Enthalpy and Chemical Reactions Combustion Reactions 1 mole

Enthalpy and Chemical Reactions Combustion Reactions 1 mole CH 4(g) + 2 O 2(g) (Exothermic) CO 2(g) + 2 H 2 O(g) + 802. 3 k. J (heat) Hcombustion = - 802. 3 k. J mol. CH 4 (. . . per mole of substance burned) Formation Reactions C(s) + 2 H 2(g) + ½ O 2(g) Hformation = - 239 1 mole (Exothermic) CH 3 OH(g) + 239 k. J (heat) k. J (. . . per mole of substance formed) mol. CH 3 OH
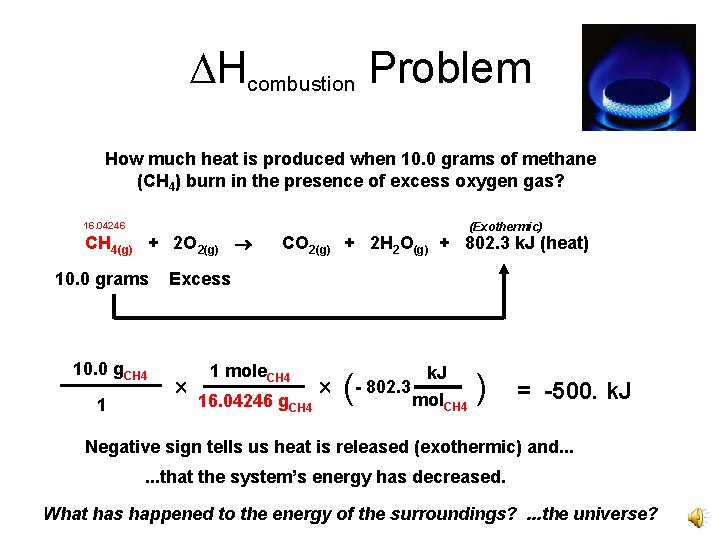
Hcombustion Problem How much heat is produced when 10. 0 grams of methane (CH 4) burn in the presence of excess oxygen gas? 16. 04246 CH 4(g) + 2 O 2(g) 10. 0 grams 10. 0 g. CH 4 1 (Exothermic) CO 2(g) + 2 H 2 O(g) + 802. 3 k. J (heat) Excess 1 mole. CH 4 16. 04246 g. CH 4 ( k. J - 802. 3 mol. CH 4 ) = -500. k. J Negative sign tells us heat is released (exothermic) and. . . that the system’s energy has decreased. What has happened to the energy of the surroundings? . . . the universe?

Enthalpy and Chemical Reactions Heats of fusion: Melting a solid into a liquid (Endothermic) 1 mole + H 2 O (s) 6. 02 k. J Hfusion = H 2 O(l) + 6. 02 k. J mol. H 2 O (. . . per mole of substance melted) Heat of vaporization: Vaporizing a liquid into a gas. (Endothermic) 2260 k. J 1 mole + H 2 O (l) Hvaporization = H 2 O(g) + 2260 Why should it require so much more energy to vaporize water in comparison to melt it? k. J mol. H 2 O (. . . per mole of substance vaporized) Because it requires much more energy to separate molecules to great distances found in gases.
- Slides: 3