Environmental Chemistry What is pollution Pollution Pollution undesirable

Environmental Chemistry What is pollution?

Pollution • Pollution: undesirable contaminant that an _______ adversely affects the ______, ______ chemical biological physical ______ or ______ characteristics of the environment • 3 Types of Pollution: – ______ air land – ______ water – ______ natural • **ALL types of pollution can be _____ or anthropogenic (human-made). ________

Air Pollution: • There are several layers to the atmosphere. Each layer of the earth has a ______ different composition ________. We will be focusing on pollution in the two closest layers of the earth’s troposphere atmosphere: the _______ stratosphere and the ________. (stratos video – 8 min)

Air Pollution Exosphere Thermosphere (506 km) Mesosphere (87 km) Stratosphere (50 km) Troposphere (11 km)

Air pollution in the Troposphere air troposphere layer of the **The _______ we breathe is in the ______ Air is a _______ gases atmosphere. _____ mixture of _______. homogeneous _____ • Homogeneous mixture: 2 or ________that evenly more substances a mixture of ____ is mixed ____ (no layers) *The percentages of the gases in the air stay fairly consistent, however there is one gas in the air that changes in abundance. H 2 O vapor humidity _____amounts change depending on the ______ of the air in a particular area.

Composition of the Troposphere

Pollutants Other changes in the composition of the air: The composition of the air can change very quickly based pollutants are added to our _____ on what ______ clean air. This is called _______. If pollution is added to the air in air pollution factories, cars houses one location because of _________or ______, it can easily move to other places because of air movement (wind) _______

Greenhouse Gases insulate the • Some of these additional substances are gases that _____ heat Without having hold earth like a blanket. They ____ in ______. troposphere the earth would be very some of these gases in the _______, cold and barren. However, too much of a blanket on the earth can Earth’s temperature increase rather than just keeping it make the _____________, a stable temperature. This process of the earth heating up is called Global warning ___________. insulators for the earth include: • Some of the gases that act as ______ – CO 2 (**most responsible for increase in global temperature) – CH 4 – H 2 O greenhouse gases They are called __________.

Greenhouse Effect trap heat energy (infrared light) • These gases _______________that we get from the sun to keep it from bouncing back out into outer space, much like greenhouses trap energy from the sun for plants. That is why this process of gases trapping heat inside the earth’s atmosphere is Greenhouse Effect called the ___________. Greenhouse Effect: the trapping and returning of heat (infrared radiation) to the earth’s surface by atmospheric substances called greenhouse gases **The more __________(CO 2, H 2 O and CH 4) we allow to build up in the atmosphere, the more heat will be trapped inside the greenhouse gases earth’s atmosphere and the ________begins to increase over time (global warming). This process is _____, gradual over time.
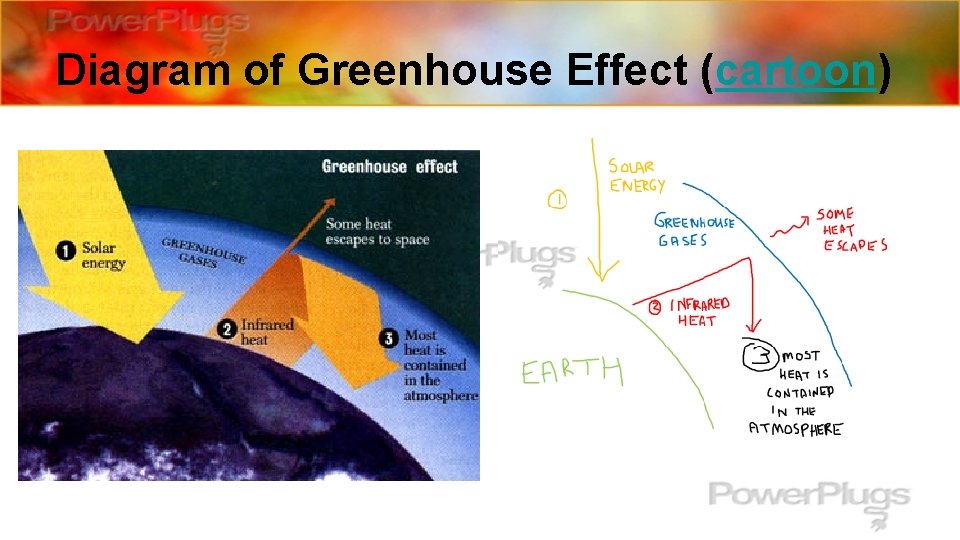
Diagram of Greenhouse Effect (cartoon)

Sources of CO 2 • Natural: – – – ______ eruptions volcanic forest fires ____ decay of dead plants and _______ animals • Anthropogenic: burning of fossil fuels (coal, – _____ natural gas and oil): cars, home, industry – – evaporation from the ocean ______ respiration ______ deforestation – _________: less trees to breath in the CO 2 waste – _____ incineration

Pictures

Sources of CH 4 • Natural: – – – swamps and marshes ____________ termites _______ bacterial processes ___________ • Anthropogenic: mining – _______ and burning of fossil fuels (coal, natural gas and oil): cars, home, industry – _______ decay of dead plants and animals – cow flatulence

Pictures

Sources of H 2 O • Natural: water cycle – _________ • Anthropogenic: – All of our water is involved in the water cycle however, with increasing global evaporate _______ temperatures more water could ________ causing more water vapor in the atmosphere. China is leading the world in greenhouse gas production. **_____
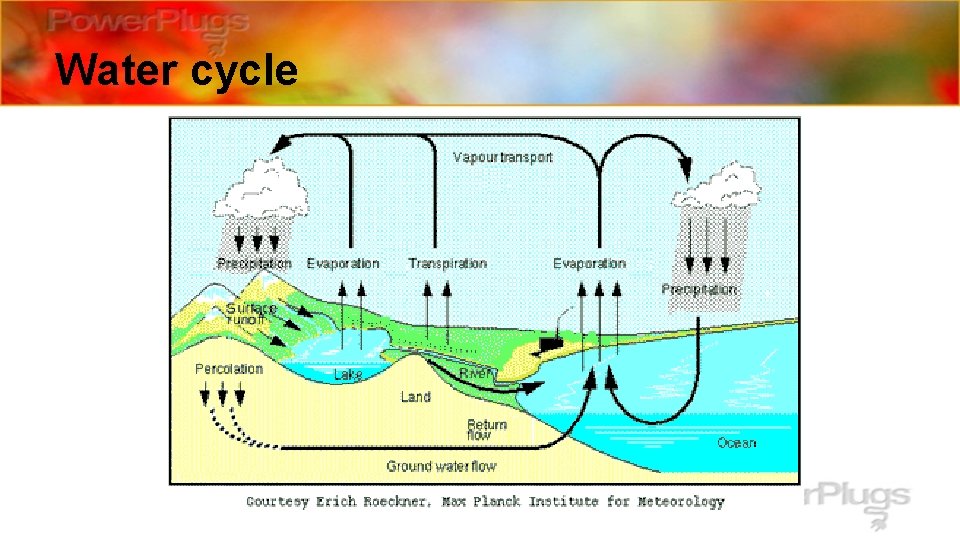
Water cycle

Implications: what does this mean for us? • Implications: Extreme weather events – ____________(hurricanes, floods, tornadoes) melting – ______of polar ice caps more fresh water and be • this means that the oceans will have ____ less ______ salty • this also means that some of the plant and animal life in the oceans die could ______ Agricultural – _________ land use changes extinction habitats – plant and animal ______ due to different _____ COMPLETE PAGES 1 AND 2 OF UNIT 10 PACKET

Air Pollution in the Stratosphere stratosphere is the layer of the earth’s atmosphere with • The _______ O 3 the _______. The ozone layer is composed of ____ ozone layer different from the ____ O 2 that we breathe. molecules, which are _____ toxic to breathe and ______ blue in color. Ozone is ____ ultraviolet • The ozone layer absorbs _______ radiation from the sun. UV rays sunburns, skin caner These ______are what cause ___________, cataracts plant life on the and _______. UV rays can also damage ____ land ocean ____ and in the ____.

CFC’s • There are some compounds that break down the O 3 in the stratosphere. Some of these compounds are called CFC’s (chlorofluorocarbons). Sources of CFC’s are all anthropogenic: – aerosol spray propellants – refrigerants or coolants (freon) – Solvents (dry-cleaning) – foam blowing agents (fire foam) – some plastics Scientists are currently trying to find viable alternatives to CFC’s. These CFC’s act as catalysts to break down ozone molecules. Because catalysts never get used up, they can keep breaking down ozone, therefore one CFC continues to break down ozone molecules for a long time.

Implications: • Implications: – sunburns – skin cancer – cataracts – lower yield of food crops – damage of phytoplankton in ocean (phytoplankton are the #1 producers of oxygen on the earth by photosynthesis) • Photosynthesis rxn: CO 2 + H 2 O C 6 H 12 O 6 + O 2

Other Air Pollutants: There are some pollutants released into the air that cause significant health problems for people. Industrialization has caused many chemicals to be put into the air that should not be there. The burning of fossil fuels such as coal, oil, and natural gas produces the following chemicals: • Sulfur dioxide (SO 2) : when mixed with the water in the clouds makes H 2 SO 4 (sulfuric acid) which contributes to acid rain which contaminates our water sources, erodes buildings, damages vegetation, cause asthma and lung disease.

Other Pollutants • Carbon Monoxide (CO) : a toxic gas which prevents the transport of oxygen in the blood and therefore can cause heart disease. • Particles: solid particles of smoke can be breathed into the lungs causing lung damage. • Nitrogen Dioxide (NO 2) : a toxic gas that irritates the lungs and cause respiratory infections such as bronchitis. • Ozone (O 3) : when ozone is in the air we breathe instead of the stratosphere, it can cause respiratory illness such as asthma, bronchitis and emphysema.

Legislation: what are we doing to stop air pollution? • Legislation: – 1970 Air Quality Act • 1 st legislation that limited the amount of pollution that can be released into the air by industries and vehicles – 1987 Montreal Protocol • International agreement to phase out all production and consumption of CFC’s by 2000, with a few exceptions – 1997 Kyoto Protocol • UN treaty to reduce the production of greenhouse gases of each country by a certain % COMPLETE PAGES 3 AND 4 OF UNIT 10 PACKET

Land Pollution Land has many uses: it is important for agriculture, ranges and pastures, homes, forests, recreation and industry. **When our land gets polluted with chemicals that were not originally on the land, OR when we remove a material that was originally on the land, all of these uses are affected. We are going to focus on three forms of land pollution: • Landfills • Deforestation • Urbanization

Landfills: a location where solid waste disposal is regulated and controlled (everything you throw into the garbage goes here) Problems: • Landfills can leak, leaching into our water system • Landfills are too compressed to allow for decomposition of waste • Many compounds are non-biodegradable and therefore takes many, many years to break down (plastics, metal and glass)

Fresh Kills Landfill on Staten Island

Landfills Possible Solutions: • Reduce: use less non-biodegradable materials • Reuse: use the same product repeatedly instead of throwing it out • Recycle: reprocessing of discarded materials into new products **Not all materials can be recycled, and generally you cannot recycle a material into the same product you started with. There also costs associated with recycling

Deforestation: the removal of trees from a forest without replacement Problems: • Trees use CO 2 and produce O 2 for us to breathe. With less trees there is less O 2 and more CO 2 which is a greenhouse gas • Loss of phytochemicals (aka phytonutrients): chemicals found in trees and plants that are later discovered to contain cures for many diseases ( any compound considered to be beneficial to human health) • Loss of biodiversity: forests are the home for many plants and animals, with a loss of their habitat many species are on the verge of extinction Possible Solutions: • Preservation of natural resources • Replanting

Urbanization: the increasing concentration of the population in cities and the transformation of green land into cities Problems: • More waste in a concentrated area • Less plant life, including forests • Filling in of wetlands/ marshes for roads, buildings Possible Solutions: • Maintaining green space in cities • Green industry

Legislation: • SEQR Act of NYS: Must get an environmental assessment done before any land use changes can be made to ensure the preservation of biodiversity and green space (only in NYS) • Wetlands Act: Class 1 wetlands are protected from development across the USA

Water Pollution: The amount of water on the earth is constant. The water cycle is a series of physical changes in water that include evaporation, condensation and precipitation.

Water Pollution **ANY pollution can be point source discharge or non-point source discharge. Point source discharge: • Pollution comes from a specific location like drain-pipes, ditches, sewer outfalls, oil wells, factory exhaust pipes Non-point source discharge: • Pollution that comes from a scattered source with no specific location like run -off from farming fields (pesticides and fertilizers), lawns, gardens, golf courses

Types of H 2 O Pollution Types of water Pollution: • Thermal: water gets heated because it is being used to cool factory processes • Ocean dumping: some cities run out of landfill space and dump solid waste into the ocean • Medical waste: some hospitals throw needles, gloves and other medical waste into the nearest water source • Pharmaceuticals: when our bodies don’t use all of a medicine, our bodies dispose of it as waste. These chemicals then go into the sewage plants and often don’t get removed before the water gets sent back to a freshwater source. Our drinking water then comes from this freshwater source.

Legislation: • Water Quality Act of 1970: Federal law that requires ship owners to clean up or pay for the cleanup of waters polluted by discharges from their ships

Connections **All three types of pollution are related. Land pollution can runoff into lakes and streams to pollute the water. Air pollution gets into the water in the clouds and becomes water pollution, and water pollution can rain down on the land. Air pollution is also a problem because due to weather patterns, it can travel many miles and rain down in relatively clean areas. COMPLETE PAGES 5 AND 6 OF UNIT 10 PACKET
- Slides: 35