Environmental Chemistry soilcompositionsoil chemistry Rani Abraham Syllabus portions

Environmental Chemistry soil-composition-soil chemistry Rani Abraham

Syllabus portions • The chemistry of processes in Lithosphere; Redox status in soil. p. E p. H predominance diagrams for redox sensitive elements. Acidity in soil materials. Acid neutralization capacity and the quantification of the soil acidity. Ion speciation in soil solution. Cation exchange capacity and exchange phase composition.

Soil chemistry • Healthy soils rely on water, oxygen, air and a balanced supply of nutrients. Chemical reactions within the soil can alter the physical structure of available nutrients. • Oxidation and reduction processes involve the oxygen exchanges that take place between water and inorganic materials. These changes affect available nutrient supplies.

Redox reactions • The chemical reactions that take place in the soil depend on the physical makeup of each material. The presence of oxygen and water in the soil can increase the likelihood of a redox reaction, depending on the physical makeup of the soil’s materials.

REDOX STATUS OF SOIL • Indicates the nature of the soil • For eg. , in wetlands it differentiates between a well drained soil, poor drained soil and submerged soil • A waterlogged or anaerobic soil is usually greyish or green in colour, has a low redox potential and contains the reduced form of various soil minerals (Ponnamperuma 1972). An anaerobic environment affects the oxidation and reduction reactions in soil that govern the formation of many species

Redox status in soil standard method measuring the voltage difference between a Pt sensing electrode and a reference electrode (e. g. , calomel or Ag/Ag. Cl) inserted directly into the soil (Mueller et al. , 1985). The voltage difference between the Pt and reference electrodes is then corrected for the reference electrode’s standard voltage relative to the standard hydrogen electrode (SHE, E = 0. 0 V), and is recorded as the Eh.

Pourbaix diagram (Electrode potential / p. H diagram) • Graphical presentation of thermodynamic equilibrium states of a metal -electrolyte system. • Axes Electrode potential of the metal vs. PH of the electrolyte. Oxidizing conditions are described by the top part of the diagram (high positive electrode potential). Reducing conditions are described by the bottom part of the diagram (high negative electrode potential). Acidic solutions are presented in the left side of the diagram (p. H lower than 6). Alkaline solutions are presented in the right side of the diagram (p. H higher than 6).

Basis of different zones on the plot-Nernst equation • The lines of the diagrams dividing different zones of the equilibrium states are calculated by the Nernst equation: E = E 0 - (0. 059/n)*ln. Cion Where: E 0 - Standard electrode potential, V; n - number of electrons transferred; Cion - molar activity (concentration) of ions.

Significance of Pourbaix diagrams • Allows one to determine the corrosion behaviour of a metal in water solutions i. e. the direction of electro-chemical processes and the equilibrium state of the metal at a certain electrode potential in a water solution at a certain value of PH. Normally the Poubaix diagrams are built for the water solutions with the concentrations of metal ions 10 -6 M and at the temperature 298 K (77ºF/25ºC).

Pourbaix diagram - illustration • An example of a Pourbaix diagram for the system iron-water is presented in the following picture.
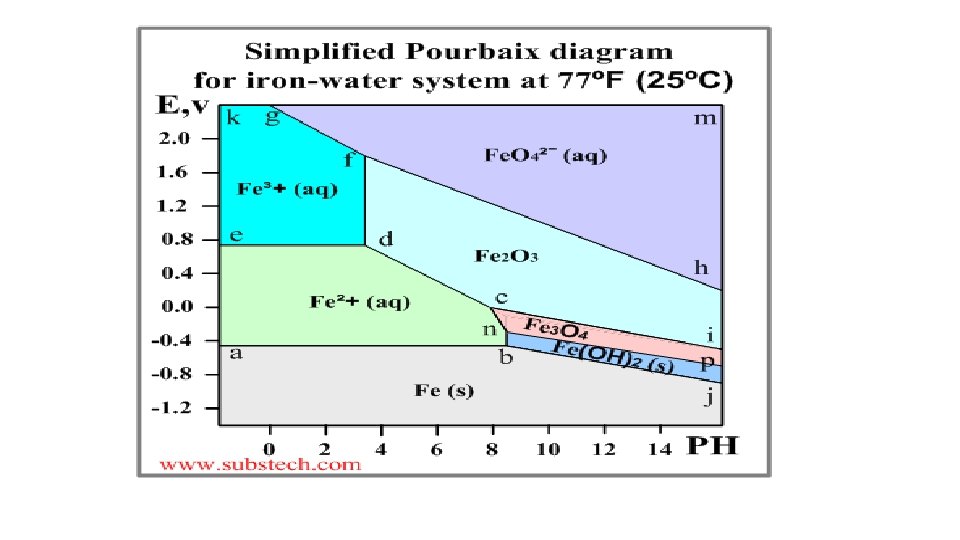
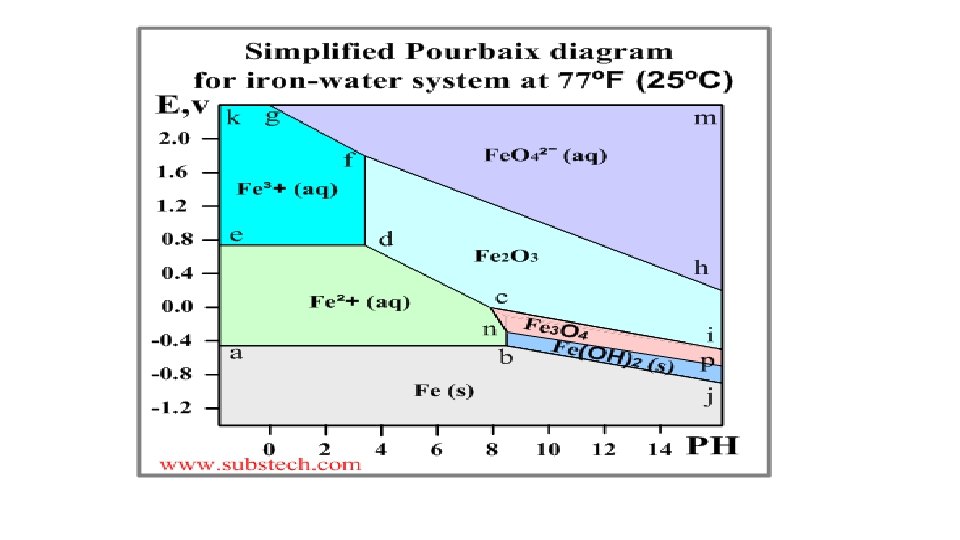
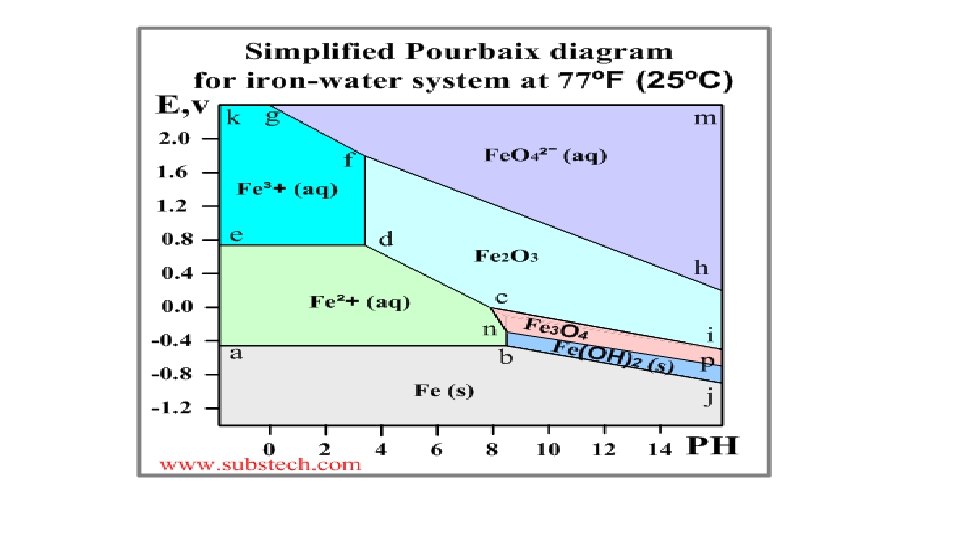
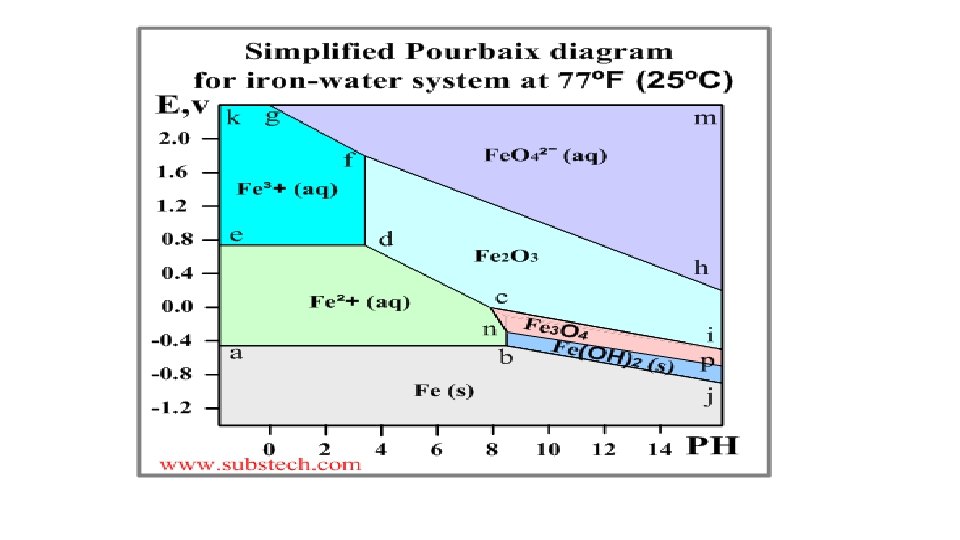
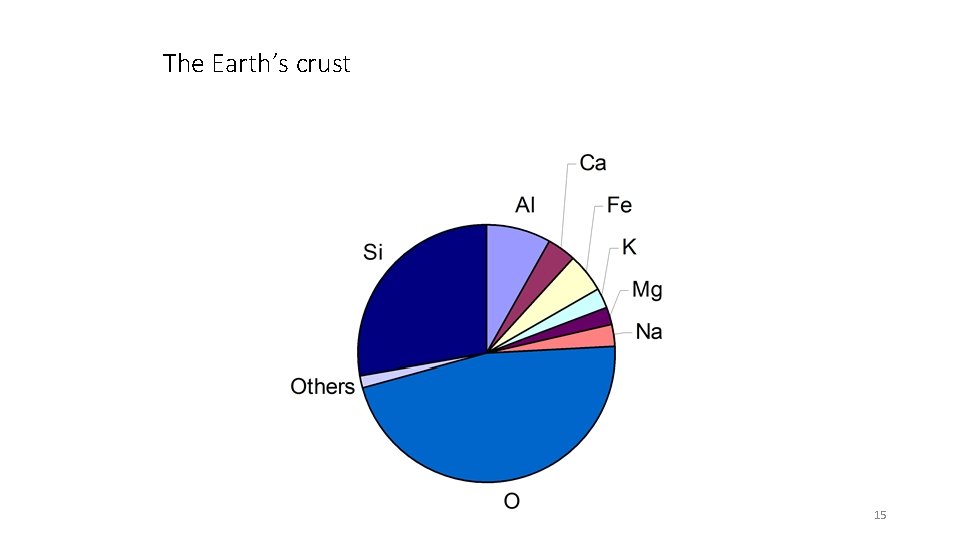
The Earth’s crust Soil Analysis Ch 5 15

Soil Components The 4 parts of soil About ½ of the soil volume is solid particles About ½ of the soil volume is pore space

Soil chemistry Study of the chemical characteristics of soil. • Soil chemistry is affected by: • mineral composition, • organic matter and • environmental factors.
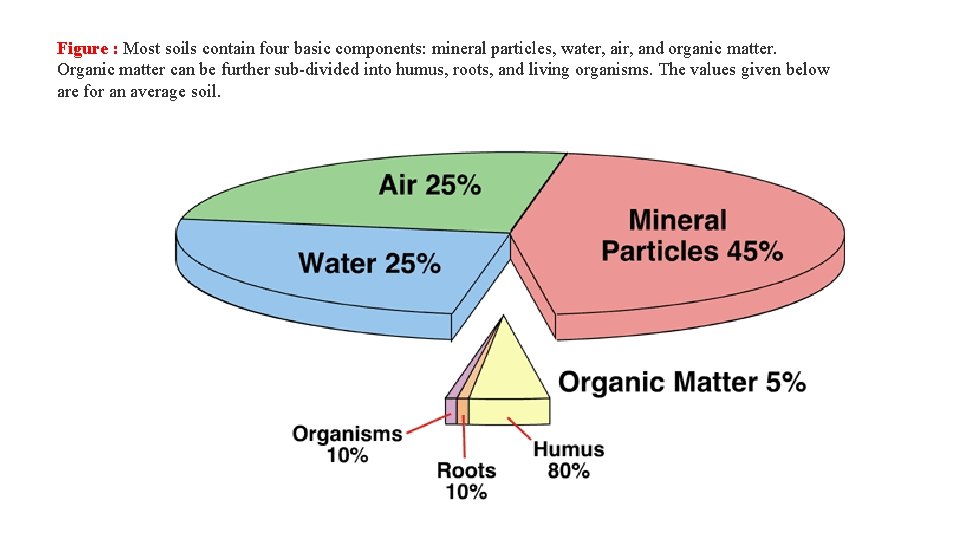
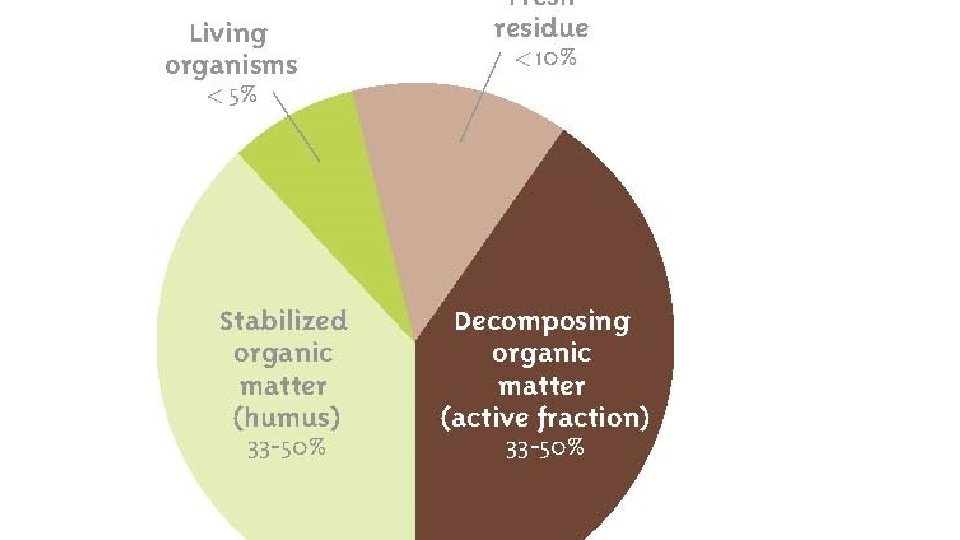
Figure : Most soils contain four basic components: mineral particles, water, air, and organic matter. Organic matter can be further sub-divided into humus, roots, and living organisms. The values given below are for an average soil.

Soil Texture • Texture of a soil refers to the size distribution of the mineral particles found in a representative sample of soil. • Particles are normally grouped into three main classes: • sand, • silt, • clay.
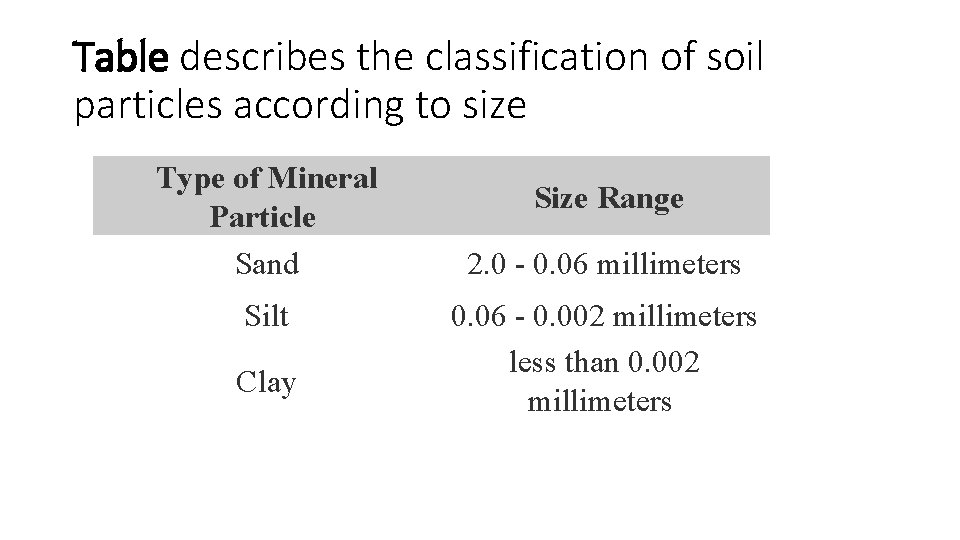
Table describes the classification of soil particles according to size Type of Mineral Particle Sand Silt Clay Size Range 2. 0 - 0. 06 millimeters 0. 06 - 0. 002 millimeters less than 0. 002 millimeters

CLAY Probably the most important type of mineral particle found in a soil Characterized by small size and very large surface area of the particles. Highly reactive due to large surface area Has the ability to attract and hold positively charged nutrient ions which are available to plant roots for nutrition. • Clay particles are also somewhat flexible and plastic because of their lattice -like design. This feature allows clay particles to absorb water and other substances into their structure. • •

Soil chemistry-why is it important • The chemistry determines what will grow and how well it will grow. One of the most important chemical properties of a soil is its acidity or alkalinity, often stated as the p. H of the soil. Factors affecting soil chemistry • Factors that affect soil formation. Soils form from the interplay of five main factors namely Parent material, Time, Climate, Relief and Organisms. • Parent material: This refers to the mineral material or organic material from which the soil is formed. •

Composition of soil • Soil is composed of a matrix of minerals, organic matter, air, and water.

OXIDATION AND REDUCTION REACTIONS • Soils that alternate between wet and dry go from having a lot of oxygen to not a lot of oxygen. The presence or absence of oxygen determines how soils chemically react. Oxidation is the loss of electrons, and reduction is the gaining of electrons at the soil surface. These type of reactions occur every day, and are responsible for creating things like rust. Soils, because they contain a lot of iron, can also rust, or if they contain a lot of water, can turn a light gray color. This is partially responsible for all of the different colors that are found, and creates the speckles usually found deeper in the soil. •

• Ion exchange involves the movement of cations (positively charged elements like calcium, magnesium, and sodium) and anions (negatively charged elements like chloride, and compounds like nitrate) through the soils • Cation exchange is the interchanging between a cation in the solution of water around the soil particle, and another cation that is stuck to the clay surface. The number of cations in the soil water solution is much smaller than the number that is attached to soil particles. •

A. Cation Exchange Capacity • Cation exchange capacity (CEC) is the total amount of cations that a soil can retain • The higher the soil CEC the greater ability it has to store plant nutrients • Soil CEC increases as 1. The amount of clay increases 2. The amount of organic matter increases 3. The soil p. H increases
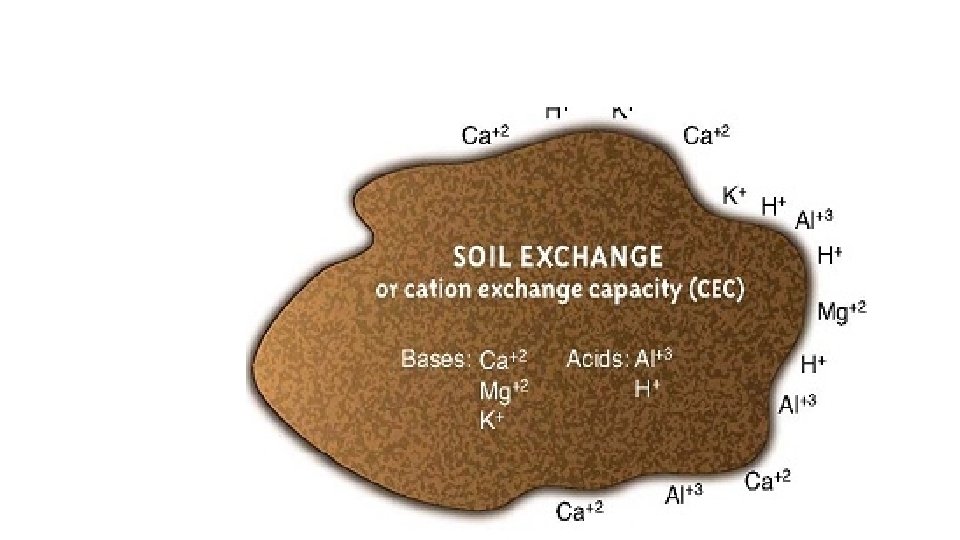

Cation exchange capacity (CEC) • The total amount of positive charges that the soil can absorb is called the cation exchange capacity (CEC). CEC impacts how quickly nutrients move through the profile. A soil with a low CEC is much less fertile because it cannot hold on to many nutrients, and they usually contain less clays. If your soil has a low CEC, it is important to apply fertilizer small doses so it does not infiltrate into the groundwater. A soil with a low CEC is less able to hold spilt chemicals. •
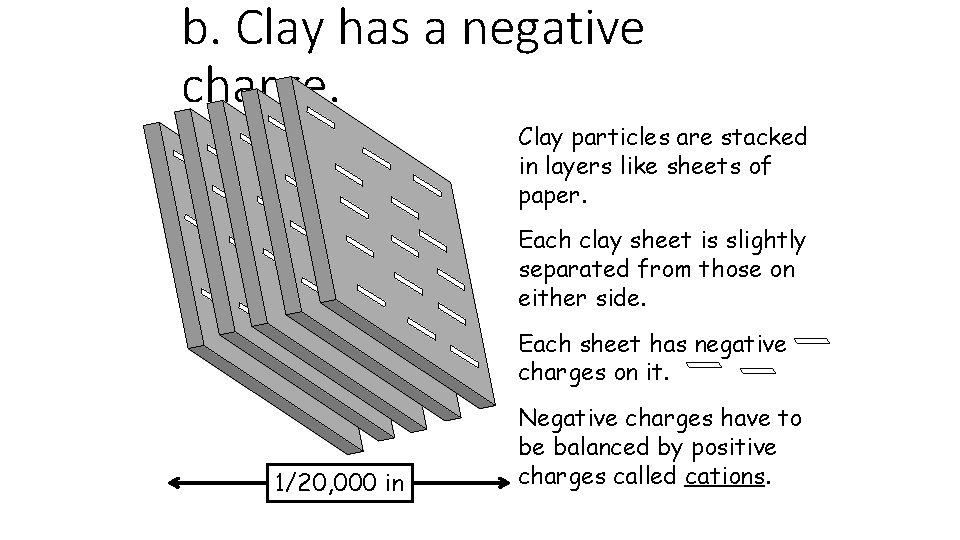
b. Clay has a negative charge. Clay particles are stacked in layers like sheets of paper. Each clay sheet is slightly separated from those on either side. Each sheet has negative charges on it. 1/20, 000 in Negative charges have to be balanced by positive charges called cations.
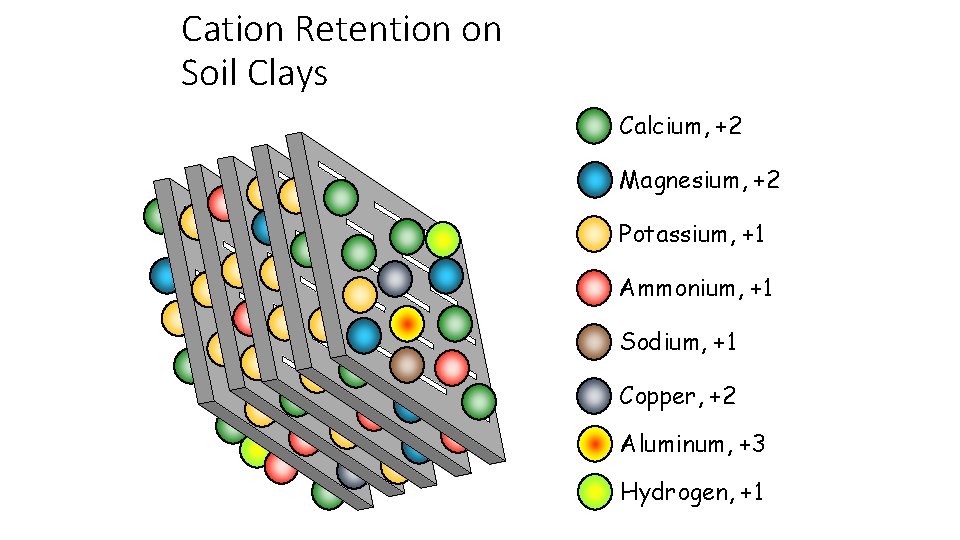
Cation Retention on Soil Clays Calcium, +2 Magnesium, +2 Potassium, +1 Ammonium, +1 Sodium, +1 Copper, +2 Aluminum, +3 Hydrogen, +1
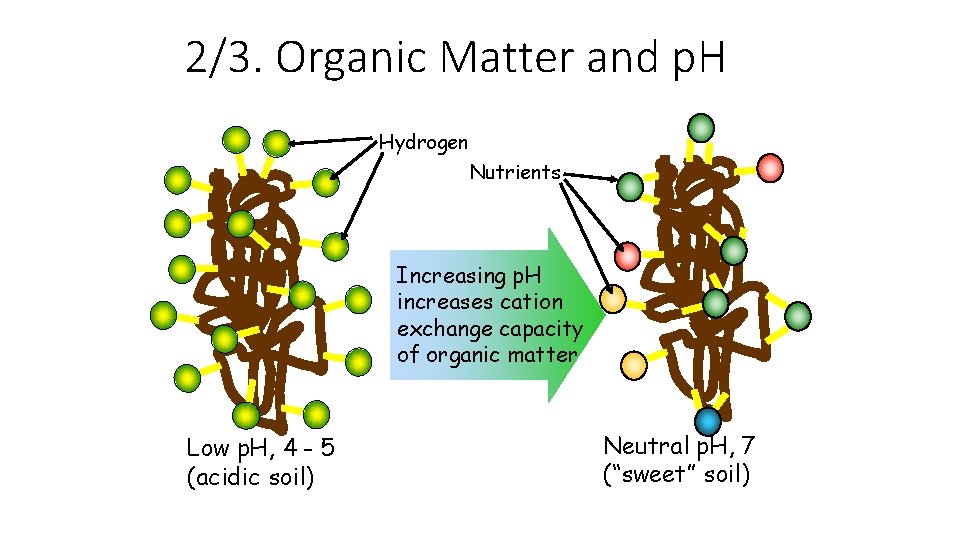
2/3. Organic Matter and p. H Hydrogen Nutrients Increasing p. H increases cation exchange capacity of organic matter Low p. H, 4 - 5 (acidic soil) Neutral p. H, 7 (“sweet” soil)

B. Negatively Charged Nutrients (Anions) • Some very important plant nutrients are anions. Nitrate Phosphate Sulfate Chloride • Soils are able to retain some of these nutrient anions. • Retention of nutrient anions varies from one anion to another
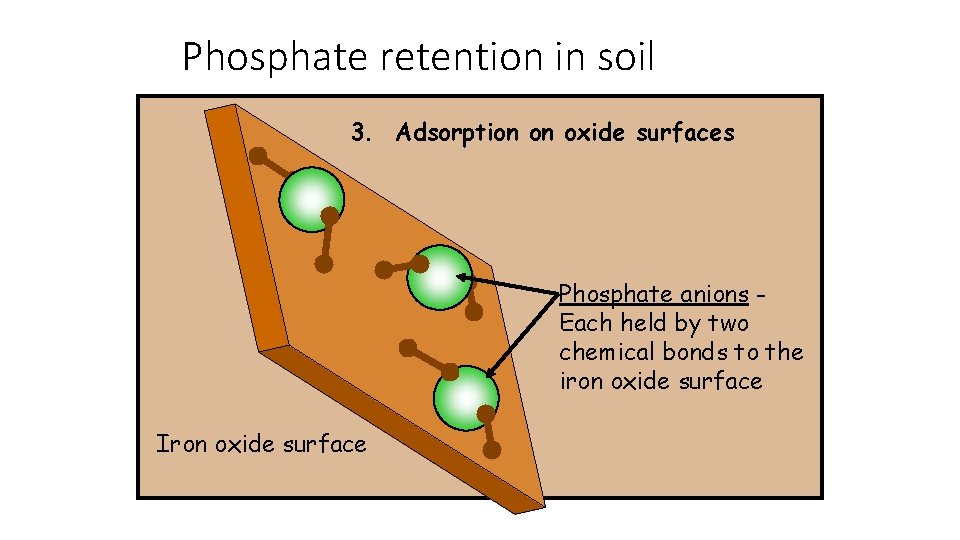
Phosphate retention in soil 3. Adsorption on oxide surfaces Phosphate anions Each held by two chemical bonds to the iron oxide surface Iron oxide surface
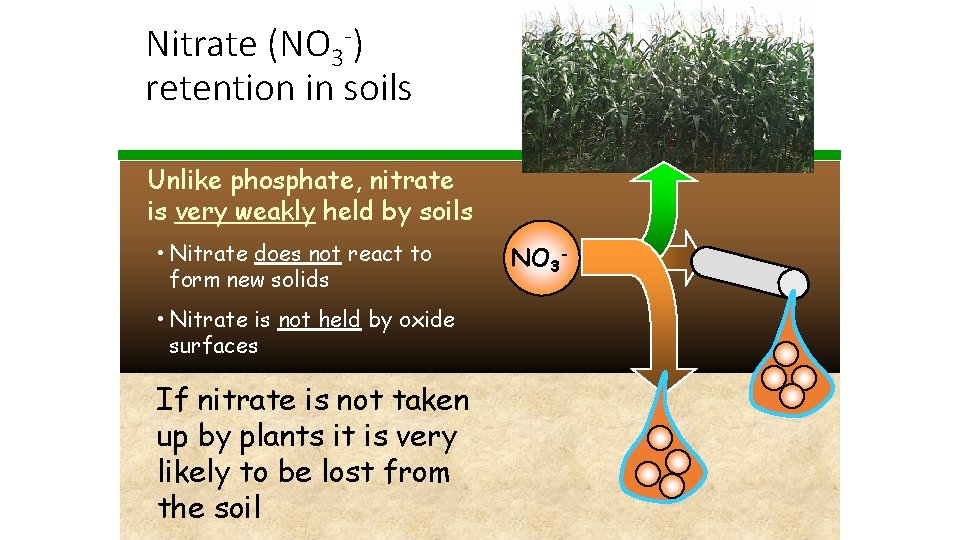
Nitrate (NO 3 -) retention in soils Unlike phosphate, nitrate is very weakly held by soils • Nitrate does not react to form new solids • Nitrate is not held by oxide surfaces If nitrate is not taken up by plants it is very likely to be lost from the soil NO 3 -
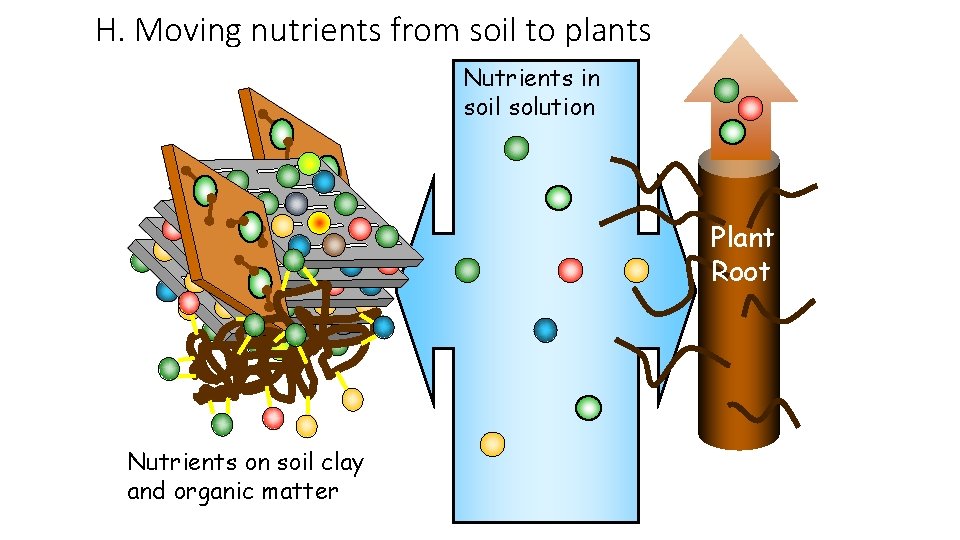
H. Moving nutrients from soil to plants Nutrients in soil solution Plant Root Nutrients on soil clay and organic matter
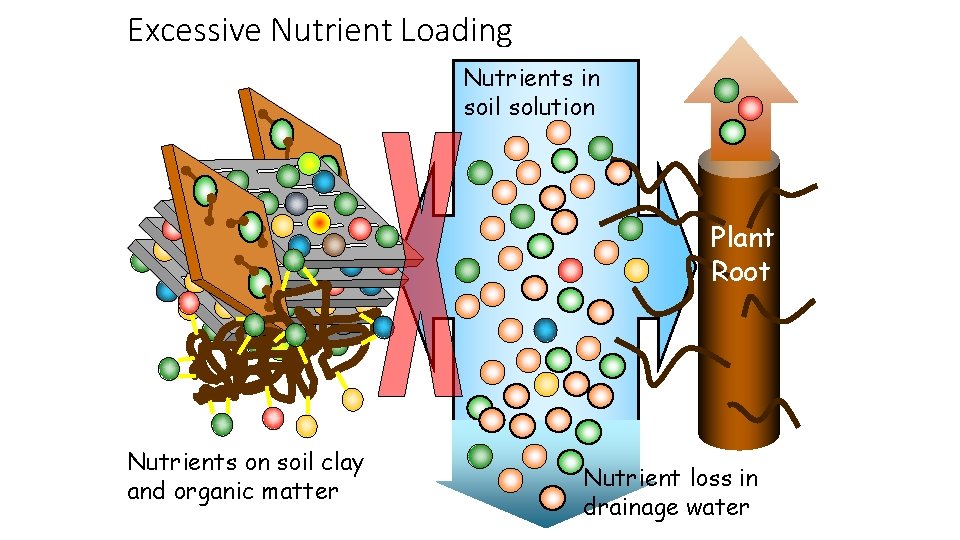
Excessive Nutrient Loading Nutrients in soil solution Plant Root Nutrients on soil clay and organic matter Nutrient loss in drainage water

SOIL PH • The soil p. H is a measure of soil acidity or alkalinity. p. H can range from 1 to 14, with values 0 -7 being acidic, and 7 -14 being alkaline. Soils usually range from 4 to 10. The p. H is one of the most important properties involved in plant growth, as well as understanding how rapidly reactions occur in the soil. . For example, the element iron becomes less available to plants a higher the p. H is. This creates iron deficiency problems. Crops usually prefer values between 5. 5 -8, but the value depends on the crop. The p. H of soil comes from the parent material during soil formation, but humans can add things to soils to change them to better suit plant growth. Soil p. H also affects organisms. •

SOIL p. H A measure of the degree to which the soil is Acidic or Basic; also known as. . . Soil Reaction

SOIL p. H Most agricultural soils p. H range from 4 to 9. . . common productive agriculture soils fall between p. H 5 to 8

SOIL p. H 2 factors affecting p. H are: 1) Amount of precipitation 2) Type of vegetation

SOIL p. H AMOUNT OF PRECIPITATION – HUMID AREAS (heavy rainfall) ACID SOILS Ca+, Mg+, and Na+ are leached downward and H+ ions replaced by
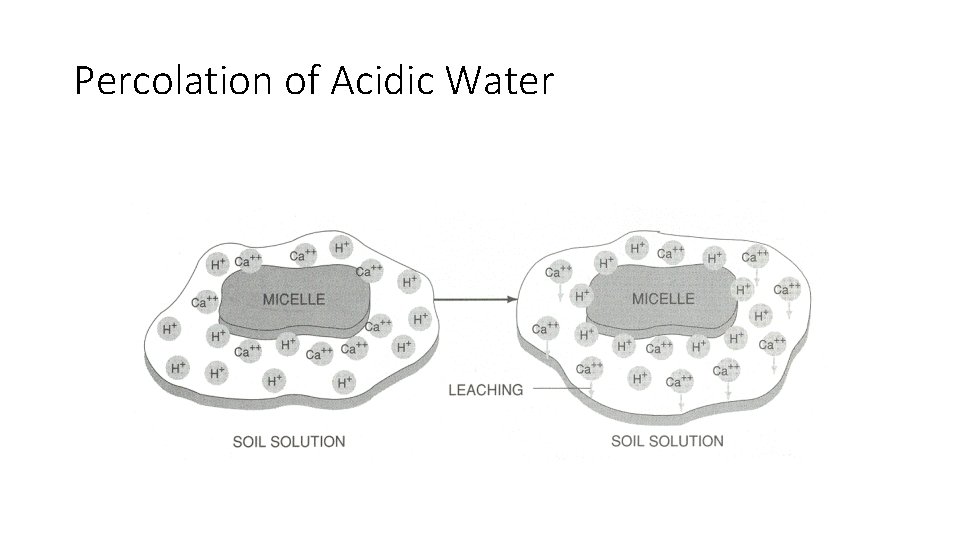
Percolation of Acidic Water
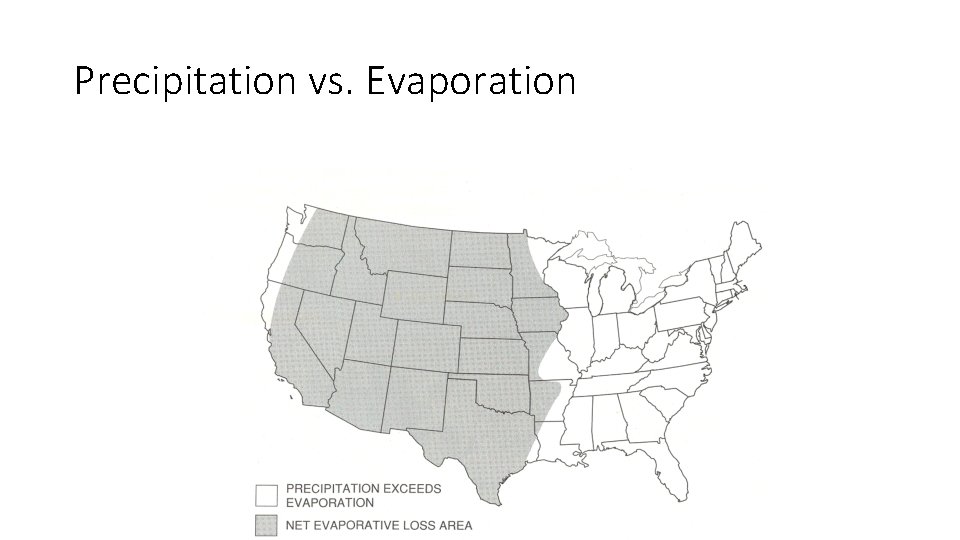
Precipitation vs. Evaporation

SOIL p. H TYPE OF VEGETATION – Vegetation affects p. H over period of years: grasslands – higher p. H forests – lower p. H

SOIL p. H The most critical effects of soil p. H are indirect. . . the availability (unavailability) of certain influenced by soil p. H nutrients is greatly

SOIL p. H Nutrients UNAVAILABLE at higher p. H: Cu – Copper Fe – Iron Mn – Manganese Zn – Zinc
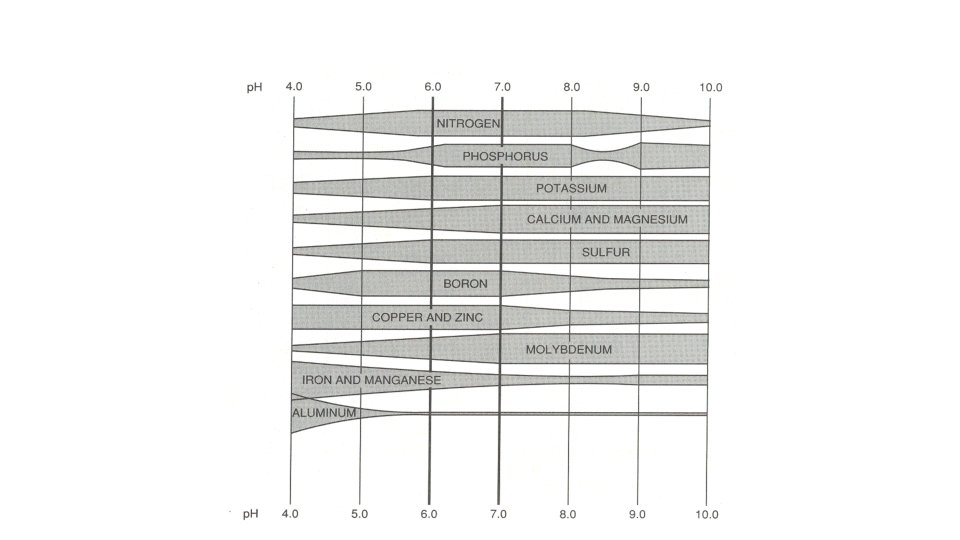

SOIL p. H additionally. . . nitrogen-fixing bacteria associated with legumes are seriously hindered in acid soils Critical with beans and peas

SOIL p. H ADJUSTING SOIL p. H - Crops vary considerably regarding soil p. H preferences
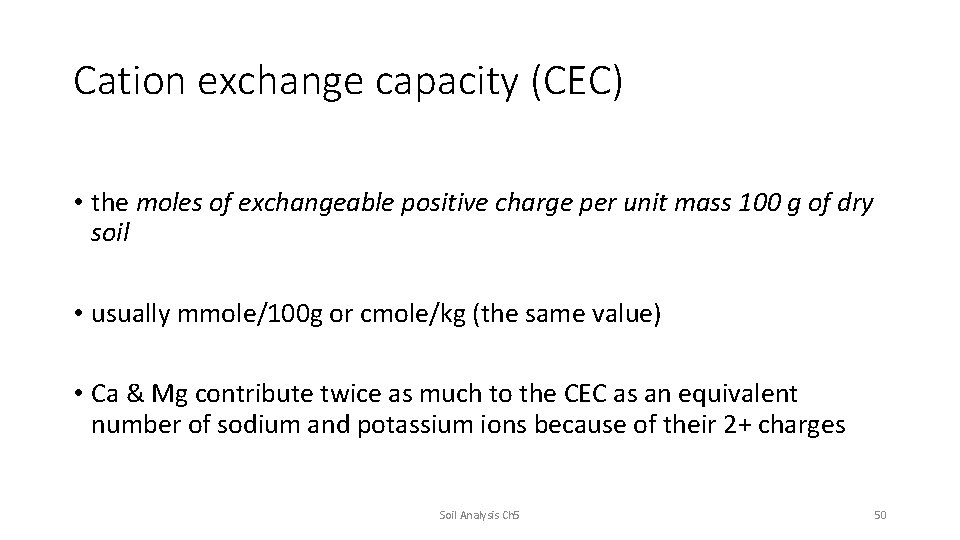
Cation exchange capacity (CEC) • the moles of exchangeable positive charge per unit mass 100 g of dry soil • usually mmole/100 g or cmole/kg (the same value) • Ca & Mg contribute twice as much to the CEC as an equivalent number of sodium and potassium ions because of their 2+ charges Soil Analysis Ch 5 50

Ion Exchange • Ions adsorbed to soil surfaces can be exchanged with ions in soil solution. • Cations and anions

Ion exchange • Organic colloids and inorganic micelles (clays) are sites of ion exchange • Where do ions in soil come from? • Release from organic matter • Rain • Weathering of parent material

Ion exchange • Exchangeable cations (on soil surfaces) cannot be removed by leaching. • Soluble cations (in solution) can be removed by leaching.

• When soil is dried… …exchangeable cations hold to adsorption sites on soil surfaces. …soluble cations (and anions) precipitate or crystallize as salts.

Examples of soluble cations precipitating

Ion exchange Exchangeable ions on soil surface trading places with ions in solution.

On soil surfaces, there are: Exchangeable and Nonexchangeable Ions : Exchangeable: Exchangeable weakly held, in contact with soil solution, ready for quick replacement. “outer sphere complex” complex Nonexchangeable: Nonexchangeable “inner sphere complex” Øadsorbed by strong bonds or held in inaccessible places Ø (e. g. , the K+ between layers of illite) Ønot part of ion exchange !

Cation exchange capacity (CEC) Sum total of exchangeable cations that a soil can adsorb. ( prevents nutrients from leaching away from roots)

CEC Expressed in: milliequivalents per 100 g (meq/100 g)

Base saturation % of exchange sites occupied by basic cations Basic cations are cations other than H+ and Al+3 Base saturation + H+ ion saturation should equal 100%

equilibrium Strive for equivalent proportions of solution and exchangeable ions. Upset equilibrium by: removal by plants leaching fertilization weathering Initiate ion exchange
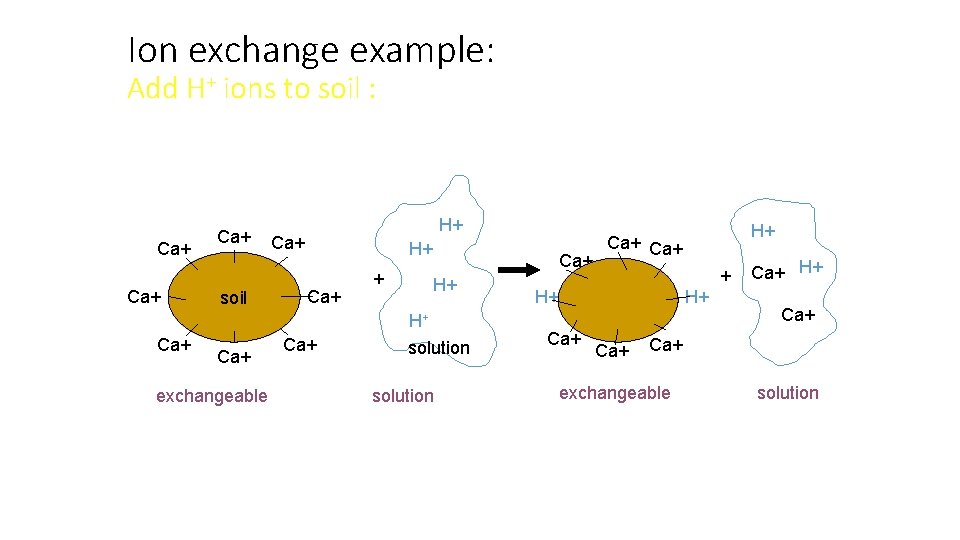
Ion exchange example: Add H+ ions to soil : Ca+ Ca+ soil Ca+ exchangeable H+ Ca+ Ca+ H+ H+ solution H+ Ca+ + H+ H+ Ca+ Ca+ H+ Ca+ exchangeable solution
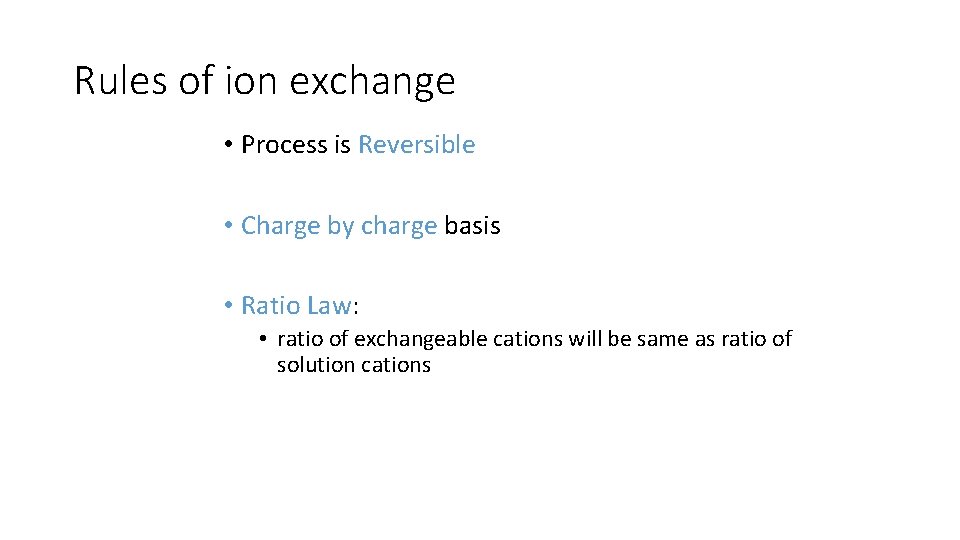
Rules of ion exchange • Process is Reversible • Charge by charge basis • Ratio Law: Law • ratio of exchangeable cations will be same as ratio of solution cations
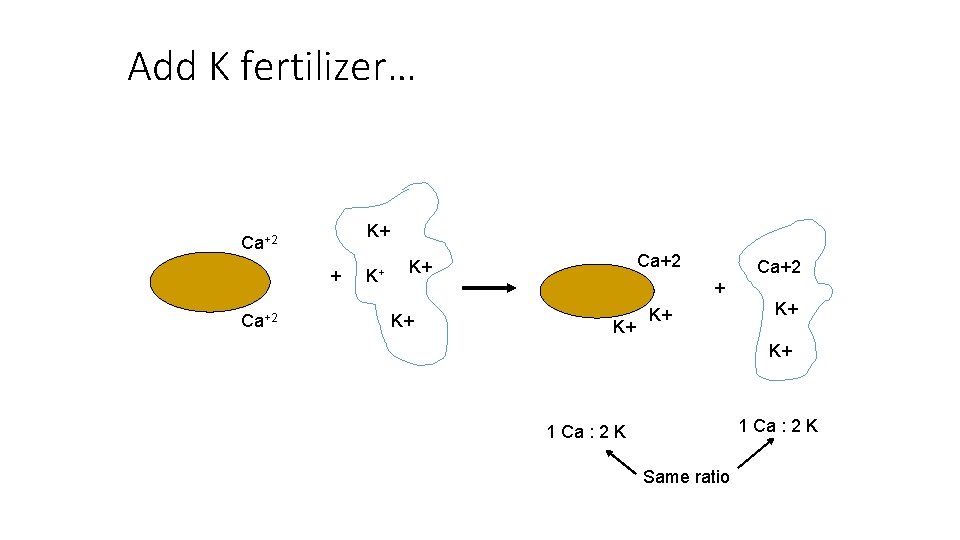
Add K fertilizer… K+ Ca+2 + K+ K+ K+ 1 Ca : 2 K Same ratio

Energy of adsorption Strong -------------------Weak Al+3 > Ca+2 > Mg+2 > [K+ = NH 4+ ] > Na+ > H+ (based on charge and hydrated radius)
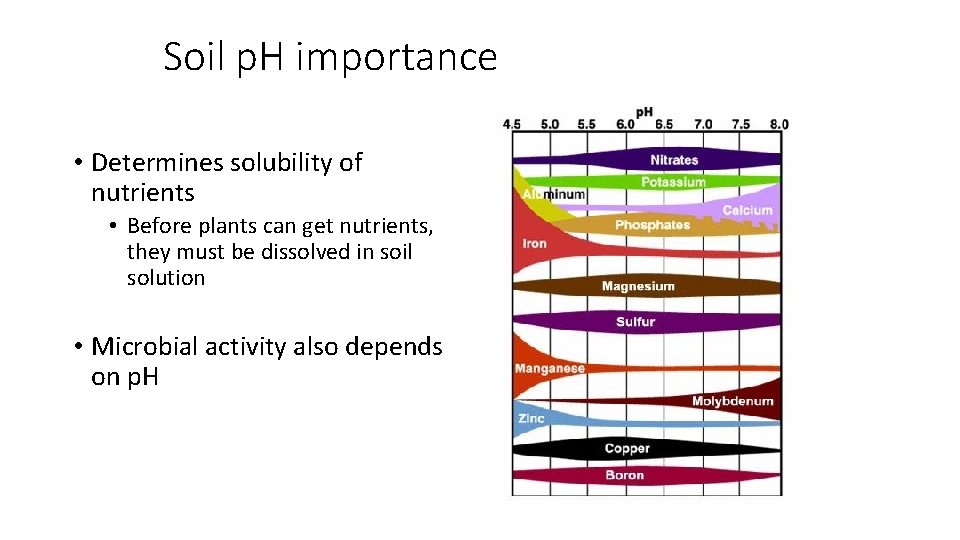
Soil p. H importance • Determines solubility of nutrients • Before plants can get nutrients, they must be dissolved in soil solution • Microbial activity also depends on p. H

p. H negative log of the hydrogen ion concentration (also a measure of OH- concentration) If H+ concentration > OH- : acidic If OH- > H+ : basic Soil p. H is p. H of solution, NOT exchange complex

General soil p. H conditions: “Slightly acid” 6. 0 – 6. 6 “Slightly basic” 7. 4 – 8. 0 “Moderately acid” 5. 0 – 6. 0 “Moderately basic” 8. 0 – 9. 0 “Strongly acid” < 5. 0 “Strongly basic” > 9. 0

In soil, both H+ and Al+3 ions produce acidity Al+3 produces H+ ions when it reacts with water. (when p. H below 6: Al+3 is the cause of acidity)

Causes of soil basicity 1. Hydrolysis of basic cations 2. Hydrolysis of carbonates

1. Hydrolysis of basic cations: (especially Ca+2, Mg+2, K+, NH 4+, Na+) (also called exchangeable bases) bases Extent to which exchangeable bases will hydrolyze depends on ability to compete with H+ ions for exchange sites. Na Na Na + Na H 2 O H Na Na + OH-

K+ and Na+ are weakly held compared to Ca+2 and Mg+2. ü Recall energy of adsorption So, K+ and Na+ are hydrolyzed easily and yield higher p. Hs.

2. Hydrolysis of carbonates (especially Ca. CO 3, Mg. CO 3, Na 2 CO 3) • As long as there are carbonates in the soil, carbonate hydrolysis controls p. H. • Calcareous soils remain alkaline because H+ ions combine with OH- to form H 2 O. • For those soils to become acid, all carbonates must be leached. • Basic cations replaced by Al+3 and H+ Ca. CO 3 + H 2 O Na 2 CO 3 + H 2 O Ca+2 + HCO 3 - + OHNa + HCO 3 - + OH- (higher p. H because Na more soluble)

Causes of soil acidity 1. Accumulation of soluble acids 2. Exchangeable acids (Al+3, H+)

1. Accumulation of soluble acids at faster rate than they can be neutralized or removed a. Carbonic acid (respiration and atmospheric CO 2) b. Mineralization of organic matter (produces organic, nitric, sulfuric acids) Precipitation increases both a and b

2. Exchangeable acids Exch. H+ or Al+3 dissociate Al+3 ties up OH- from water, releases an equivalent amount of H+ ions. Al+3 + H 2 O Al. OH+2 + H+

CEC and p. H Only 2: 1 silicate clays do not have p. H-dependent CECs. Others are p. H-dependent: 1: 1 kaolinite: low p. H: low CEC high p. H: high CEC Oxidic clays
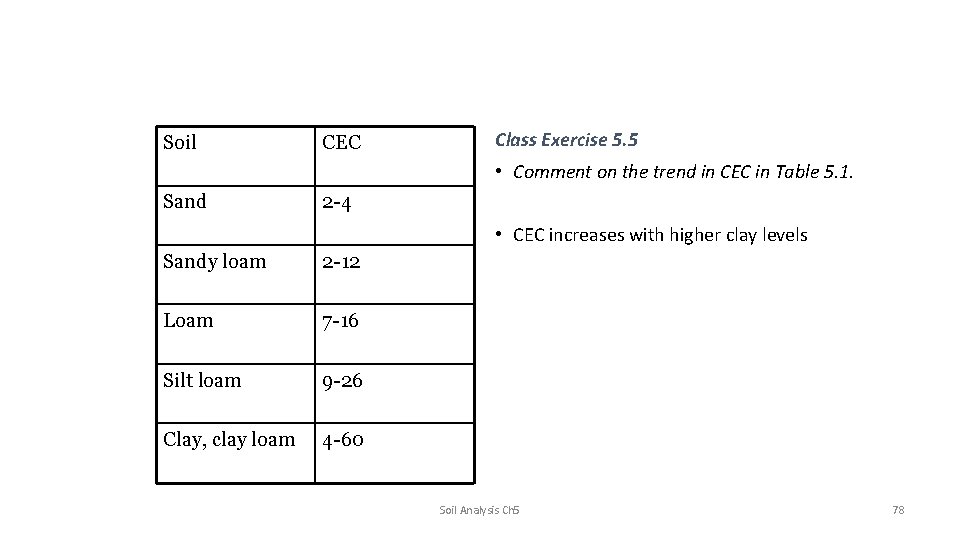
Soil CEC Class Exercise 5. 5 • Comment on the trend in CEC in Table 5. 1. Sand 2 -4 • CEC increases with higher clay levels Sandy loam 2 -12 Loam 7 -16 Silt loam 9 -26 Clay, clay loam 4 -60 Soil Analysis Ch 5 78

Significance of CEC • uptake of nutrient ions from plant roots occurs from solution only • as cations are absorbed into the roots, they are replaced in the soil solution by H+ ions • when the exchange equilibrium is disturbed, some of that ion will desorb from the soil particles • replaced by another ion • if the nutrient is a weakly adsorbed one, such as K, there may not be enough adsorbed to replenish the soil, presenting a fertility problem • K is the most likely cation to be in short supply Soil Analysis Ch 5 79

Anion exchange • the important soil anions, nitrate and phosphate, behave very different at exchange sites • nitrate and chloride are only weakly held at positive sites • more likely to be found in soil solution • phosphate and sulfate are very strongly bound to the exchange sites • phosphate can become covalently and irreversibly bound Soil Analysis Ch 5 80

Soil p. H • one of its most important properties • it affects so many other soil properties, (eg ion exchange and nutrient availability) • soil p. H comes about from a balance between acidic and alkaline species • reflects mainly the levels of dissolved H+ and OH-, but also the adsorbed H+ on cation exchange sites • normally ranges from 4 -9 Soil Analysis Ch 5 81

Sources of soil acidity • rain - polluted or fresh will be slightly acidic due to dissolved gases • microbial and root respiration – this produces CO 2, which is slightly acidic in solution • oxidation of organic matter – this produces organic acids known as humic acids, together with nitric and sulfuric acids Soil Analysis Ch 5 82

Sources of soil alkalinity • carbonate minerals – calcium and magnesium carbonate are common materials in minerals • they are slightly soluble in water, and produce OH- as they dissolve • these cations and Na & K are known as bases because of their association with alkaline soils • mineral weathering – many primary minerals as they weather release hydroxide salts of the basic cations Soil Analysis Ch 5 83

Trends in soil p. H • as soils age by weathering and leaching, they tend to become more acidic • primary minerals that release alkaline materials are replaced by neutral or slightly acidic secondary minerals • leaching removes the carbonate minerals • weathering occurs from the surface downwards so that the A and B horizons will tend to be more acidic than the C horizon Soil Analysis Ch 5 84

Significance of soil p. H • nutrient availability – the ability of plants to take up nutrients is very much dependent on the soil p. H Soil Analysis Ch 5 85

Significance of soil p. H • effect on soil organisms – soil organisms prefer different p. H levels • acid sulfate soils - soils that are rich in inorganic sulfide minerals, such as pyrites, • can lead to the formation of excessive levels of sulfuric acid through oxidation • soil p. H dives to very low levels • causes solubilisation of toxic levels of aluminium, manganese and iron from soil minerals • plant preferences – most alkaline soils; a few which need acidic soils Soil Analysis Ch 5 86

Soil p. H management • soils tend towards lower p. H values as they age • the main need for p. H management is to making the soil more alkaline • most common method by liming • agricultural lime is a mixture dominated by Ca. CO 3, but also containing Mg. CO 3 and Ca(OH)2 • comes from ground limestone, • add the nutrients calcium and magnesium to the soil • dolomite lime has a higher proportion of magnesium carbonate • to reduce p. H , add Fe, S or peat Soil Analysis Ch 5 87

Exercise 5. 9 • What factors will affect the amount of liming required? • buffering capacity • p. H Soil Analysis Ch 5 88

Redox potential (Eh) • a measure of its ability to produce oxidation or reduction of chemical species in it • the most important soil property indicated by the soil Eh is whether it is aerobic or anaerobic • aerobic soils give a positive value • the lower the value the more anaerobic the conditions • a value that is affected by soil p. H Soil Analysis Ch 5 89

SORPTION AND PRECIPITATION • Soil particles have the ability to capture different nutrients and ions. Sorption is the process in which one substance takes up or holds another. In this case, soils that have high sorption can hold a lot of extra environmental contaminents, like phosporus, onto the particles. Soil precipitation occurs during chemical reactions when a nutrient or chemical in the soil solution (water around soil particles) transforms into a solid. This is really important if soils are really salty. Soil chemists study the speed of these reactions under many different conditions. •

SOIL ORGANIC MATTER INTERACTIONS • Soil chemists also study soil organic matter (OM), which are materials derived from the decay of plants and animals. They contain many hydrogen and carbon compounds. The arrangement and formation of these compounds influence a soils ability to handle spilt chemicals and other pollutants. •
- Slides: 92