Enthalpy Cycles Combustion Data What is the enthalpy

Enthalpy Cycles: Combustion Data What is the enthalpy of formation of methane (CH 4) from carbon and hydrogen? 1. Construct a cycle (combustion products at the bottom). 2. Determine the direction of the routes 3. Work out the equation with respect to ΔH 1. 4. Calculate the values for ΔH 2 and ΔH 3. 5. Substitute the values into the equation. Δc. H (k. J mol-1) C(s) -394 H 2(g) -286 CH 4(g) -890
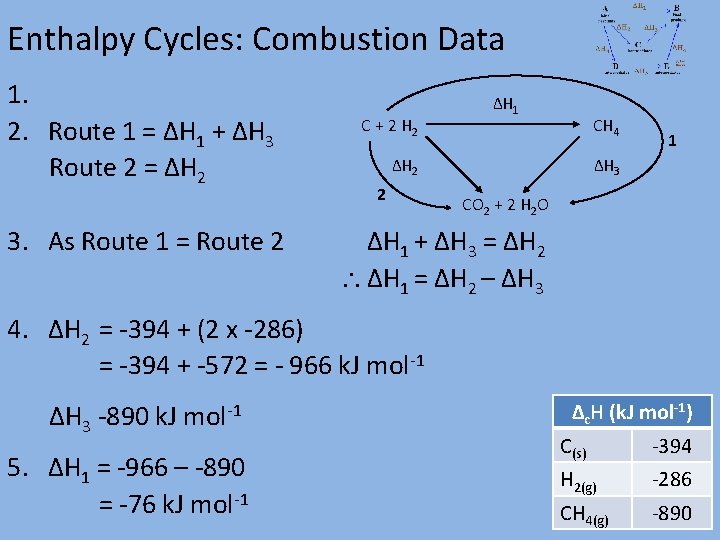
Enthalpy Cycles: Combustion Data 1. 2. Route 1 = ΔH 1 + ΔH 3 Route 2 = ΔH 2 3. As Route 1 = Route 2 C + 2 H 2 ΔH 1 CH 4 ΔH 2 2 1 ΔH 3 CO 2 + 2 H 2 O ΔH 1 + ΔH 3 = ΔH 2 ΔH 1 = ΔH 2 – ΔH 3 4. ΔH 2 = -394 + (2 x -286) = -394 + -572 = - 966 k. J mol-1 ΔH 3 -890 k. J mol-1 5. ΔH 1 = -966 – -890 = -76 k. J mol-1 Δc. H (k. J mol-1) C(s) -394 H 2(g) -286 CH 4(g) -890
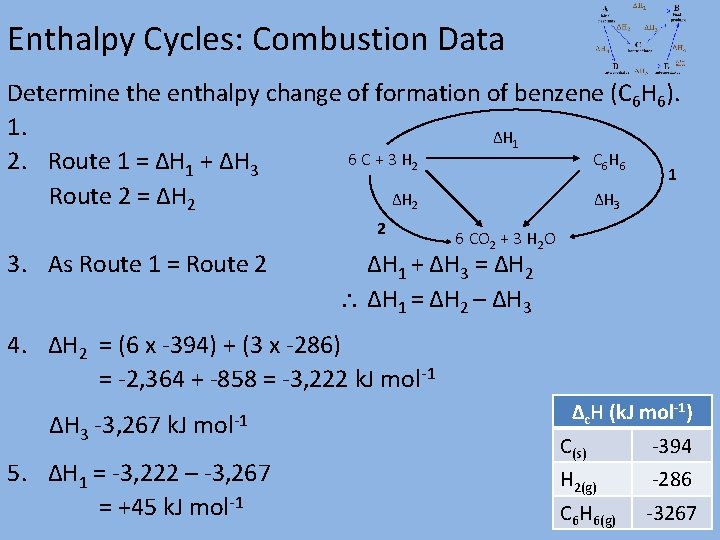
Enthalpy Cycles: Combustion Data Determine the enthalpy change of formation of benzene (C 6 H 6). 1. ΔH 1 6 C + 3 H 2 C 6 H 6 2. Route 1 = ΔH 1 + ΔH 3 1 Route 2 = ΔH 2 ΔH 3 2 3. As Route 1 = Route 2 6 CO 2 + 3 H 2 O ΔH 1 + ΔH 3 = ΔH 2 ΔH 1 = ΔH 2 – ΔH 3 4. ΔH 2 = (6 x -394) + (3 x -286) = -2, 364 + -858 = -3, 222 k. J mol-1 ΔH 3 -3, 267 k. J mol-1 5. ΔH 1 = -3, 222 – -3, 267 = +45 k. J mol-1 Δc. H (k. J mol-1) C(s) -394 H 2(g) -286 C 6 H 6(g) -3267

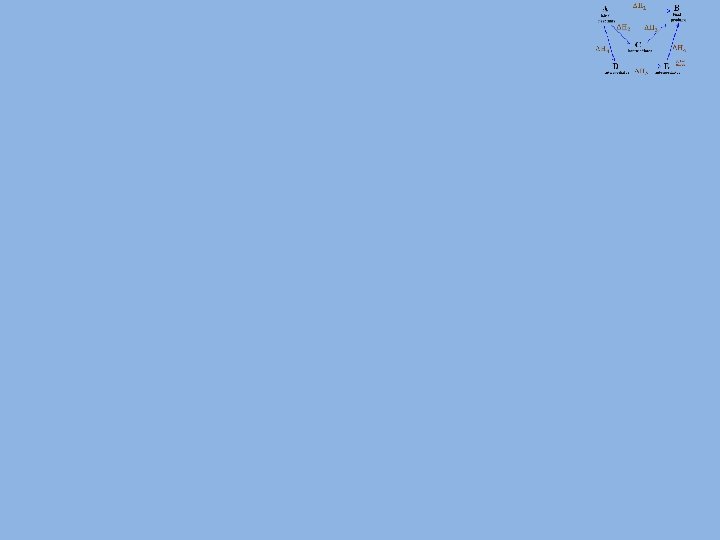
Enthalpy Cycles: Formation Data What is the enthalpy of reaction for the reaction between hydrogen chloride and ethene (CH 2=CH 2)? 1. Construct a cycle (elements in standard state at bottom). 2. Determine the direction of the routes • If the route is in the opposite direction to the cycle arrow then the value is reversed. 3. Work out the equation with respect to ΔH 1. 4. Calculate the values for ΔH 2 and ΔH 3. 5. Substitute the values into the equation. Δf. H (k. J mol-1) C 2 H 4(g) +52 HCl(g) -92 C 2 H 5 Cl(g) -109
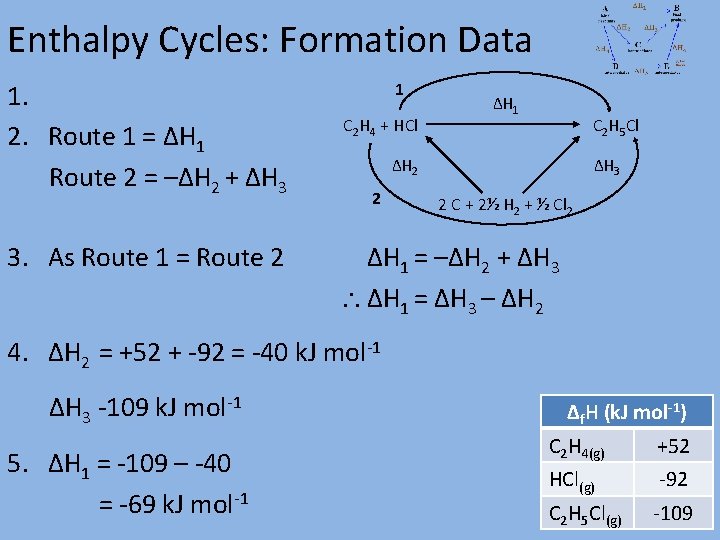
Enthalpy Cycles: Formation Data 1. 2. Route 1 = ΔH 1 Route 2 = –ΔH 2 + ΔH 3 3. As Route 1 = Route 2 1 C 2 H 4 + HCl ΔH 1 C 2 H 5 Cl ΔH 2 2 ΔH 3 2 C + 2½ H 2 + ½ Cl 2 ΔH 1 = –ΔH 2 + ΔH 3 ΔH 1 = ΔH 3 – ΔH 2 4. ΔH 2 = +52 + -92 = -40 k. J mol-1 ΔH 3 -109 k. J mol-1 5. ΔH 1 = -109 – -40 = -69 k. J mol-1 Δf. H (k. J mol-1) C 2 H 4(g) +52 HCl(g) -92 C 2 H 5 Cl(g) -109
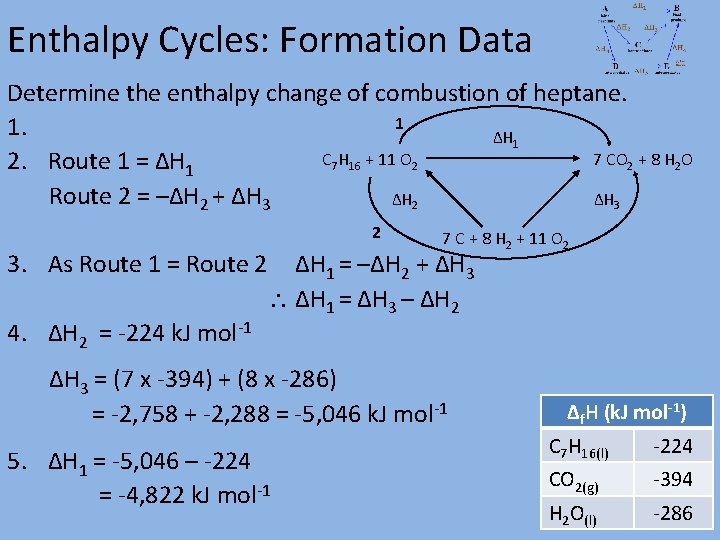
Enthalpy Cycles: Formation Data Determine the enthalpy change of combustion of heptane. 1 1. ΔH 1 C 7 H 16 + 11 O 2 7 CO 2 + 8 H 2 O 2. Route 1 = ΔH 1 Route 2 = –ΔH 2 + ΔH 3 ΔH 2 ΔH 3 2 3. As Route 1 = Route 2 4. ΔH 2 = -224 k. J mol-1 7 C + 8 H 2 + 11 O 2 ΔH 1 = –ΔH 2 + ΔH 3 ΔH 1 = ΔH 3 – ΔH 2 ΔH 3 = (7 x -394) + (8 x -286) = -2, 758 + -2, 288 = -5, 046 k. J mol-1 5. ΔH 1 = -5, 046 – -224 = -4, 822 k. J mol-1 Δf. H (k. J mol-1) C 7 H 16(l) -224 CO 2(g) -394 H 2 O(l) -286
- Slides: 7