ENTHALPY OF COMBUSTION Hc Enthalpy of Combustion the

ENTHALPY OF COMBUSTION ΔHc � Enthalpy of Combustion: the heat energy given out when one mole of a substance burns in oxygen. �Always exothermic, so ΔHc always negative
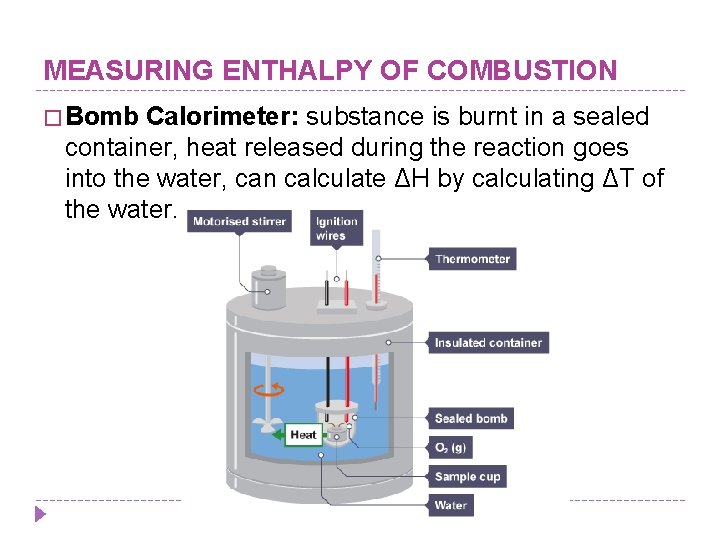
MEASURING ENTHALPY OF COMBUSTION � Bomb Calorimeter: substance is burnt in a sealed container, heat released during the reaction goes into the water, can calculate ΔH by calculating ΔT of the water.
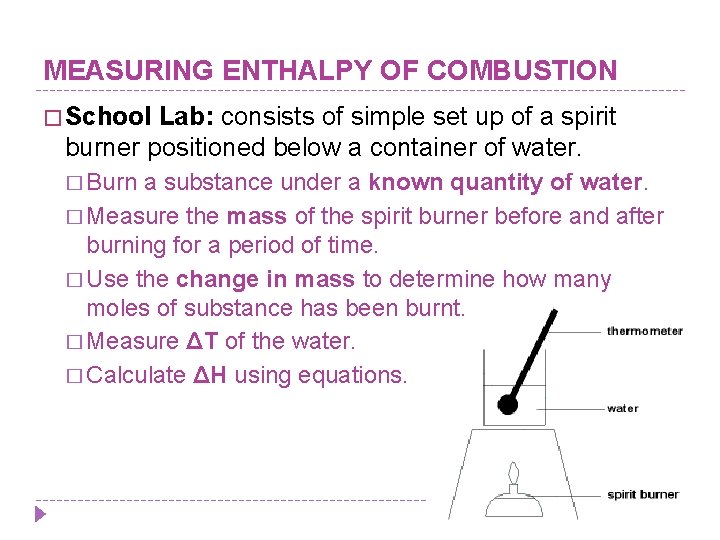
MEASURING ENTHALPY OF COMBUSTION � School Lab: consists of simple set up of a spirit burner positioned below a container of water. � Burn a substance under a known quantity of water. � Measure the mass of the spirit burner before and after burning for a period of time. � Use the change in mass to determine how many moles of substance has been burnt. � Measure ΔT of the water. � Calculate ΔH using equations.

ENTHALPY OF COMBUSTION ΔHc A spirit burner used 1. 00 g of methanol to raise the temperature of 100. 0 g water in a metal can from 25°C to 55°C. Assuming the heat capacity of water is 4. 18 J. g 1. °C-1, calculate the molar enthalpy of combustion of methanol in k. J. mol-1.

ENTHALPY OF COMBUSTION ΔHc Ethanol was placed in a spirit burner and used to heat 200 cm 3 of water in a copper can. When the temperature of the water had increased by 5°C, the mass of the burner and ethanol had decreased by 0. 36 g. Calculate the enthalpy of combustion of ethanol.
- Slides: 5