Entropy of an Ideal Monatomic Gas 1 We














- Slides: 15

Entropy of an Ideal Monatomic Gas 1 We wish to find a general expression ω(U, V, N) for a system of N weakly-interacting particles of an ideal monatomic gas, confined to a volume V, with the total energy in the range U to U + U. Since U = p 2/2 m, the total momentum lies in the ranges ± p to ± (p + p). In order to count microstates, we imagine the phase-space to be divided into cells of “area” ΔpiΔqi ≈ h, per degree of freedom. N particles moving in 3 dimensions is represented by a point in 6 N-dimensional phase space, while the number of degrees of freedom of the system is f = 3 N. Note that f may be more than 3 N if a particle is vibrating in simple harmonic motion, since degrees of freedom are associated both 1 with kinetic energy and with vibrational potential energy.

Entropy of an Ideal Monatomic Gas 2 A state describing N particles moving in 3 dimensions is a point in 6 N-dimensional phase space, The number of degrees of freedom of the system is f = 3 N. For f = 1 (one particle moving in one dimension), the relation U = p 2/2 m means that p lies between the values ± p to ± (p + p). Thus ω(U, V, N) = 2 p L/h, where is the length of the container, 2 so that ω L = V 1/3 and ω p 0 U 0.

Entropy of an Ideal Monatomic Gas 3 • f = 1 (1 atom in 1 dimension) ω is the number of squares contained in the two rectangles; i. e. ω = 2 p L/h p 0 L U 0 V 1/3. • f = 2 (1 atom in 2 dimensions or 2 atoms in 1 dimension) ω is the number of squares contained in the area between the circles; i. e. ω = 2πp p L 2/h 2 p 1 L 2 U 1/2 V 2/3. • f = 3 (1 atom in 3 dimensions or 3 atoms in 1 dimension) ω is the number of squares contained in the area between the spheres; i. e. ω = 4πp 2 p L 3/h 3 p 2 L 3 U 1 V. 3

Entropy of an Ideal Monatomic Gas 4 • General result for f degrees of freedom ω p(f – 1) Lf or p(f – 1) Vf/3. ω (m. U)(f – 1)/2 Vf/3 = (m. U)(3 N – 1)/2 VN ≈ (m. U)3 N/2 VN for large N. Thus ω(U, V, N) = A(N) m 3 N/2 U 3 N/2 VN For a single species of mass m, we may write B(N) = A(N) m 3 N/2. Thus ω(U, V, N) = B(N) VN U 3 N/2, so that S(U, V, N) = k [C(N) + N ln. V + (3 N/2) ln. U]. Assuming that S is an extensive function, we have S(U, V, N) = N s(U, V) = Nk [K + lnv + (3/2) ln u]. Using the results u =(3/2)k. T and v = V/N, we obtain S(U, V, N) = Nk [D + (3/2)ln. T + ln(V/N)]. 4

Mixing of gases 1: Gibbs’ Paradox If N molecules of an ideal monatomic gas make a free expansion from a V to 2 V, ΔU = 0, so that ΔS = Sf – Si = Nk ln(2 V/V) = Nk ln 2. If the container of volume 2 V is now redivided into equal parts, so that (N/2) molecules are in each half, then ΔS’ = Sf’ – Si’ = 2(N/2)k ln(V/2 V) = – Nk ln 2. This is Gibbs’ paradox, which is removed by assuming that the molecules are indistiguishable; i. e. that ω(U, V, N) = B(N) U 3 N/2 VN/N!. The term Nk ln. V in the expression for S is replaced by Nk ln(V/N!), where (by Stirling’s theorem), 5 Nk ln(V/N!) ≈ Nk [ln(V/N) + 1].

Entropy of an Ideal Monatomic Gas 5 • If S is assumed to be an extensive quantity, Gibbs’ paradox may be avoided. • Letting S = Ns, with s(U, V) = k [ln. C + lnv +(3/2) lnu], where C = B(N)/N, v = V/N, and u = U/N. • Thus, S(U, V, N) = Nk [ln. C + (3/2)ln(U/N) + ln(V/N)]. • Temperature is defined as 1/T ( S/ U)V, N = (3/2)Nk/U, since U = (3/2)Nk. T, so that S(U, V, N) = Nk [D + (3/2)ln. T + ln(V/N)]. • Note that P/T = ( S/ V)T, N = Nk/V, so that PV = Nk. T. Mixing gases a. Gas A in the left compartment, a different gas B in the right compartment. ΔStot = ΔS 1 + ΔS 2. b. Gas A in the left compartment, the same gas B in the right compartment. ΔStot = Sfinal – S 1 initial – S 2 initial. 6

Partition Function of an Ideal Monatomic Gas 1 z = i exp(– i) → d 3 x d 3 p exp(– )/h 3 (classical system). 7

Partition Function of an Ideal Monatomic Gas 2 8

Entropy of an Ideal Monatomic Gas 6 Z =z. N/N!, where z = V(2πmk. T/h 2)3/2, and U = (3/2)N k. T. Also S = k(ln. Z + U) = k [ln z. N – ln. N! + (3/2)N], = Nk [lnz – ln. N + 1 + 3/2]. Thus S = Nk [K + ln(V/N) + (3/2) ln T], with K = ln (2πmk/h 2)3/2 + 5/2. 9

Equipartition Theorem 1 10

Equipartition Theorem 2 11

Degrees of Freedom • Applying the equipartition theorem is simplified by using the concept of degrees of freedom f, which is the minimum number of independent coordinates needed to the specify the motion of a system of particles. * • For a single atom (assumed to be a point), there are 3 directions of motion so that f =3. • For a molecule consisting of n point-atoms, it is necessary to separate translational, rotational and vibrational motions; however the total number of degrees of freedom must be 3 f. • For linear molecules, ftrans= 3, frot= 2 (since there can be no rotation about the axis for point-atoms), so that fvib= 3 n – 5. • For non-linear molecules, ftrans= 3, frot= 3, so that fvib= 3 n – 6. * Beware! Some texts, such as Carter, define f differently.

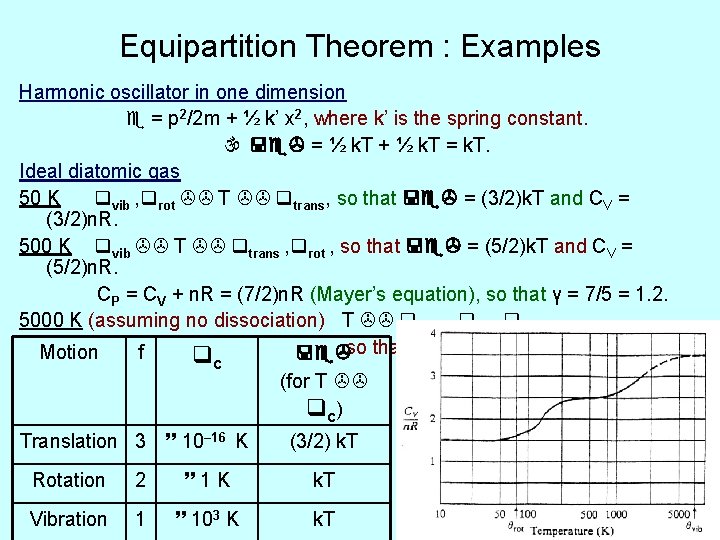
Equipartition Theorem : Examples Harmonic oscillator in one dimension = p 2/2 m + ½ k’ x 2, where k’ is the spring constant. = ½ k. T + ½ k. T = k. T. Ideal diatomic gas 50 K vib , rot T trans, so that = (3/2)k. T and CV = (3/2)n. R. 500 K vib T trans , rot , so that = (5/2)k. T and CV = (5/2)n. R. CP = CV + n. R = (7/2)n. R (Mayer’s equation), so that γ = 7/5 = 1. 2. 5000 K (assuming no dissociation) T trans , rot , vib , so that CV = (7/2)n. R. Motion f c (for T c) Translation 3 10– 16 K Rotation Vibration 2 1 K 1 103 K (3/2) k. T 13

Harmonic Oscillator in One Dimension 1 For a 1 D harmonic oscillator, n = (n + ½) ħω, where ω = √(k/m). 14

Harmonic Oscillator in One Dimension 2 15