Entropy and Gibbs Free Energy Chemistry Mrs Coyle

Entropy and Gibbs Free Energy Chemistry Mrs. Coyle

Spontaneous Process p A process that occurs without outside intervention. p Spontaneous processes can be fast or slow!

What Factors Determine the Spontaneity of a Reaction? p Entropy p Enthalpy p What ties the two together is: Gibbs Free Energy.

Entropy p Symbol p. A S measure of molecular randomness or disorder.

Disorder p Chemical processes spontaneously go to a direction of increased entropy. p Why? p Probability

Probability of Disorder p Is there a higher probability your room will be messy or neat as time goes on?

System the part of the universe under investigation.

Entropy of the System p Is greater in: n n n Gases than solids. Larger volumes of gases than smaller volumes. Larger number of gas molecules than smaller number of gas molecules.
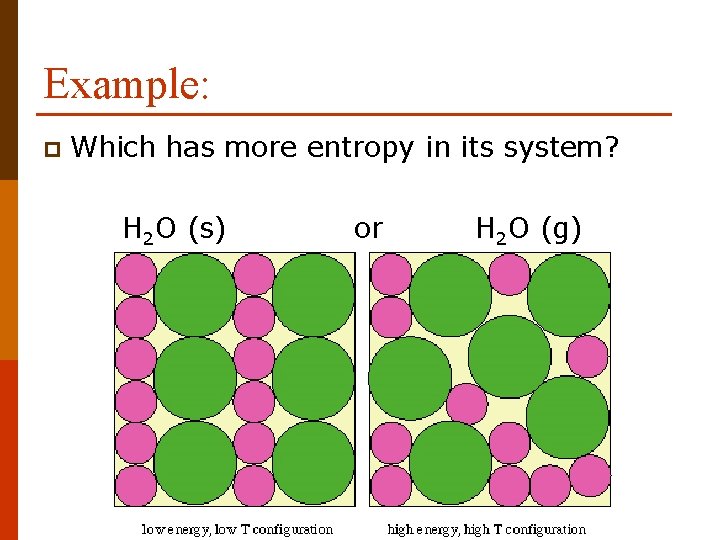
Example: p Which has more entropy in its system? H 2 O (s) or H 2 O (g)

Enthalpy (Heat of Reaction) p Spontaneity is favored when the process is exothermic (DH<0).

Example of a spontaneous exothermic reaction: p 2 SO 2(g)+ O 2(g) 2 SO 3(g) + heat p DH<0

So…… : p When DH <0 (exothermic) and DSsystem>0 (Greater Disorder) the reaction would be spontaneous.

p Are all spontaneous reactions exothermic and with a greater system disorder? p Answer: No.

What about exothermic and less disorder? Use Gibbs Free Energy p Gibbs Free Energy can be used to predict the spontaneity and it ties together the DH and the DS, the two driving forces of reactions. DG= DH-TDS (all quantities refer to the system)

Gibbs Free Energy that can be converted to work. DG<0 for spontaneous processes. DG=0 at equilibrium.
- Slides: 15