Entropy heat engine Lets consider the efficiency of









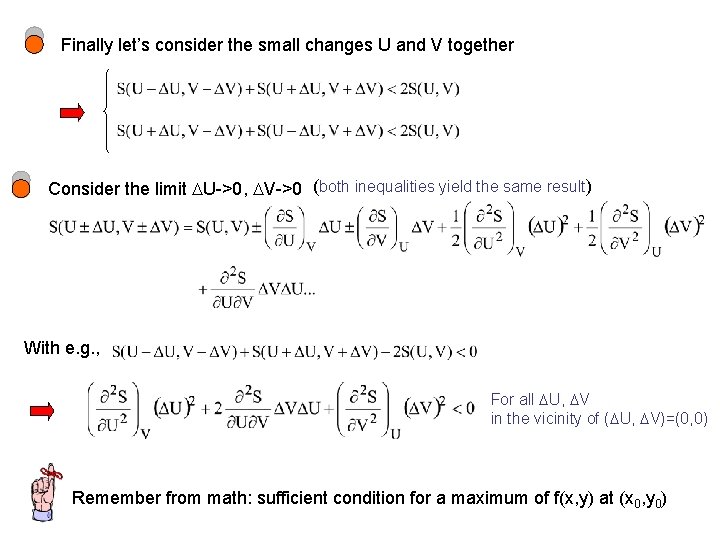
- Slides: 40

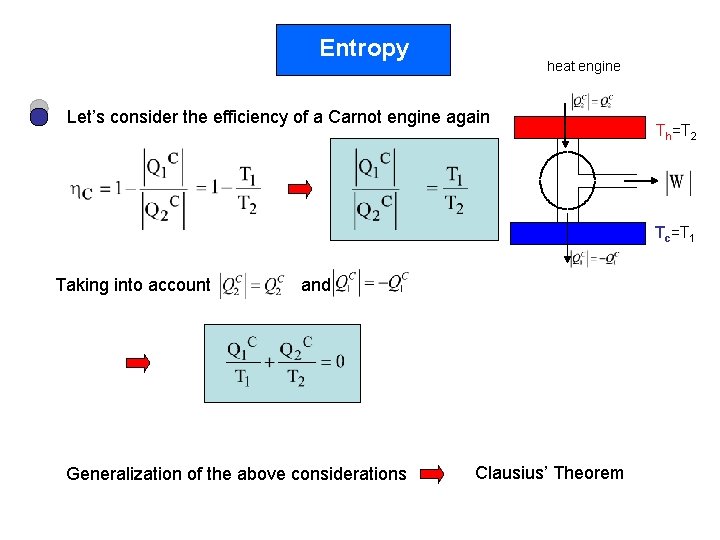
Entropy heat engine Let’s consider the efficiency of a Carnot engine again Th=T 2 Tc=T 1 Taking into account and Generalization of the above considerations Clausius’ Theorem

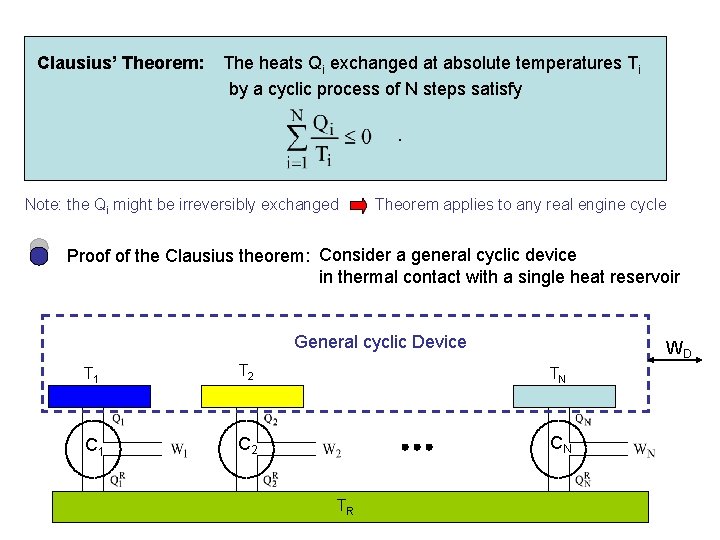
Clausius’ Theorem: The heats Qi exchanged at absolute temperatures Ti by a cyclic process of N steps satisfy. Note: the Qi might be irreversibly exchanged Theorem applies to any real engine cycle Proof of the Clausius theorem: Consider a general cyclic device in thermal contact with a single heat reservoir General cyclic Device WD T 1 T 2 TN C 1 C 2 CN TR

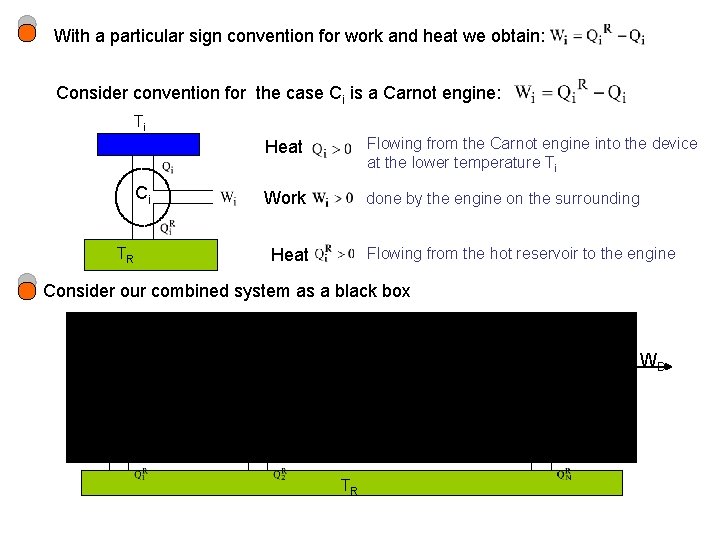
With a particular sign convention for work and heat we obtain: Consider convention for the case Ci is a Carnot engine: Ti Ci Heat Flowing from the Carnot engine into the device at the lower temperature Ti Work done by the engine on the surrounding TR Flowing from the hot reservoir to the engine Heat Consider our combined system as a black box General cyclic Device T 1 T 2 TN C 1 C 2 CN TR WD

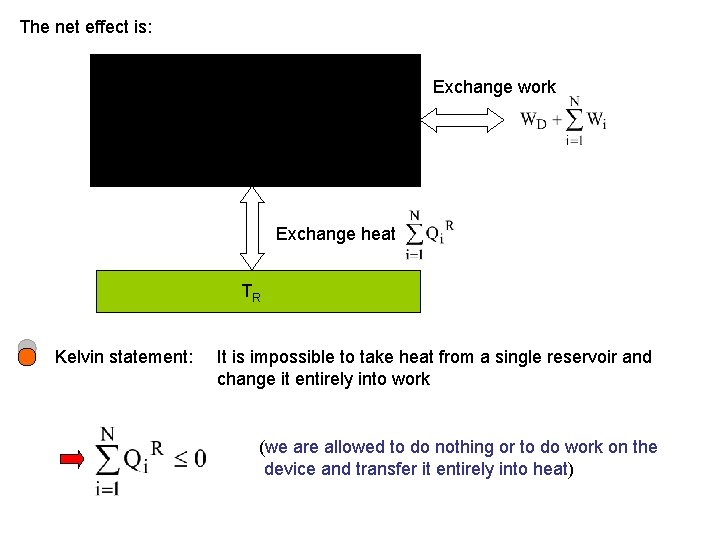
The net effect is: Exchange work Exchange heat TR Kelvin statement: It is impossible to take heat from a single reservoir and change it entirely into work (we are allowed to do nothing or to do work on the device and transfer it entirely into heat)

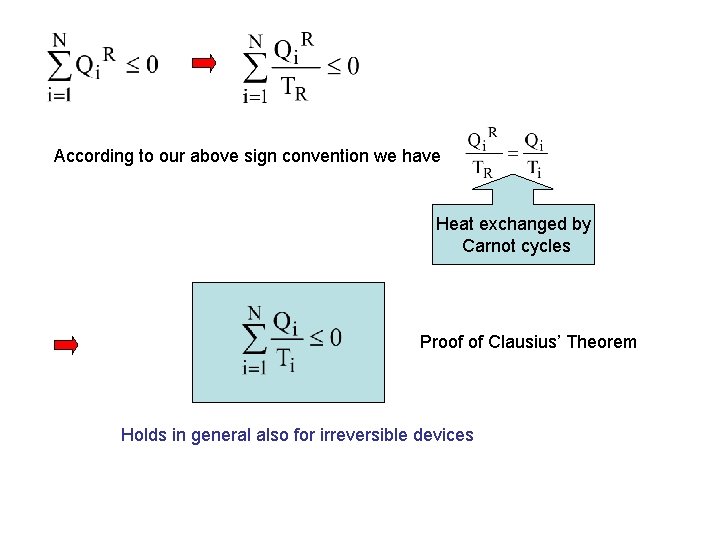
According to our above sign convention we have Heat exchanged by Carnot cycles Proof of Clausius’ Theorem Holds in general also for irreversible devices

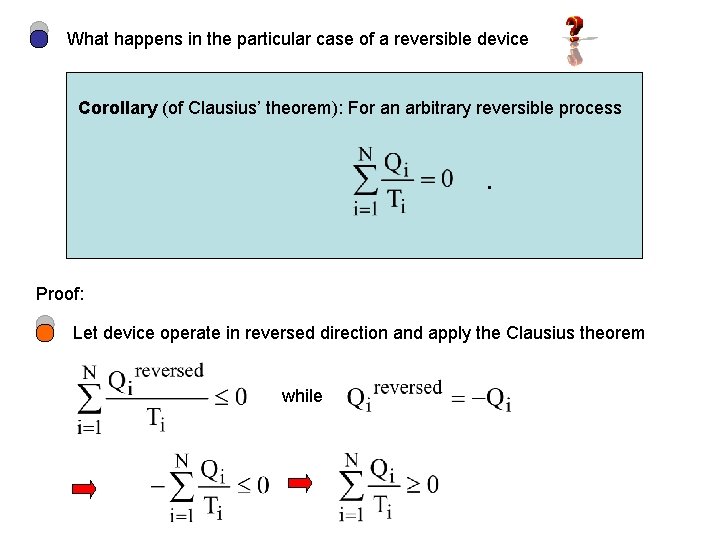
What happens in the particular case of a reversible device Corollary (of Clausius’ theorem): For an arbitrary reversible process . Proof: Let device operate in reversed direction and apply the Clausius theorem while

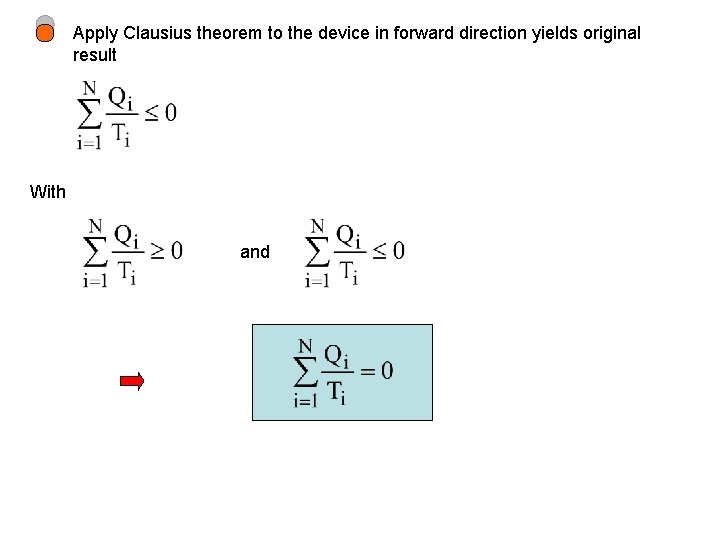
Apply Clausius theorem to the device in forward direction yields original result With and

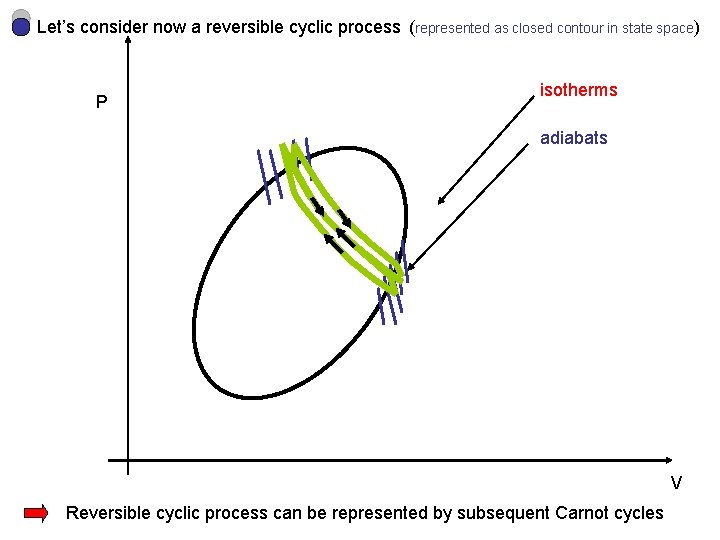
Let’s consider now a reversible cyclic process (represented as closed contour in state space) P isotherms adiabats V Reversible cyclic process can be represented by subsequent Carnot cycles

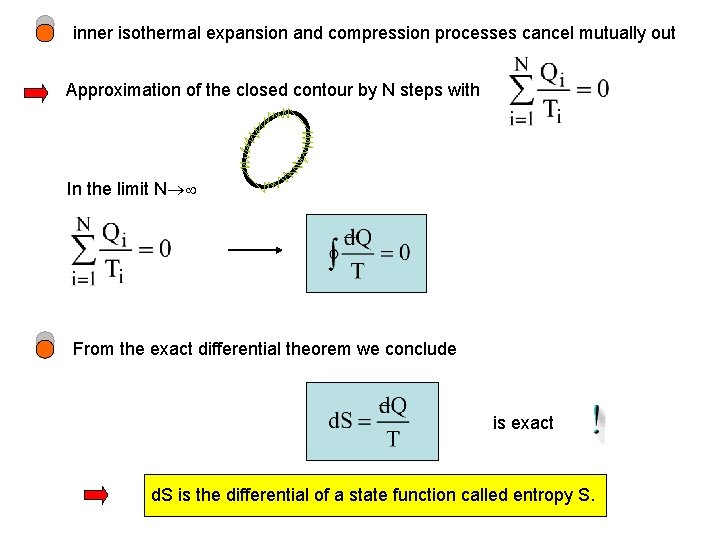
inner isothermal expansion and compression processes cancel mutually out Approximation of the closed contour by N steps with In the limit N From the exact differential theorem we conclude is exact d. S is the differential of a state function called entropy S.

Entropy determined up to an additive constant ( which will be specified later by the 3 rd law) Since is extensive, S and d. S are extensive In some cases an inexact differential can be changed into an exact one by multiplying with an integrating factor is the integrating factor which makes exact (for more info go to ) Exact differentials and theory of differential equations Consequences of Existence of Entropy Relationship between the internal energy and the equation of state

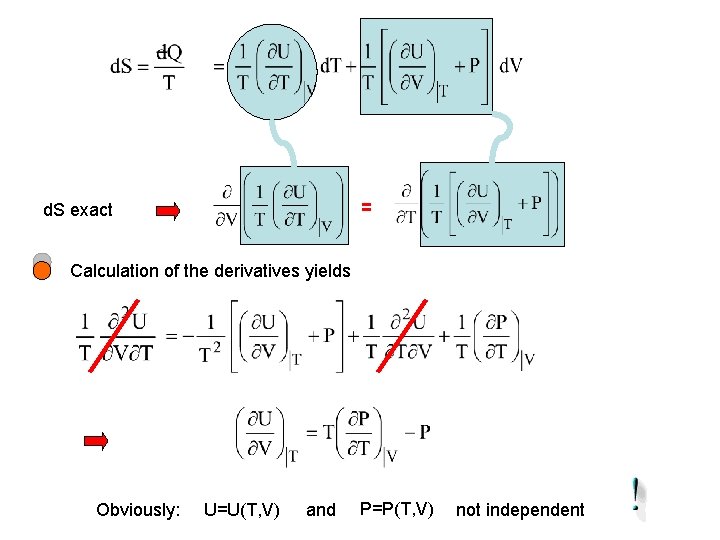
= d. S exact Calculation of the derivatives yields Obviously: U=U(T, V) and P=P(T, V) not independent

Example: We show for the ideal gas: is a consequence of U=U(T) 1 U=U(T) From 2 U=U(T) is derived from U independent of V U=U(T)

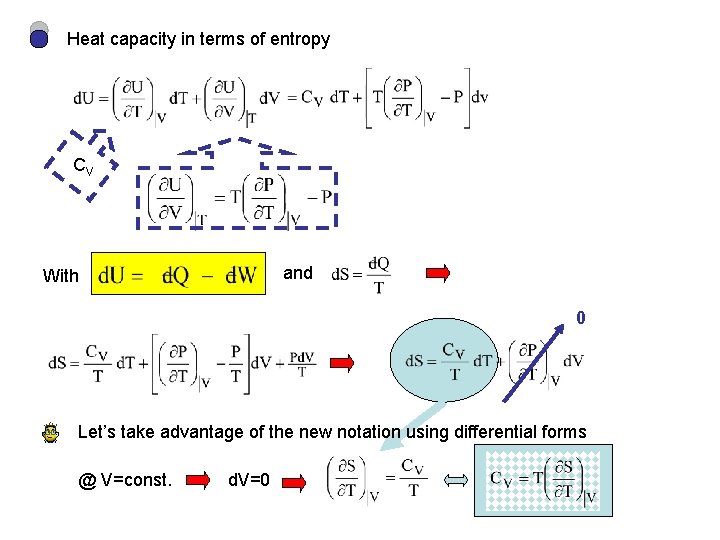
Heat capacity in terms of entropy CV and With 0 Let’s take advantage of the new notation using differential forms @ V=const. d. V=0

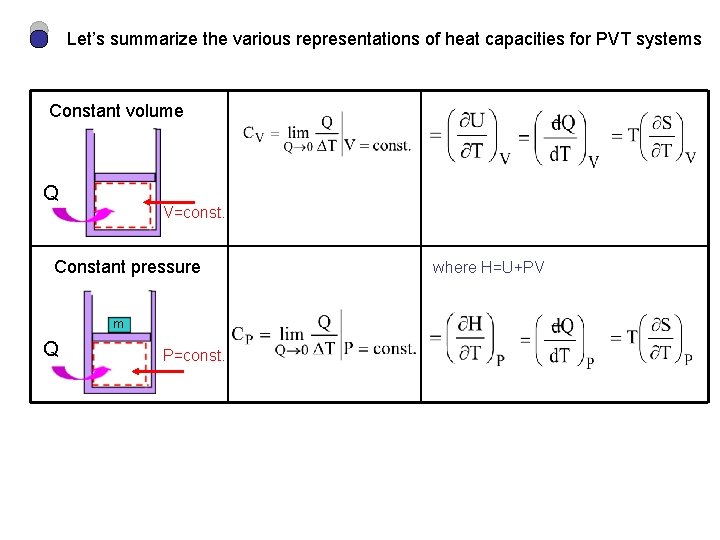
Let’s summarize the various representations of heat capacities for PVT systems Constant volume Q V=const. Constant pressure m Q P=const. where H=U+PV

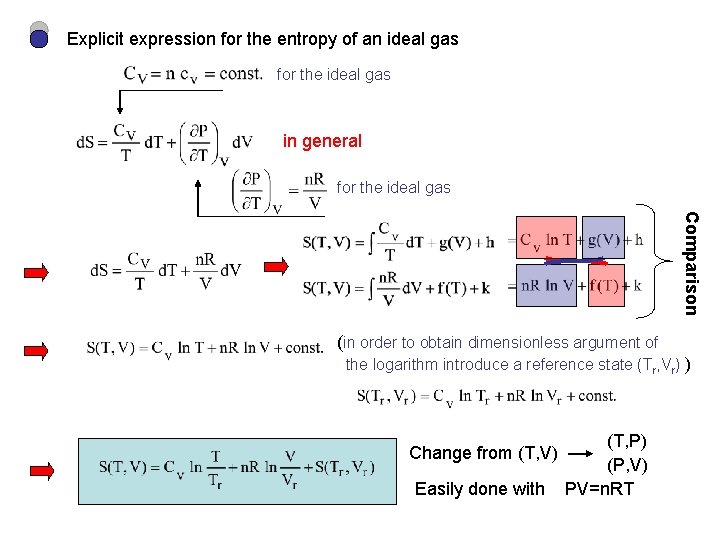
Explicit expression for the entropy of an ideal gas for the ideal gas in general for the ideal gas Comparison (in order to obtain dimensionless argument of the logarithm introduce a reference state (Tr, Vr) ) Change from (T, V) Easily done with (T, P) (P, V) PV=n. RT

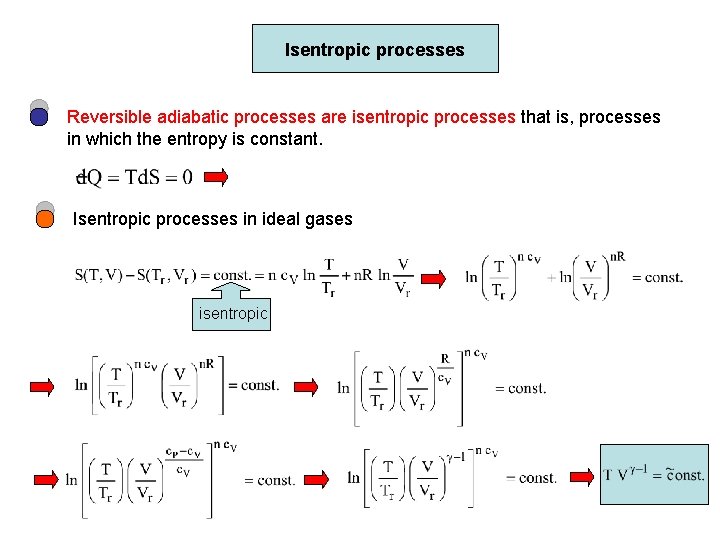
Isentropic processes Reversible adiabatic processes are isentropic processes that is, processes in which the entropy is constant. Isentropic processes in ideal gases isentropic

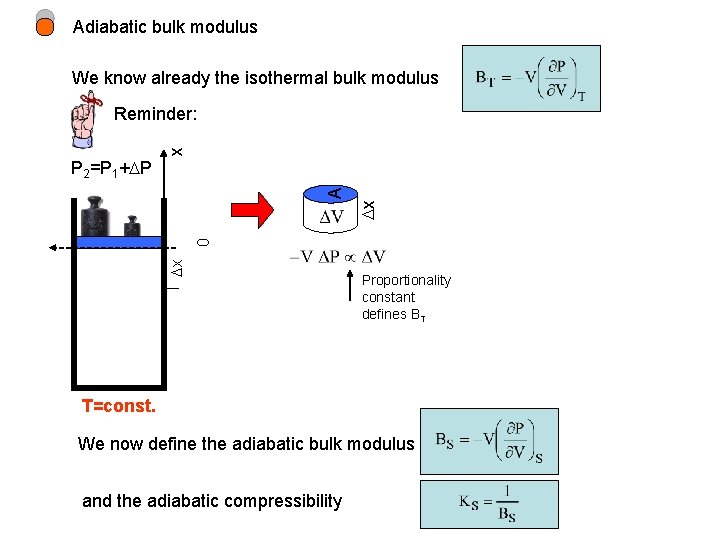
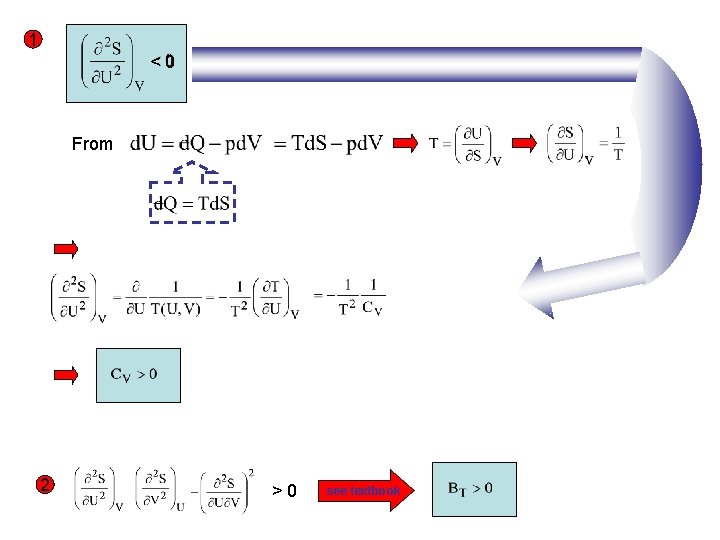
Adiabatic bulk modulus We know already the isothermal bulk modulus x Reminder: x 0 x A P 2=P 1+ P Proportionality constant defines BT T=const. We now define the adiabatic bulk modulus and the adiabatic compressibility

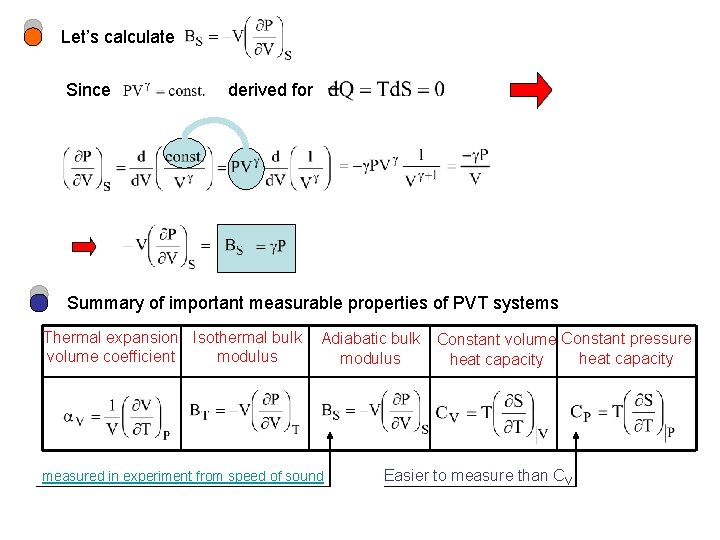
Let’s calculate Since derived for Summary of important measurable properties of PVT systems Thermal expansion Isothermal bulk volume coefficient modulus Adiabatic bulk modulus measured in experiment from speed of sound Constant volume Constant pressure heat capacity Easier to measure than CV

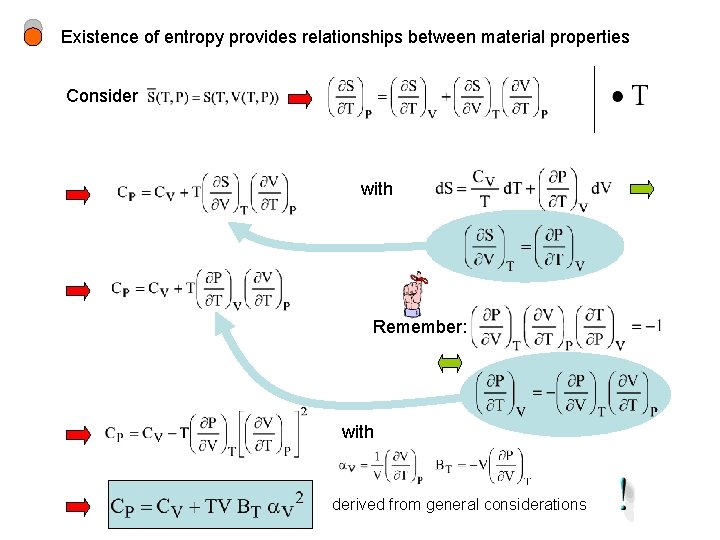
Existence of entropy provides relationships between material properties Consider with Remember: with derived from general considerations

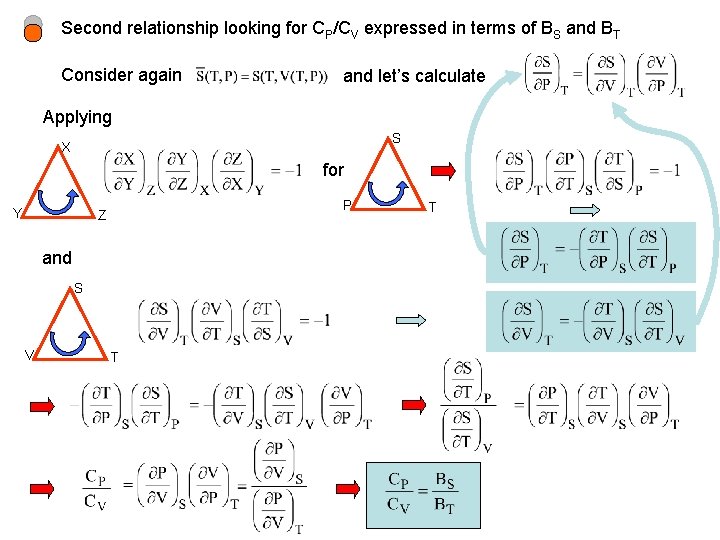
Second relationship looking for CP/CV expressed in terms of BS and BT Consider again and let’s calculate Applying S X for Y P Z and S V T T

Entropy and the Second Law We are going to show the meaning of the sloppy phrase: Entropy never decreases Therefore: Reminder: Clausius´ theorem In a cyclic process of N steps Heat exchanged @ absolute temperatures How to calculate the entropy change involved in a real (irreversible) process Answer: Find a reversible equilibrium process that takes the system between the initial and the final states of the actual processes.

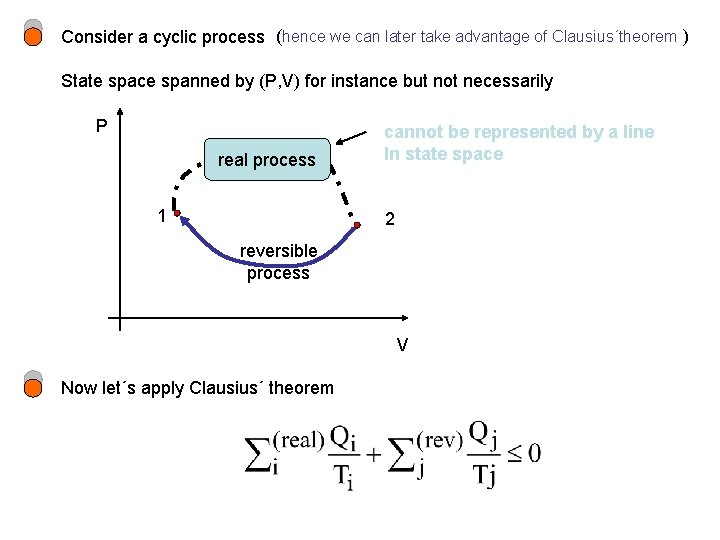
Consider a cyclic process (hence we can later take advantage of Clausius´theorem ) State space spanned by (P, V) for instance but not necessarily P real process 1 cannot be represented by a line In state space 2 reversible process V Now let´s apply Clausius´ theorem

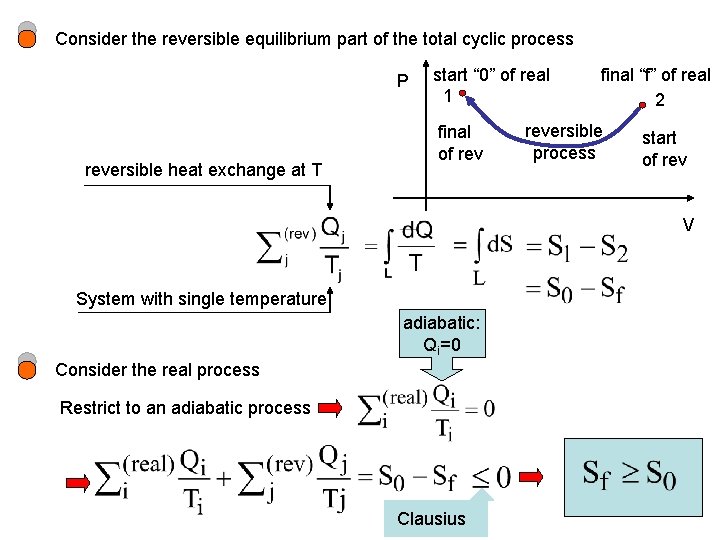
Consider the reversible equilibrium part of the total cyclic process P reversible heat exchange at T start “ 0” of real 1 final of rev final “f” of real 2 reversible process start of rev V System with single temperature adiabatic: Qi=0 Consider the real process Restrict to an adiabatic process Clausius

Entropy statement of the second law: The total entropy of an adiabatically isolated system never decreases. The idea that entropy will increase, if it can is emphasized in Clausius summary of the second law: The entropy of the universe* tends toward a maximum. *Universe is a synonym for a completely isolated system (in contrast to adiabatically isolated systems of the above statement )

Clausius statement and the principle of increase of entropy adiabatically isolated -Entropy change of the high temperature bath. -Entropy change of the low temperature bath. Heat is leaving the low temperature bath Total entropy change because Existence of a non-Clausius device violates entropy statement of 2 nd law

Kelvin statement and the entropy statement of the 2 nd law adiabatically isolated -Entropy change of the high temperature bath: -Device: only effect is to accept heat from the reservoir and transform it entirely into work Device in the same state at the start and finish of the process Total entropy change Existence of a non-Kelvin device violates entropy statement of 2 nd law

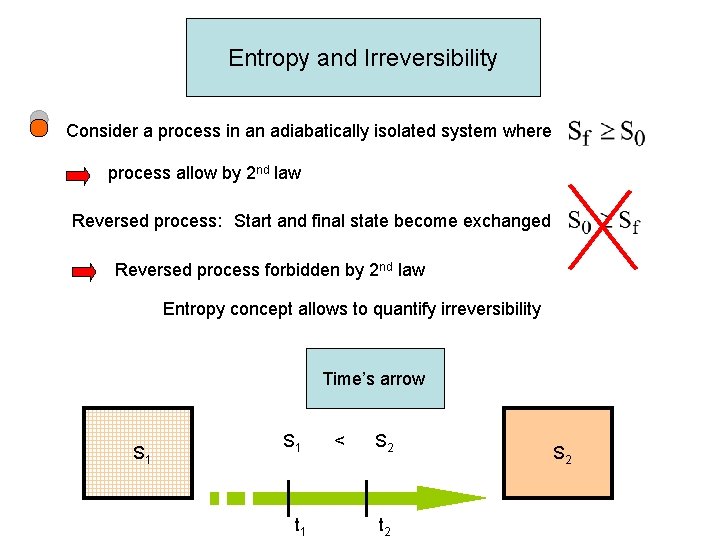
Entropy and Irreversibility Consider a process in an adiabatically isolated system where process allow by 2 nd law Reversed process: Start and final state become exchanged Reversed process forbidden by 2 nd law Entropy concept allows to quantify irreversibility Time’s arrow S 1 t 1 < S 2 t 2 S 2

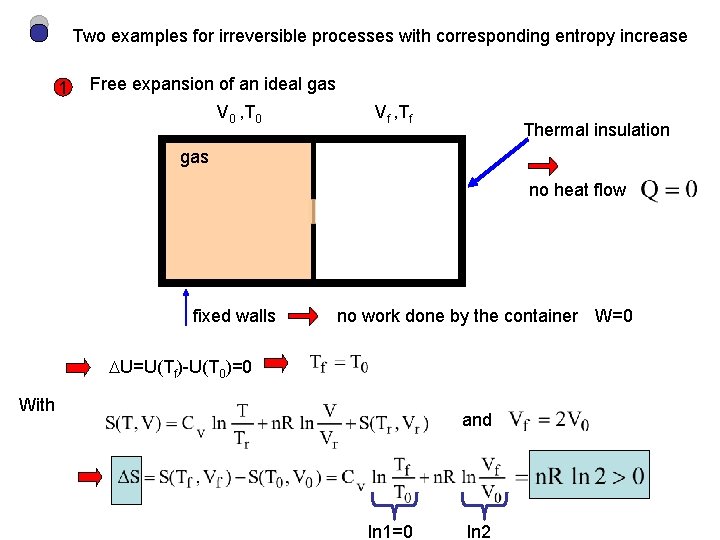
Two examples for irreversible processes with corresponding entropy increase 1 Free expansion of an ideal gas V 0 , T 0 Vf , Tf Thermal insulation gas no heat flow fixed walls no work done by the container W=0 U=U(Tf)-U(T 0)=0 With and ln 1=0 ln 2

Fast free expansion is certainly not a sequence of reversible equilibrium processes only initial and final states can be represented in state space Let’s calculate S via an integral in state space using a path, L, of reversible equilibrium processes taking the system from the initial to the final states of the actual process. We know that free expansion of the ideal gas takes place at T=const. =T 0=Tf We can therefore bring the system from (T 0, V 0) to (Tf, Vf) via an isothermal expansion T=const.

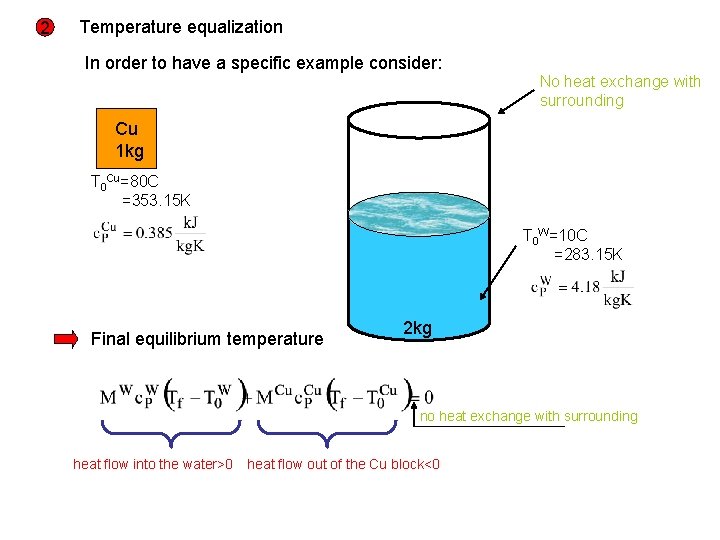
2 Temperature equalization In order to have a specific example consider: No heat exchange with surrounding Cu 1 kg T 0 Cu=80 C =353. 15 K T 0 W=10 C =283. 15 K Final equilibrium temperature 2 kg no heat exchange with surrounding heat flow into the water>0 heat flow out of the Cu block<0

with Total entropy change Entropy of the Cu block decreases Total entropy of the isolated system increases

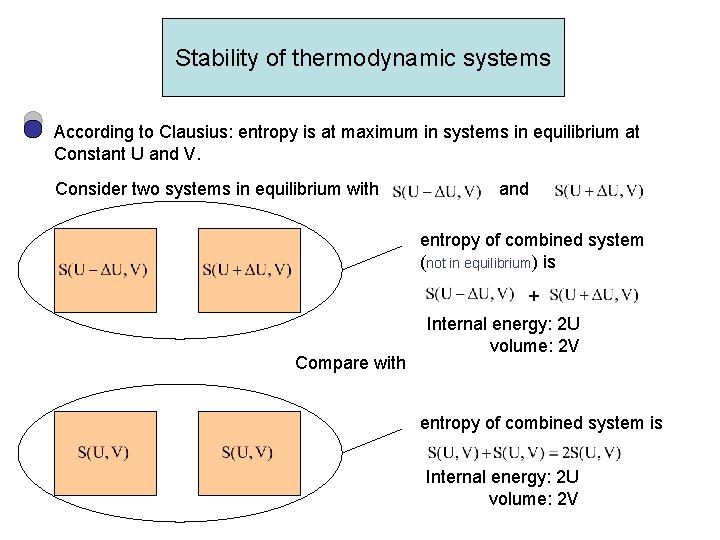
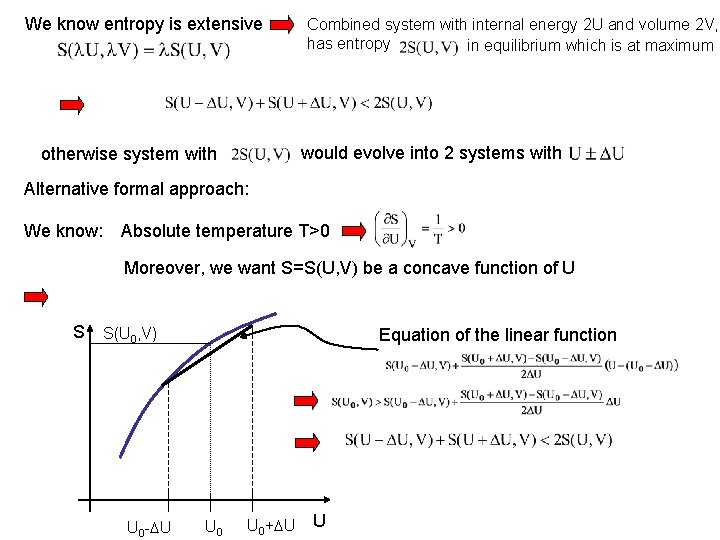
Stability of thermodynamic systems According to Clausius: entropy is at maximum in systems in equilibrium at Constant U and V. Consider two systems in equilibrium with and entropy of combined system (not in equilibrium) is + Compare with Internal energy: 2 U volume: 2 V entropy of combined system is Internal energy: 2 U volume: 2 V

We know entropy is extensive Combined system with internal energy 2 U and volume 2 V, has entropy in equilibrium which is at maximum would evolve into 2 systems with otherwise system with Alternative formal approach: We know: Absolute temperature T>0 Moreover, we want S=S(U, V) be a concave function of U S S(U 0, V) U 0 - U Equation of the linear function U 0+ U U

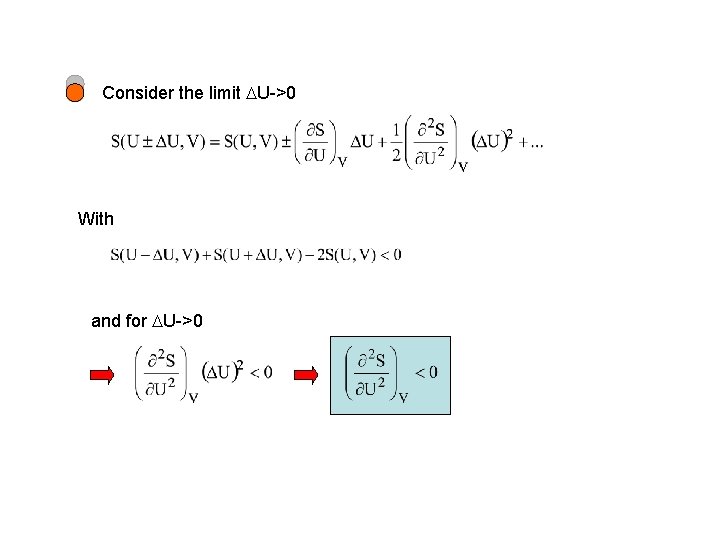
Consider the limit U->0 With and for U->0

Let’s apply the same consideration for the volume Consider the limit V->0 With For V->0
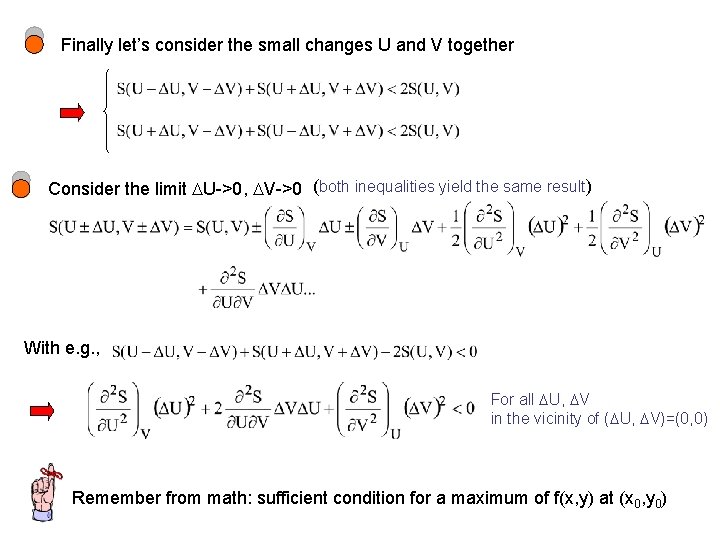
Finally let’s consider the small changes U and V together Consider the limit U->0, V->0 (both inequalities yield the same result) With e. g. , For all U, V in the vicinity of ( U, V)=(0, 0) Remember from math: sufficient condition for a maximum of f(x, y) at (x 0, y 0)

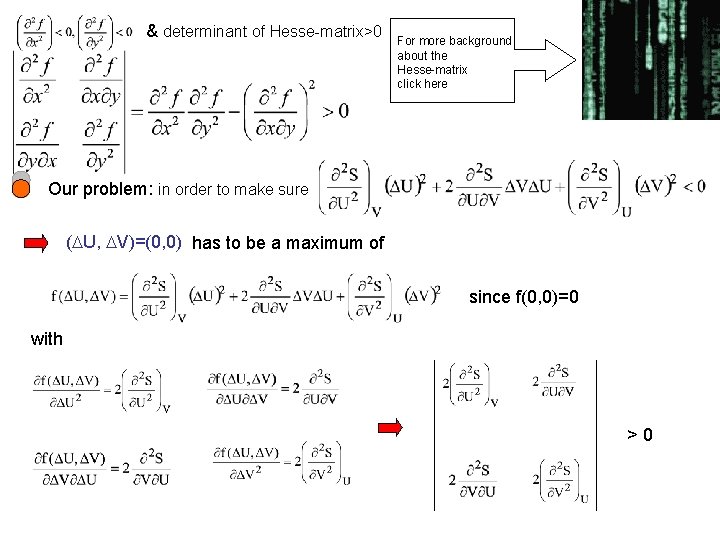
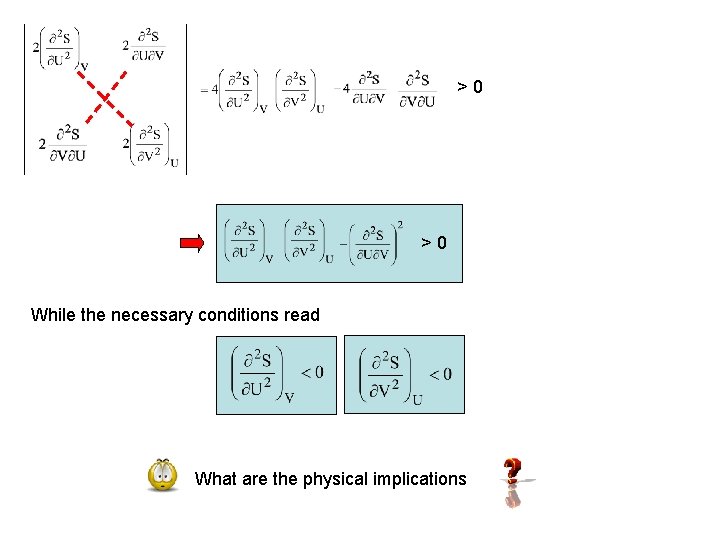
& determinant of Hesse-matrix>0 For more background about the Hesse-matrix click here Our problem: in order to make sure ( U, V)=(0, 0) has to be a maximum of since f(0, 0)=0 with >0

>0 >0 While the necessary conditions read What are the physical implications

1 <0 From 2 >0 see textbook

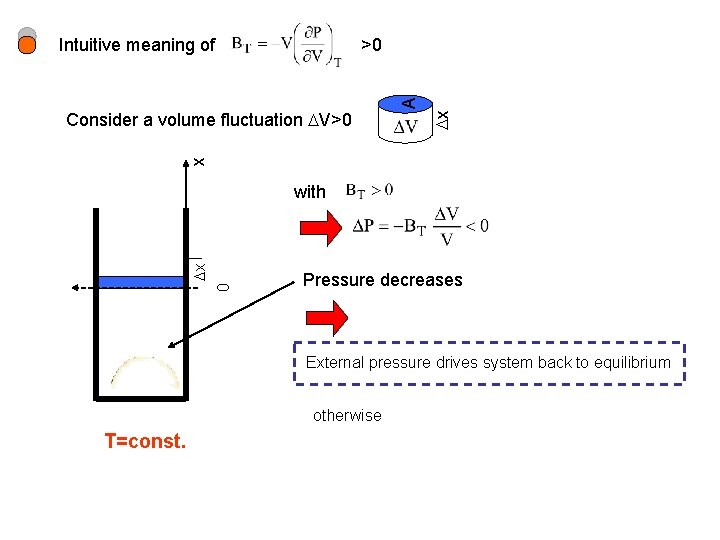
>0 x x Consider a volume fluctuation V>0 A Intuitive meaning of 0 x with Pressure decreases External pressure drives system back to equilibrium otherwise T=const.