Environmental Engineering Lecture Note Week 10 Transport Processes





















- Slides: 32

Environmental Engineering Lecture Note Week 10 (Transport Processes) Joonhong Park Yonsei CEE Department 2016. 5. 11 CEE 3330 Y 2013 WEEK 3

Transport Processes (I) 4. A Basic Concepts and Mechanisms – – Contaminant Flux Advection Diffusion Dispersion 4. D Transport in Porous Media – Fluid Flow through Porous Media – Contaminant Transport in Porous Media CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

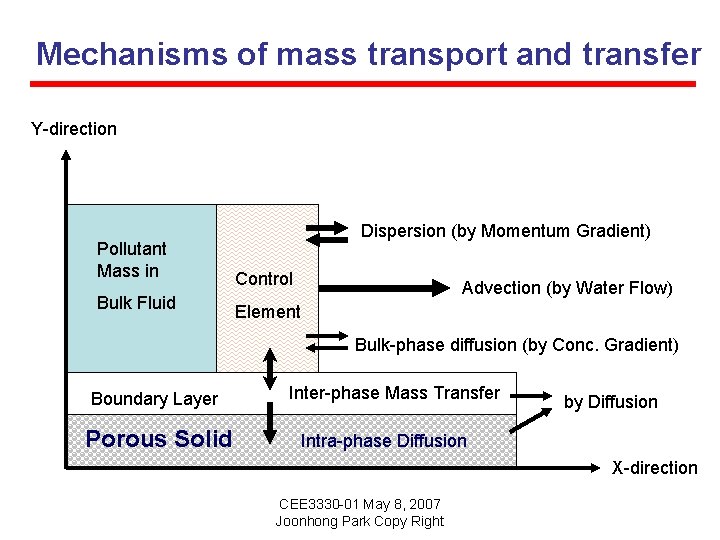
Mechanisms of mass transport and transfer Y-direction Dispersion (by Momentum Gradient) Pollutant Mass in Control Bulk Fluid Element Advection (by Water Flow) Bulk-phase diffusion (by Conc. Gradient) Boundary Layer Porous Solid Inter-phase Mass Transfer by Diffusion Intra-phase Diffusion X-direction CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Definition: Flux of material i (Ni) = The number of moles of material i transported per unit cross-sectional area per unit time = # mole of material i d. A * dt CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

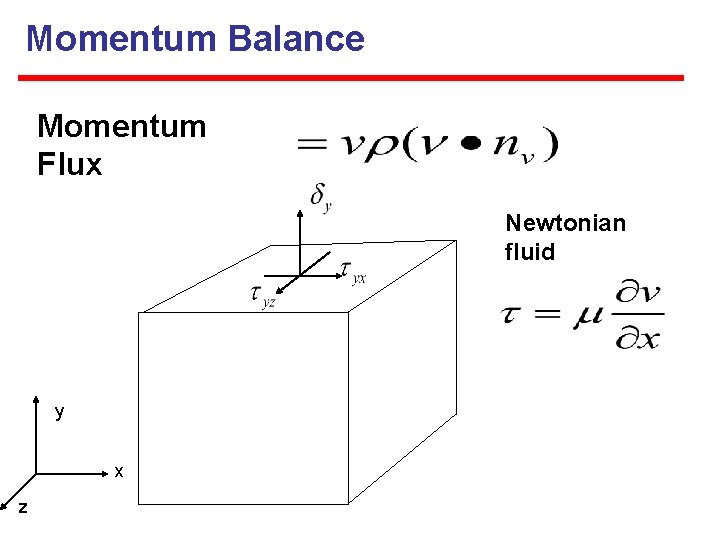
Reynolds transport theorem: Mass continuity Magnitude of the molar flux normal to a differential element of surface area, d. A ~. = |Ni| cos θ = Ni nv ~ Flux is a Vector: Ni = Nix ix + Niy iy + Niz iz CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Characterization of Flow Steady Flow Uniform Flow Unsteady Flow Nonuniform Flow Steady State For Turbulent Flow

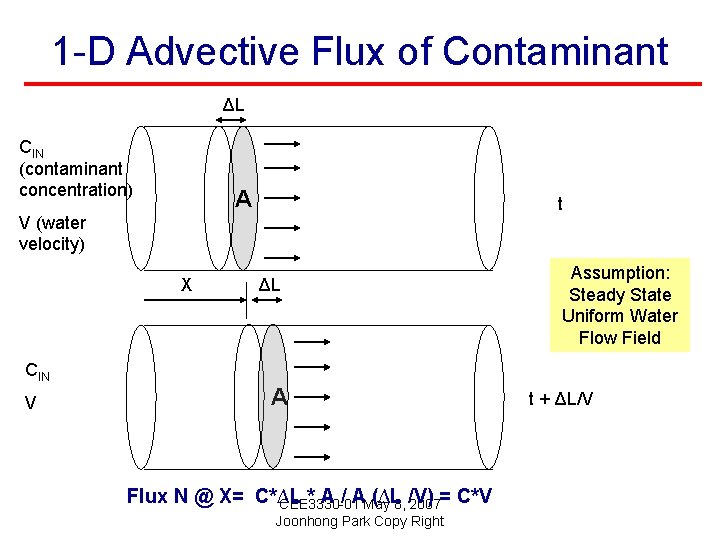
1 -D Advective Flux of Contaminant ΔL CIN (contaminant concentration) A t V (water velocity) X CIN V ΔL A Flux N @ X= C*∆L * A / AMay (∆L 8, /V) CEE 3330 -01 2007= C*V Joonhong Park Copy Right Assumption: Steady State Uniform Water Flow Field t + ΔL/V

Molecular Diffusion The random motion of fluid molecules causes a net movement of species from regions of high concentration to regions of low concentration. The rate of movement depends on the spatial gradient of concentration of a solute. Our discussion is restricted to conditions in which the diffusing species is present at a low mole fraction (the infinite dilution condition). CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

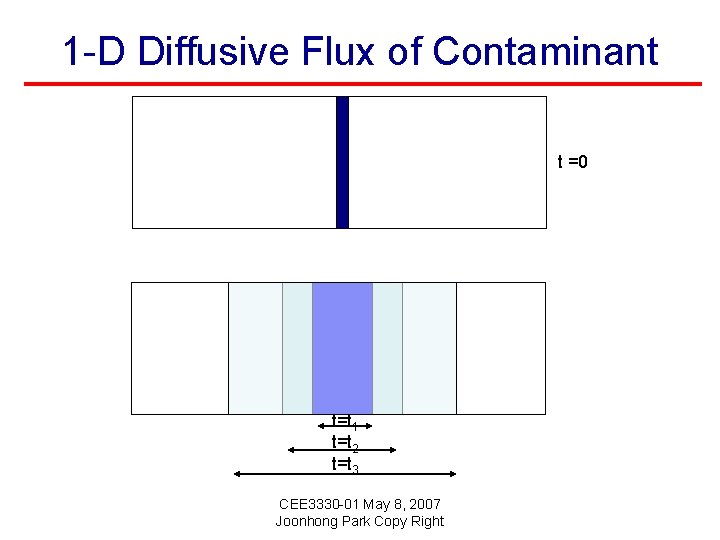
1 -D Diffusive Flux of Contaminant t =0 t=t 1 t=t 2 t=t 3 CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Fick’s 1 st Laws Di : Diffusion coefficient or diffusivity a property of the diffusing species For molecules in air, typically D values is 0. 1 cm 2/s For molecules in water, typically D values is 10 -5 cm 2/s CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

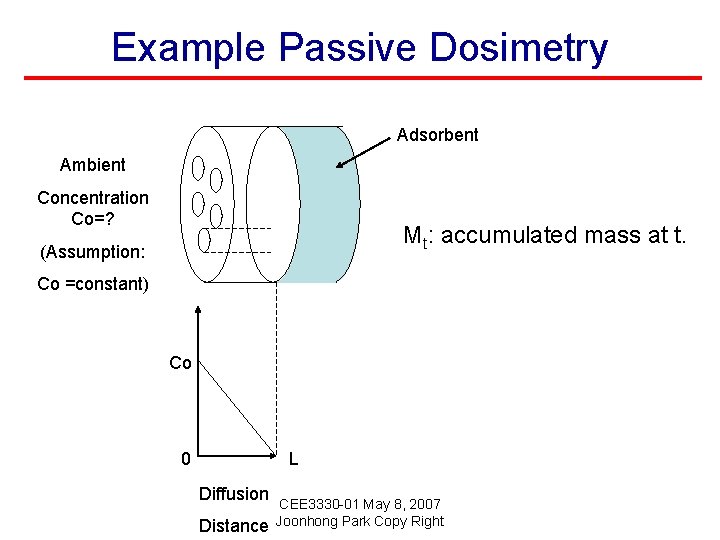
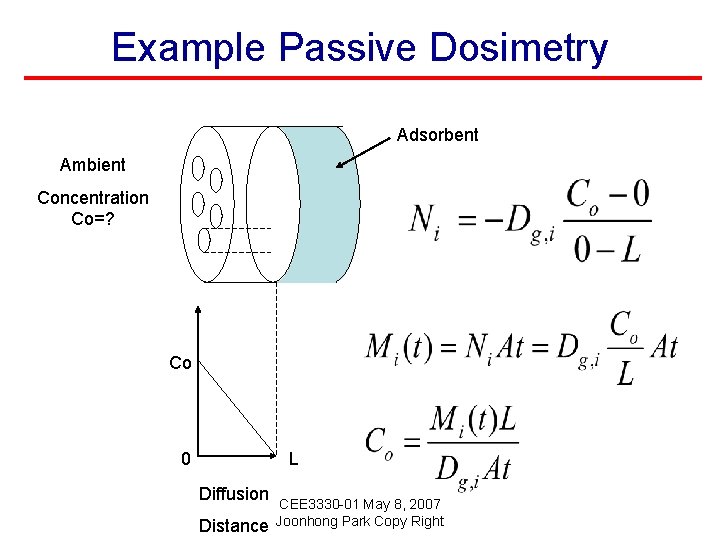
Example Passive Dosimetry Adsorbent Ambient Concentration Co=? Mt: accumulated mass at t. (Assumption: Co =constant) Co 0 L Diffusion Distance CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Fick’s 1 st Laws Di : Diffusion coefficient or diffusivity a property of the diffusing species For molecules in air, typically D values is 0. 1 cm 2/s For molecules in water, typically D values is 10 -5 cm 2/s CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Albert Einstein’s Solution X: traveling distance t: traveling time D: Diffusion coefficient CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Example Passive Dosimetry Adsorbent Ambient Concentration Co=? Co 0 L Diffusion Distance CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

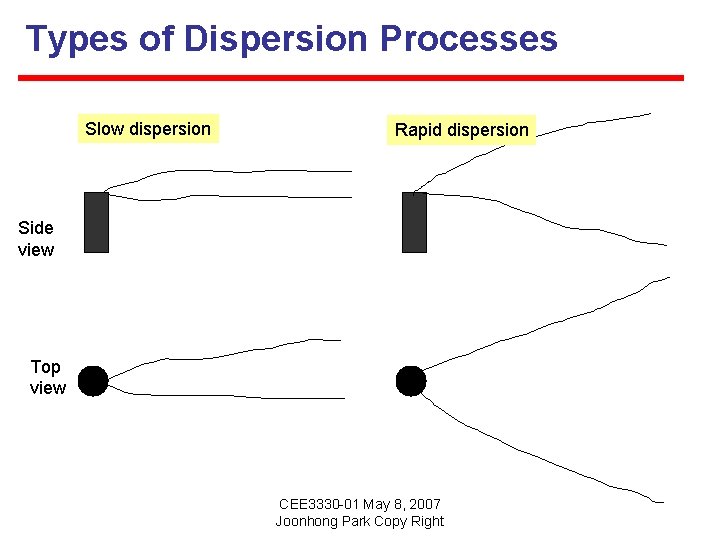
Dispersion The spreading of contaminants by nonuniform flow is called dispersion. This is not a fundamentally distinct transport process. Instead, dispersion is caused by nonuniform advection and influenced by diffusion. A phenomenon caused by the gradient of momentum, which is expressed by a tensor. CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Types of Dispersion Processes Slowdispersion Rapid dispersion Side view Top view CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Types of Dispersion Processes Taylor (Shear Flow) Dispersion: occurs in laminar flow (pipes and narrow channels); transverse direction of solute movement driven by solute concentration gradient Turbulent (eddy) dispersion: velocity fluctuations created by fluid turbulence acting across large advection-dominated fields; large channels, rivers, streams, and lakes. Hydrodynamic and mechanical dispersion: flow in porous media (activated carbon filters; groundwater) CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Shear Flow Dispersion (in a laminar flow) Fluid velocity profile Concentration gradient Injected pulse @ t=0 Dispersed pulse @ t=t Factors to cause the dispersion -Average effect -Concentration gradient due to velocity gradient (momentum gradient) CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

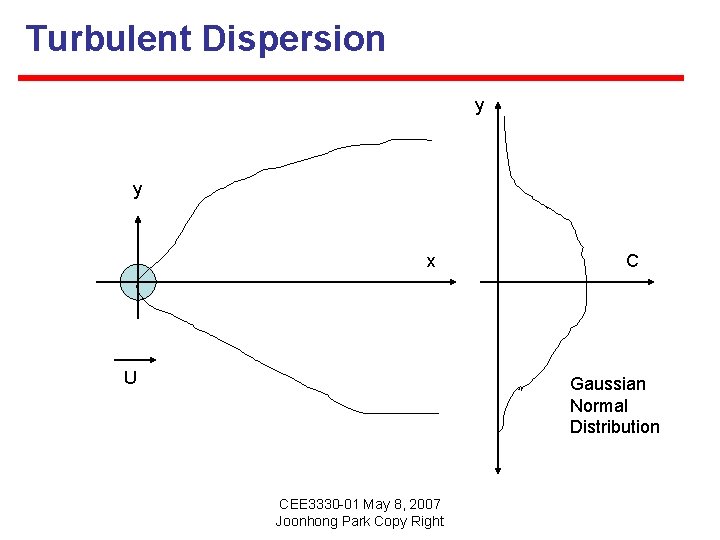
Turbulent Dispersion y y x U C Gaussian Normal Distribution CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

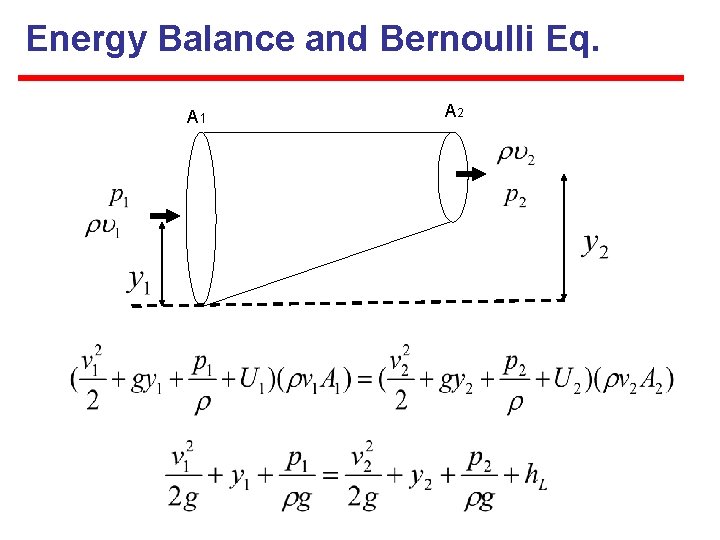
Energy Balance and Bernoulli Eq. A 1 A 2

Momentum Balance Momentum Flux Newtonian fluid y x z

Dispersion Equation Dispersivity (Tensor) Free-Liquid Molecular Diffusion Coefficient (Scalar) Identity Matrix CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Dispersion Distance X: traveling distance t: traveling time Ddd: Dispersion coefficient CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Water Flow in Porous Media - History and equation. - Determination of K and k. CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Darcy’s Experiment (1856) Flow of water in homogeneous sand filter under steady conditions A: cross area Sand Porous Medium h 2 L h 1 Datum Q = - K * A * (h 2 -h 1)/L CEE 3330 -01 May 8, 2007 K= hydraulic conductivity Joonhong Park Copy Right

Darcy’s Experiment (1856) Flow of water in homogeneous sand filter under steady conditions A: cross area Sand Porous Medium h 2 L h 1 Datum Q = - K * A * (h 2 -h 1)/L CEE 3330 -01 May 8, 2007 K= hydraulic conductivity Joonhong Park Copy Right

Darcy’s Law Q = - K * A * (Φ 2 - Φ 1)/L Φ piezometric head In a 1 -D differential form, Darcy’s law may be: q = Q/A = - K * [dΦ/d. L] Hydraulic Conductivity, K (L/T) KΞk*ρ*g/μ Here, k = intrinsic permeability (L 2) ρ: fluid density (M L-3); g: gravity (LT-2) μ: fluid viscosity (M L-1 T-1) CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

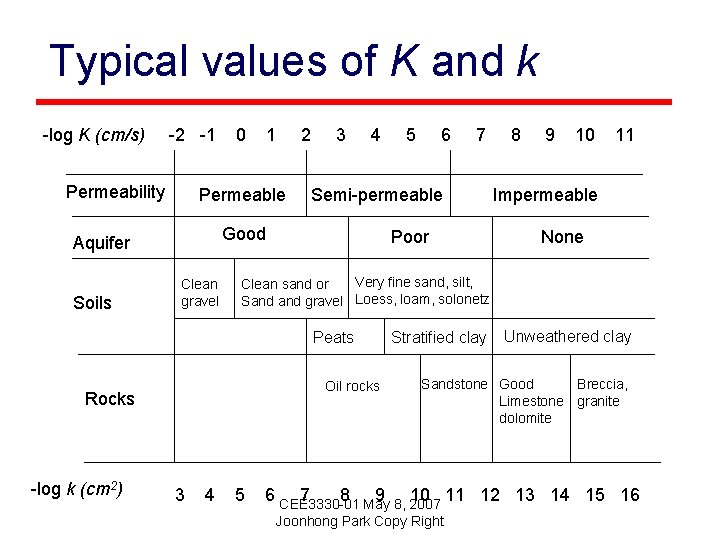
Typical values of K and k -log K (cm/s) -2 -1 Permeability 1 2 Permeable 3 4 Clean gravel Rocks 4 7 8 9 10 11 Impermeable None Very fine sand, silt, Clean sand or Sand gravel Loess, loam, solonetz Oil rocks 3 6 Poor Peats -log k (cm 2) 5 Semi-permeable Good Aquifer Soils 0 5 Stratified clay Unweathered clay Sandstone Good Breccia, Limestone granite dolomite 6 CEE 3330 -01 7 8 May 9 8, 2007 10 11 12 13 14 15 16 Joonhong Park Copy Right

Hydrodynamic Dispersion Mechanical Dispersion (Tensor) In one-D system, α, dispersivity V, pore velocity (=q/n) Molecular Diffusion (Scalar) Identity Matrix CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

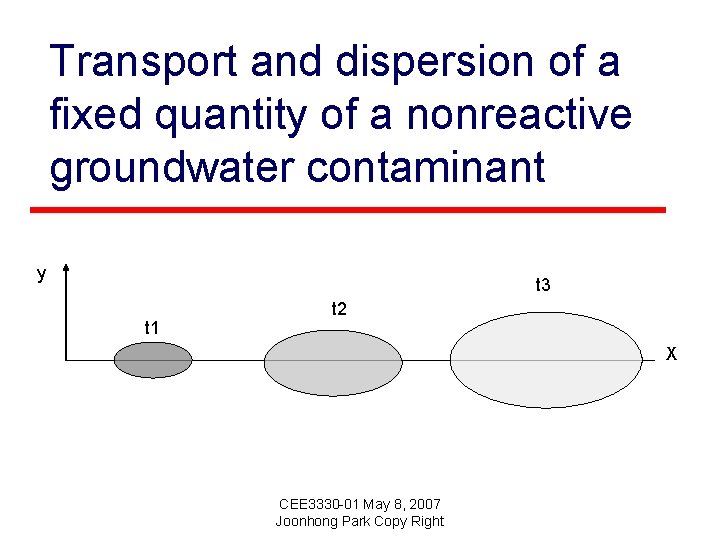
Transport and dispersion of a fixed quantity of a nonreactive groundwater contaminant y t 3 t 1 t 2 X CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

Reading Assignment Read p. 159 -172 Practice EXHIBIT 4. A. 1 at p. 165 EXAMPLE 4. D. 1 at p. 196 -197 CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right

HW Problem 4. 1 Problem 4. 4 Problem 4. 6 Problem 4. 12 CEE 3330 -01 May 8, 2007 Joonhong Park Copy Right