Entropy Production in a System of Coupled Nonlinear










![Limit cycle property r(t) = [1 – (1 - r 0 -2 )exp(-2 t)]-½ Limit cycle property r(t) = [1 – (1 - r 0 -2 )exp(-2 t)]-½](https://slidetodoc.com/presentation_image_h2/710a8e2c121ca407a18aaa0c57e4a8b3/image-20.jpg)



















- Slides: 48

Entropy Production in a System of Coupled Nonlinear Driven Oscillators Mladen Martinis, Vesna Mikuta-Martinis Ruđer Bošković Institute, Theoretical Physics Division Zagreb, Croatia MATH/CHEM/COMP, Dubrovnik-2006

Motivation Nonequilibrium thermodynamics of complex biological networks What are thermodynamic links between biosphere and environment? How to bring nonequilibrium thermodynamics to the same level of clarity and usefulness as equilibrium thermodynamics? Ø Energy balance analysis Ø Entropy production as a measure of Bio Env interaction

"A violent order is disorder; and a great disorder is an order. These two things are one. “ Wallace Stevens, Connoisseur of Chaos, 1942 v Non-equilibrium may be a source of order v Irrevesible processes may lead to disspative structures v Order is a result of far-from-equilibrium (dissipative) systems trying to maximise stress reduction. 3

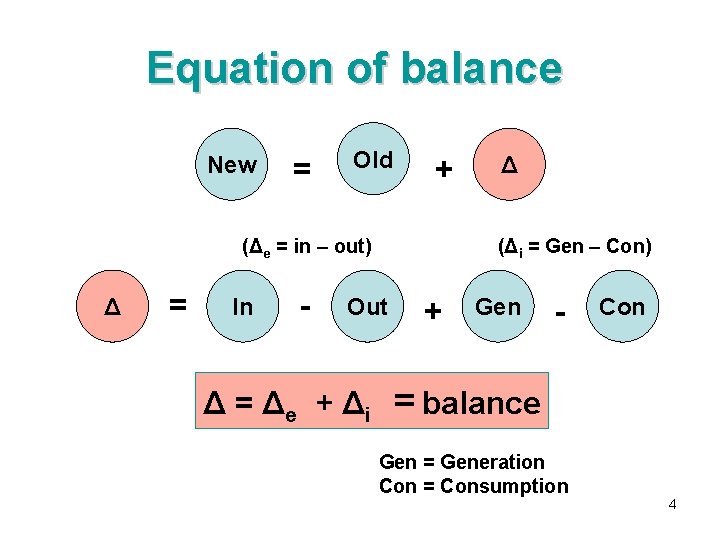
Equation of balance New = Old + (Δe = in – out) Δ = In - (Δi = Gen – Con) Out Δ = Δe + Δi Δ + Gen - Con = balance Gen = Generation Con = Consumption 4

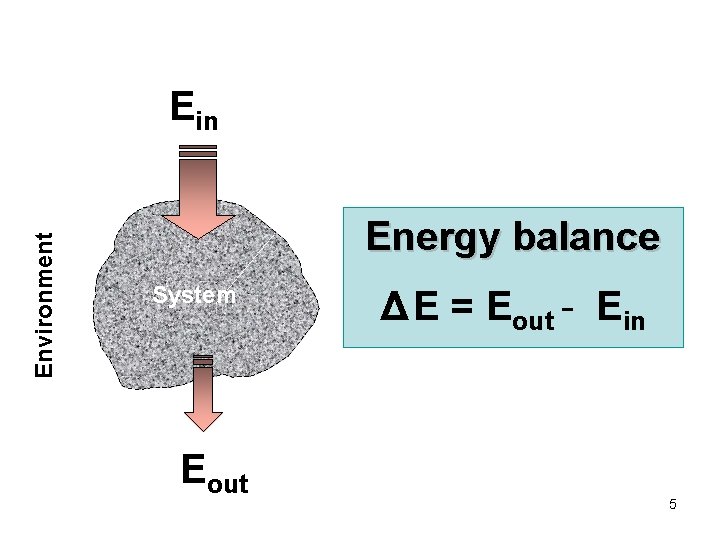
Environment Ein Energy balance System Eout Δ E = Eout - Ein 5

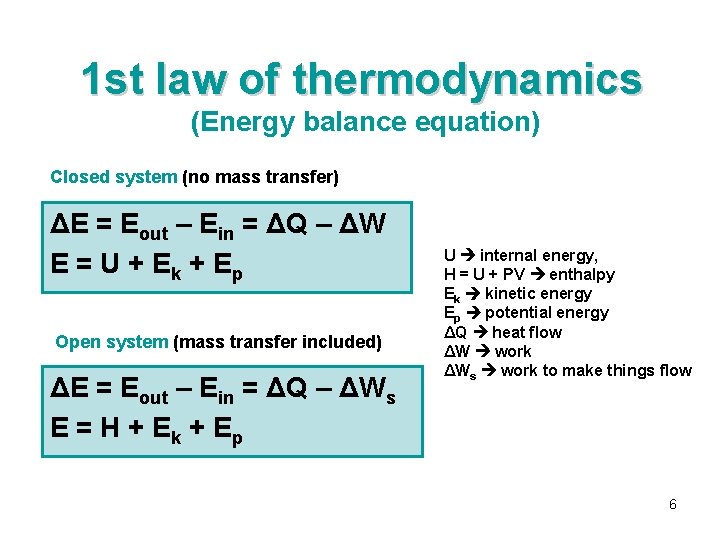
1 st law of thermodynamics (Energy balance equation) Closed system (no mass transfer) ΔE = Eout – Ein = ΔQ – ΔW E = U + E k + Ep Open system (mass transfer included) ΔE = Eout – EEin = ΔQ – ΔWs out E = H + Ek + Ep U internal energy, H = U + PV enthalpy Ek kinetic energy Ep potential energy ΔQ heat flow ΔW work ΔWs work to make things flow Energy 6

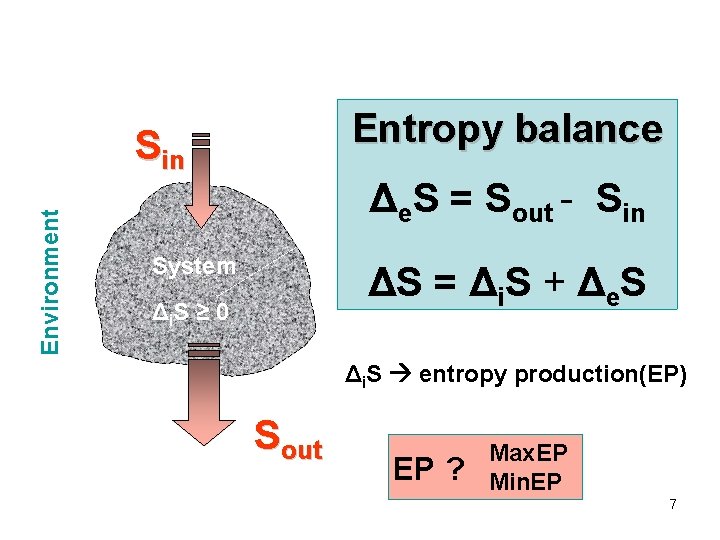
Entropy balance Environment Sin Δe. S = Sout - Sin System ΔS = Δi. S + Δe. S Δ i. S ≥ 0 Δi. S entropy production(EP) Sout EP ? Max. EP Min. EP 7

2 nd law of thermodynamics • Entropy production (di. S/dt) • d. S = de. S + di. S with di. S ≥ 0 • Entropy production includes many effects: dissipation, mixing, heat transfer, chemical reactions, . . . 8

Coupled oscillators Ø Many (quasi)periodic phenomena in physics, chemistry, biology and engineering can be described by a network of coupled oscillators. Ø The dynamics of the individual oscillator in the network, can be either regular or complicated. Ø The collective behavior of all the oscillators in the network can be extremely rich, ranging from steady state (periodic oscillations) to chaotic or turbulent motions. 9

Biological oscillators Ø It is well known that cells, tissues and organs behave as nonlinear oscillators. Ø By the evolution of the organism, they are multiple hierarchicaly and functionally interconnected → complex biological network. 10

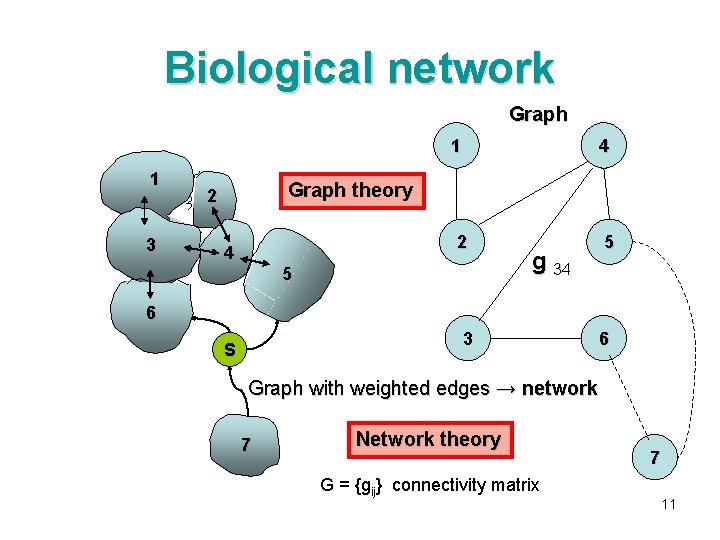
Biological network Graph 1 11 3 4 Graph theory 2 2 4 5 5 g 34 6 3 S 6 Graph with weighted edges → network 7 Network theory G = {gij} connectivity matrix 7 11

Biological homeostasis (Dynamic self-regulation) Ø Homeostasis (resistance to change) is the property of an open system, (e. g. living organisms), to regulate its internal physiological environment: Ø to maintain its stability under external varying conditions, Ø by means of multiple dynamic equilibrium adjustments, controlled by interrelated negative feedback regulation mechanisms. Ø Most physiological functions are mainteined within relatively narrow limits ( → state of physiological homeostasis). 12

What is feedback ? Ø It is a connection between the output of a system and its input ( effect is fed back to cause ). Ø Feedback can be • negative (tending to stabilise the system order) or • positive (leading to instability chaos). Ø Feedback results in nonlinearities leading to unpredictability. 13

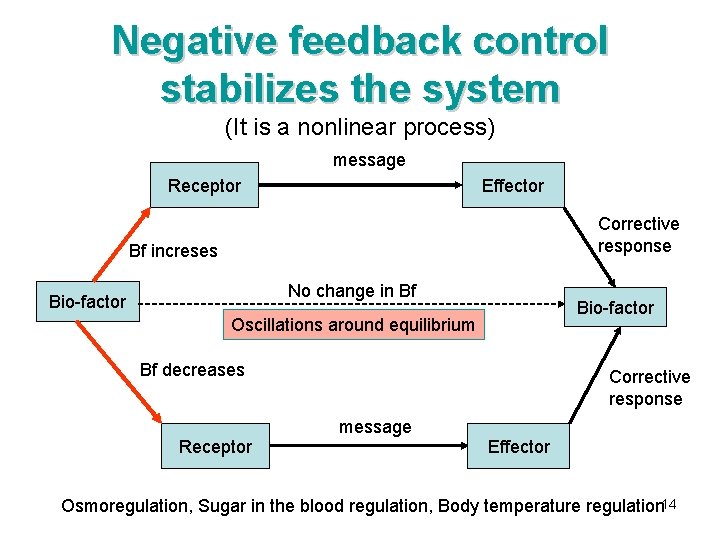
Negative feedback control stabilizes the system (It is a nonlinear process) message Receptor Effector Corrective response Bf increses No change in Bf Bio-factor Oscillations around equilibrium Bf decreases Receptor Corrective response message Effector Osmoregulation, Sugar in the blood regulation, Body temperature regulation 14

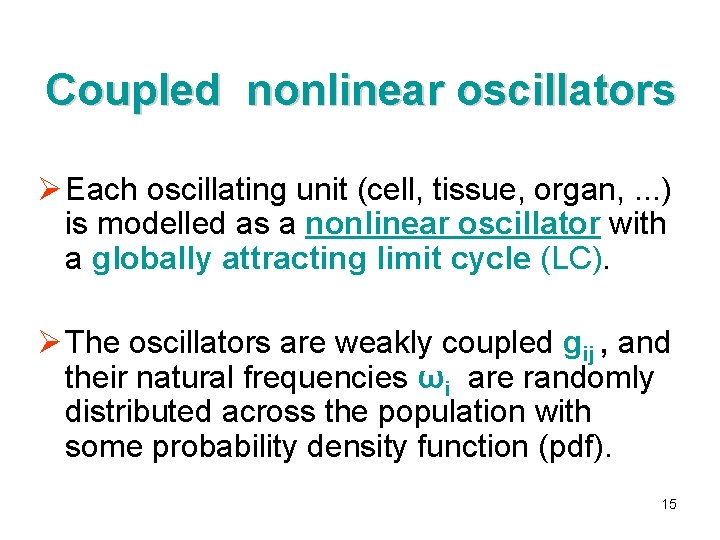
Coupled nonlinear oscillators Ø Each oscillating unit (cell, tissue, organ, . . . ) is modelled as a nonlinear oscillator with a globally attracting limit cycle (LC). Ø The oscillators are weakly coupled gij , and their natural frequencies ωi are randomly distributed across the population with some probability density function (pdf). 15

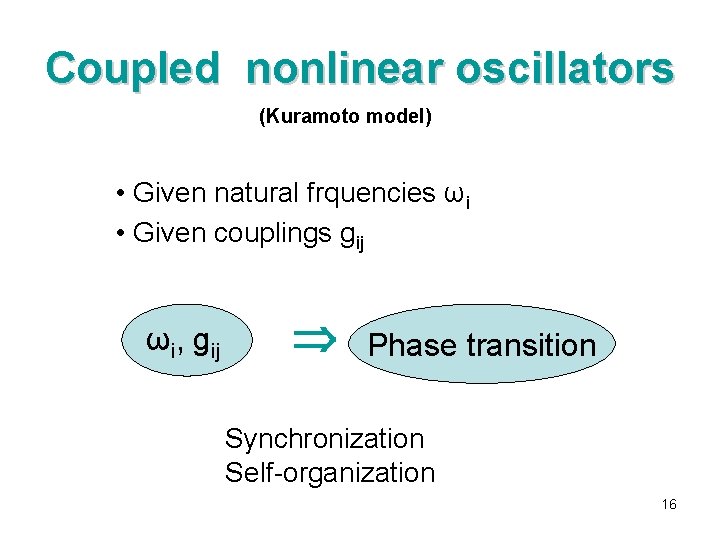
Coupled nonlinear oscillators (Kuramoto model) • Given natural frquencies ωi • Given couplings gij ωi, gij Phase transition Synchronization Self-organization 16

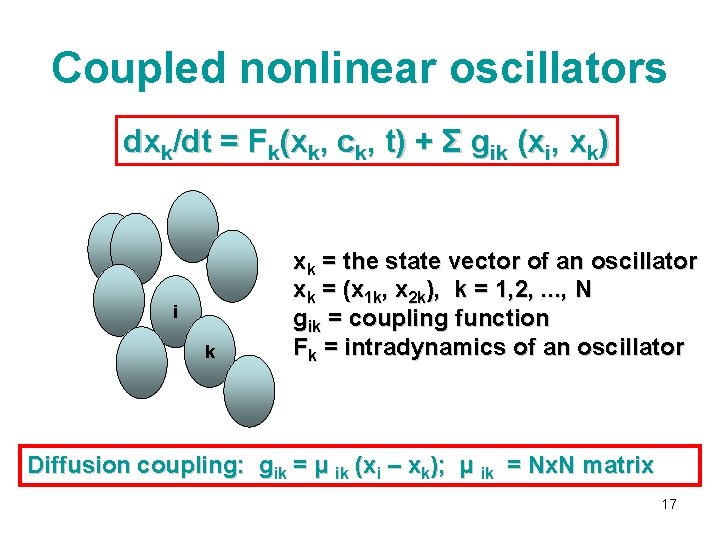
Coupled nonlinear oscillators dxk/dt = Fk(xk, ck, t) + Σ gik (xi, xk) i k xk = the state vector of an oscillator xk = (x 1 k, x 2 k), k = 1, 2, . . . , N gik = coupling function Fk = intradynamics of an oscillator Diffusion coupling: gik = μ ik (xi – xk); μ ik = Nx. N matrix 17

Complex phase space Standard phase space: s(t), sn = biological signal dts(t), sn+1 = rate of change dts(t) = f(s, t) or sn+1 = f(sn) dts sn+1 Free oscillator dt 2 s + ω2 s = 0, s(t) = Acos( ωt + φ ) Complex phase space: s, sn Im z z z(t) = ωs(t) – i dts(t) dtz(t) = F(z, z*, t) or zn+1 = F(zn, z*n) Free oscillator: dtz = i ω z , |z|2 = const z = r e iθ , dtr = 0, dt θ = ω ωs = Re z = r cosθ r z - plane Re z = r cosθ Im z = r sinθ θ Re z 18

Limit Cycle Oscillator(LCO) negative feedback effect Example of LCO: LCO dtz = (a 2 + iω - |z|2)z dtr = (a 2 – r 2)r, dt θ = ω ωs(t) = r(t)cos θ(t) r 0>a z-plane r 0<a a Limit cycle Solution: r(t) =a/u(t), θ(t) = ωt + θ 0 u(t) = [1 – (1 - u 02)exp(-2 a 2 t)]½ ωs(t) = (a/u(t))cos( ωt + θ 0), ω = 2π / T 19
![Limit cycle property rt 1 1 r 0 2 exp2 t½ Limit cycle property r(t) = [1 – (1 - r 0 -2 )exp(-2 t)]-½](https://slidetodoc.com/presentation_image_h2/710a8e2c121ca407a18aaa0c57e4a8b3/image-20.jpg)
Limit cycle property r(t) = [1 – (1 - r 0 -2 )exp(-2 t)]-½ 20

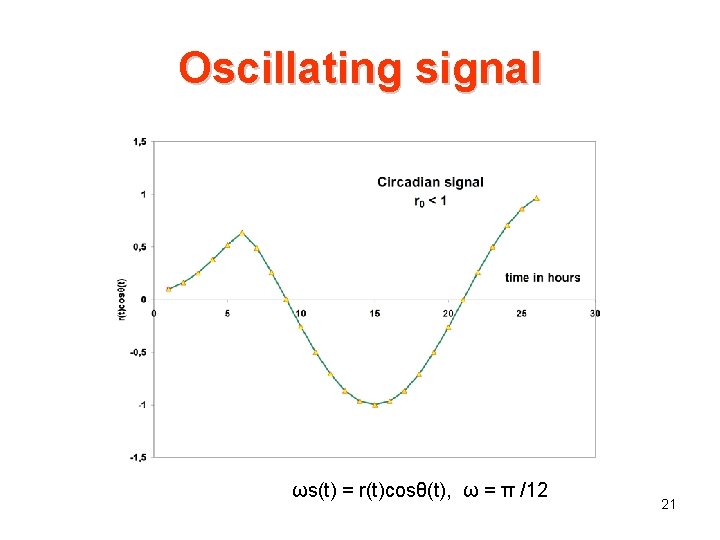
Oscillating signal ωs(t) = r(t)cosθ(t), ω = π /12 21

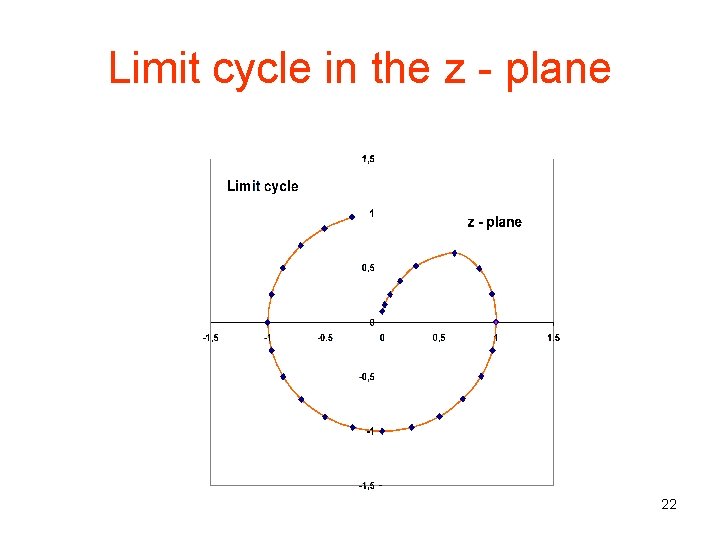
Limit cycle in the z - plane 22

Entropy production in a driven LC oscillator • Biological systems are generically out of equilibrium. • In an environment with constant temperature the source of nonequilibrium are usually mechanical (external forces) or chemical (imbalanced reactions) stimuli with stochastic character of the nonequilibrium processes. Stochastic (Langevin) description of a driven LC oscillator representing stochastic trajectory (dts(t), s(t))) in (r, θ)-phase space dtr(t) = (a 2 – r 2)r + ς(t), dtθ(t) = ω ς(t) Gaussian white noise < ς(t) ς(t’)> = 2 dδ(t – t’) 23

Non- equilibrium entropy dt Se Si(t) = - ∫rdr p(r, t) lnp(r, t) ≡ <si(t)> si(t) = - lnp(r, t), dtse(t) = dtq(t)/ T = (a 2 – r 2)r dtr, D = T dt Si p(r, t) is the probability to find the LCO in the state r dt S = d t Si + dt Se ≥ 0 p(r, t) is the solution of the Fokker-Planck equation with a given initial condition p(r, 0) = p 0(r) ∂tp(r, t) = - ∂rj(r, t) = - ∂r[(a 2 – r 2)r - D∂r]p(r, t) 24

Applications

Biological Rhythms (BRs) Ø Ø Ø BRs are observed at all levels of living organisms. BRs can occur daily, monthly, or seasonally. Circadian (daily) rhythms (CRs) vary in length from species to species (usually lasts approximately 24 hours).

Biological Clocks (BCs) Ø Biological clocks are responsible for maintaining circadian rhythms, which affect our sleep, performance, mood and more. Circadian clocks enhance the fitness of an organism by improving its ability to adapt to environmental influences, specifically daily changes in light, temperature and humidity.

Modelling circadian rhythmus as coupled oscillators 1. Blood pressure circadian 2. Heart rate circadian 3. Body temperature circadian 28

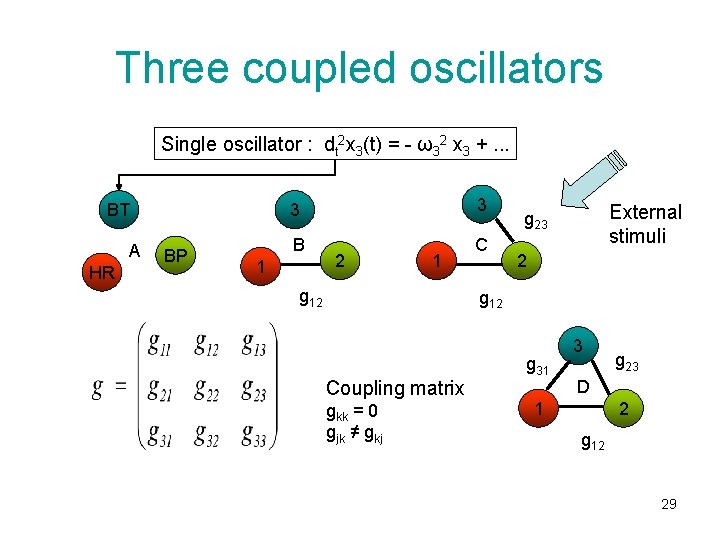
Three coupled oscillators Single oscillator : dt 2 x 3(t) = - ω32 x 3 +. . . BT A HR BP 3 3 B C 1 2 1 g 12 External stimuli g 23 2 g 12 Coupling matrix gkk = 0 gjk ≠ gkj g 31 3 g 23 D 1 2 g 12 29

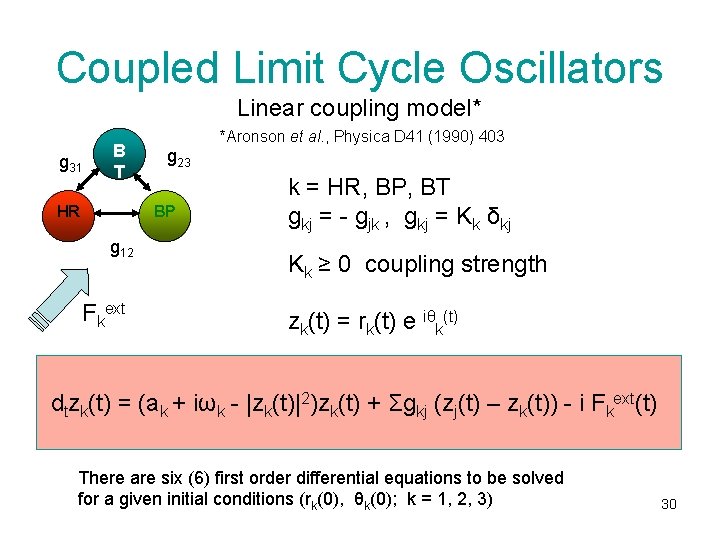
Coupled Limit Cycle Oscillators Linear coupling model* g 31 B T HR *Aronson et al. , Physica D 41 (1990) 403 g 23 BP g 12 Fkext k = HR, BP, BT gkj = - gjk , gkj = Kk δkj Kk ≥ 0 coupling strength zk(t) = rk(t) e iθk(t) dtzk(t) = (ak + iωk - |zk(t)|2)zk(t) + Σgkj (zj(t) – zk(t)) - i Fkext(t) There are six (6) first order differential equations to be solved for a given initial conditions (rk(0), θk(0); k = 1, 2, 3) 30

Consequences • Coupled limit cycle oscillator model has variety of stationary and nonstationary solutions which depend on the coupling K, the limit cycle radius a and the frequency differencies ∆kj = |ωk – ωj|. • Weak coupling (K ~ 0): the oscillators behave as independent units , subjected each to the influence of the external stimuli (Fext (t)). • With increasing coupling (K> 1) two important classes of stationary solutions are possible: – The amplitude death (r 1, r 2 or r 3 → 0 as t → ∞ ) – The frequency locking (synchronization) 31

Conclusion • We have developed a mathematical models of BP, HR and BT circadian oscillations using the coupled LC oscillators approach. • Coupled LC oscillator-model can have variety of stationary and nonstationary solutions which depend on the coupling K, the limit cycle radius a and the frequency differencies ∆kj = |ωk – ωj|. • Weakly coupled oscillators behave as independent units but with coupled phases. • They are subjected each to the influence of the external disturbancies (Fext (t)) which can change circadian organization of the organism and become an important cause of morbidity. 32

END 33

34

35

Self-organization in biological systems relies on functional interactions between populations of structural units (molecules, cells, tissues, organs, or organisms). . 36

Synchronization There are several types of synchronization : • • Phase synchronization (PS), Lag synchronization (LS), Complete synchronization (CS), and Generalized synchronization (GS) (usually observed in coupled chaotic systems) 37

Relationship between entropy and self-organization The relationship between entropy and self-organization tries to relate organization to the 2 nd Law of Thermodynamics order is a necessary result of farfrom-equilibrium (dissipative) systems trying to maximise stress reduction. This suggests that the more complex the organism then the more efficient it is at dissipating potentials, a field of study sometimes called 'autocatakinetics' and related to what has been called 'The Law of Maximum Entropy Production'. Thus organization does not 'violate' the 2 nd Law (as often claimed) but seems to be a direct result of it. 38

What are dissipative systems ? • Systems that use energy flow to maintain their form are said to be dissipative (e. g. living systems ). • Such systems are generally open to their environment. 39

Biological signals Ø Every living cell, organ, or organism generates signals for internal and external communication. Ø In-out relationship is generated by a biological process (electrochemical, mechanical, biochemical or hormonal). Ø The received signal is usually very distorted by the transmission channel in the body. 40

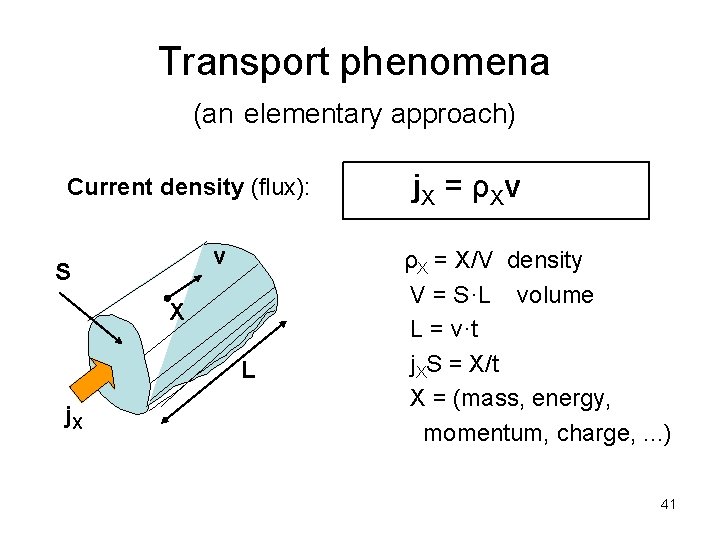
Transport phenomena (an elementary approach) Current density (flux): v S X L j. X j X = ρXv ρX = X/V density V = S·L volume L = v·t j. XS = X/t X = (mass, energy, momentum, charge, . . . ) 41

Transport phenomena (an elementary approach) • Continuity equation ∂tρX + div j. X = 0 • Transport equation j. X = - αX grad ρX αX(from kinetic theory) ~ vℓ ℓ - mean free path 42

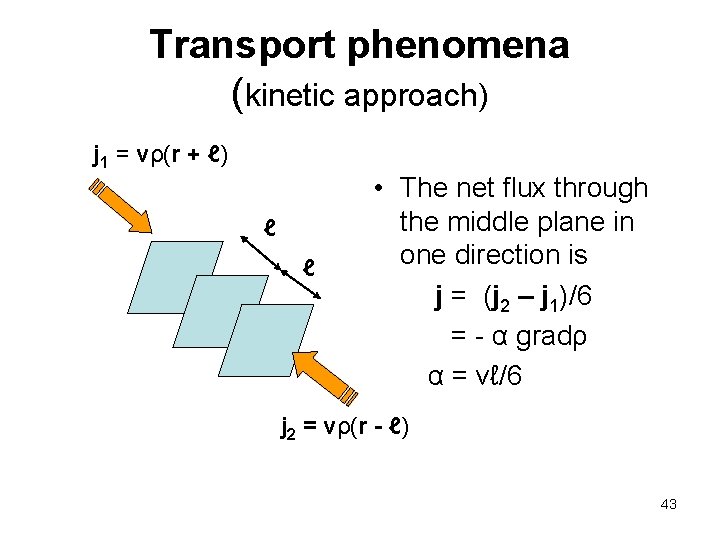
Transport phenomena (kinetic approach) j 1 = vρ(r + ℓ) ℓ ℓ • The net flux through the middle plane in one direction is j = (j 2 – j 1)/6 = - α gradρ α = vℓ/6 j 2 = vρ(r - ℓ) 43

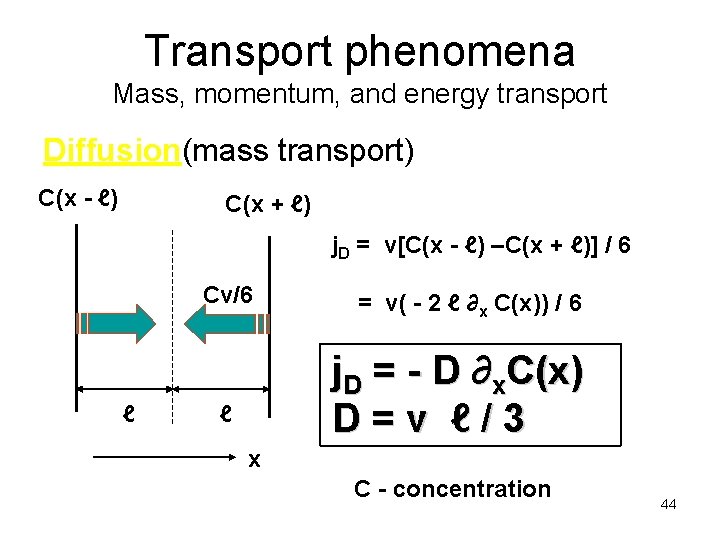
Transport phenomena Mass, momentum, and energy transport Diffusion(mass transport) C(x - ℓ) C(x + ℓ) j. D = v[C(x - ℓ) –C(x + ℓ)] / 6 Cv/6 ℓ = v( - 2 ℓ ∂x C(x)) / 6 j. D = - D ∂x. C(x) D=v ℓ/3 ℓ x C - concentration 44

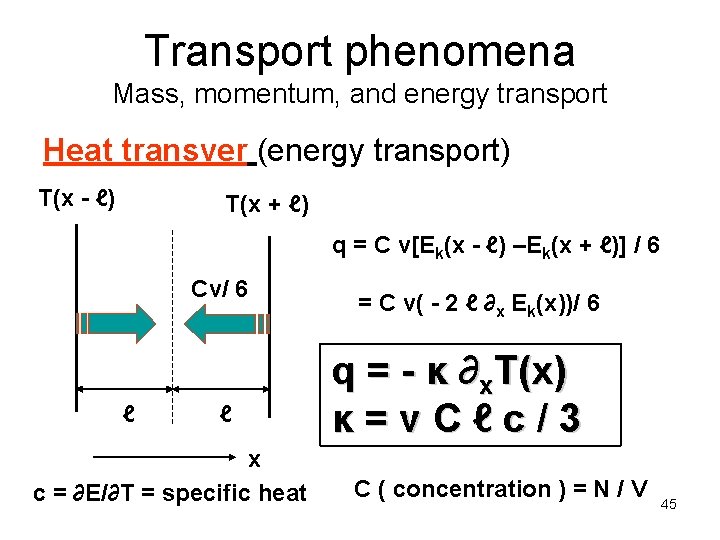
Transport phenomena Mass, momentum, and energy transport Heat transver (energy transport) T(x - ℓ) T(x + ℓ) q = C v[Ek(x - ℓ) –Ek(x + ℓ)] / 6 Cv/ 6 ℓ ℓ x c = ∂E/∂T = specific heat = C v( - 2 ℓ ∂x Ek(x))/ 6 q = - κ ∂x. T(x) κ=v. Cℓc/3 C ( concentration ) = N / V 45

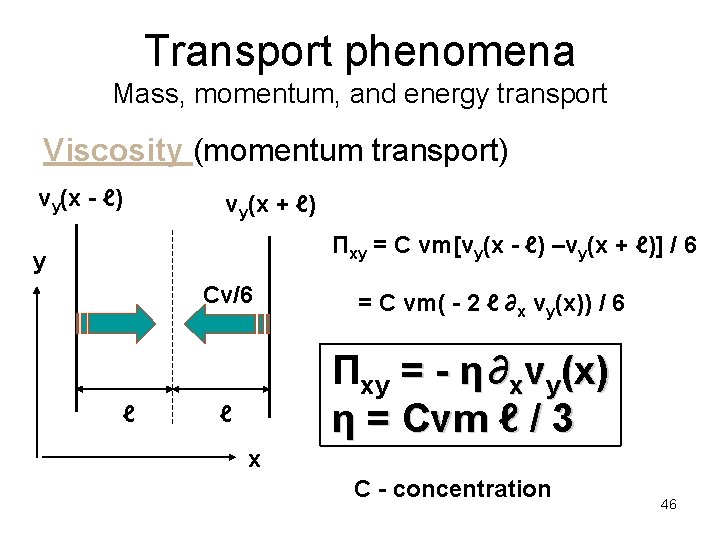
Transport phenomena Mass, momentum, and energy transport Viscosity (momentum transport) vy(x - ℓ) vy(x + ℓ) Πxy = C vm[vy(x - ℓ) –vy(x + ℓ)] / 6 y Cv/6 ℓ = C vm( - 2 ℓ ∂x vy(x)) / 6 Πxy = - η ∂xvy(x) η = Cvm ℓ / 3 ℓ x C - concentration 46

• • • • ENTROPY PRODUCTION At the very core of the second law of thermodynamics we find the basic distinction between “reversible” and “irreversible processes” (1). This leads ultimately to the introduction of entropy S and the formulation of the second law of thermodynamics. The classical formulation due to Clausius refers to isolated systems exchanging neither energy nor matter with the outside world. The second law then merely ascertains the existence of a function, the entropy S, which increases monotonically until it reaches its maximum at the state of thermodynamic equilibrium, (2. 1) It is easy to extend this formulation to systems which exchange energy and matter with the outside world. (see fig. 2. 1). Fig. 2. 1. The exchange of entropy between the outside and the inside. 47

• • • • To extend thermodynamics to non-equilibrium processes we need an explicit expression for the entropy production. Progress has been achieved along this line by supposing that even outside equilibrium entropy depends only on the same variables as at equilibrium. This is the assumption of “local” equilibrium (2). Once this assumption is accepted we obtain for P, the entropy production per unit time, (2. 3) : dt. Si = Σ Jα Fα where the Jp are the rates of the various irreversible processes involved (chemical reactions, heat flow, diffusion. . . ) and the F the corresponding generalized 266 Chemistry 1977 forces (affinities, gradients of temperature, of chemical potentials. . . ). This is the basic formula of macroscopic thermodynamics of irreversible processes. 48