (amt of a. c. ) + Cw*Vw + KHO*Cw*Vg 200 mg of activated carbon (a. c. )

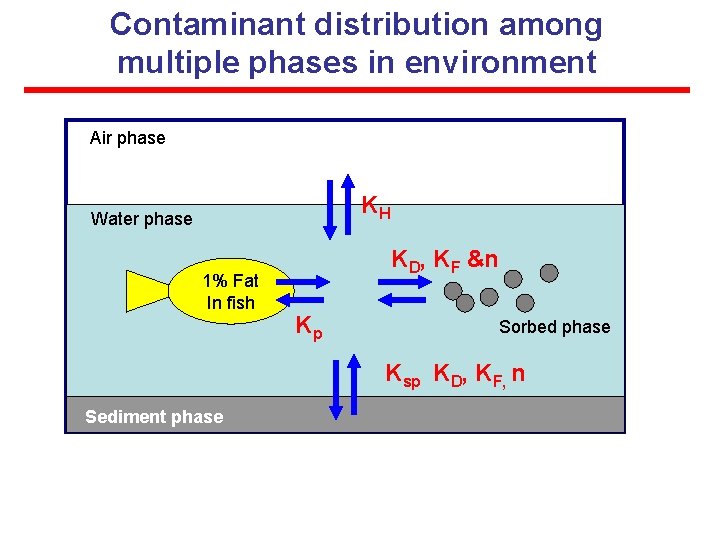
Contaminant distribution among multiple phases in environment Air phase KH Water phase 1% Fat In fish KD, KF &n Kp Sorbed phase Ksp KD, KF, n Sediment phase

Contaminant distribution among multiple phases in environment FOR distribution of a solute (species A) between multi-phases @ equilibrium (used for heterogeneous rxn) Gas & Water : Henry constant, KH PA = KH * CA, (Pi= [ni / V]*R*T) here, P=partial pressure, C=molar conc. Water & Organic phase: Partition Coeff. , Kp C A in organic = Kp * CA in water Solid & Water: Q = KF * Cwn in water (or = KD Cw) Ksp=[A] a[B]b Aqueous Solubility, Cs: Max. concentration of a solute in water Vapor Pressure, PO : Max. concentration of a solute in air

HW #2 (Due April 16) • • • Problem Problem Problem 3. 7 (humidity exercises) 3. 10 (N 2 dissolved in water) 3. 11 (Henry’s constant) 3. 12 (Henry’s law application) 3. 14 (solid solubility) 3. 17 (sorption isotherms) 3. 36 (stoichiometry) 3. 39 (stoichiometry) 3. 45 (kinetics) 3. 53 (kinetics)