Enterobius vermicularis Department of Medical Parasitology Introduction Enterobius

Enterobius vermicularis Department of Medical Parasitology

Introduction • Enterobius vermicularis, commonly known as pinworm or seatworm • E. vermicularis is parasitic only to humans • Adults inhabit the ileocecus, that is, cecum and adjacent ascending colon and distal ileum. The infection of E. vermicularis may cause Enterobiasis • World-wide distribution, it is commonly found in kindergarten and primary school students

Classification Phylum Nemathelminthes Class Nematoda Order Oxyurata Family Oxyuridae

Morphology -- Adult • Female -- fusiform body with a long, thin, tapering tail, 8 to 13 mm • Male -- “ 6” shape, curved tail, 2 to 5 mm. Males die right after mating, thus are rarely seen • White in color

• Cephalic alae The anterior end tapers and is flanked on each side by cuticular extensions of head • Pharyngeal bulb The esophagus is slender, terminating in a prominent posterior bulb

Morphology -- Egg • Oval in shape, 50~60× 25μm in average, a larva inside • Clear, colorless and doubly refractive egg shell, flattened on one side
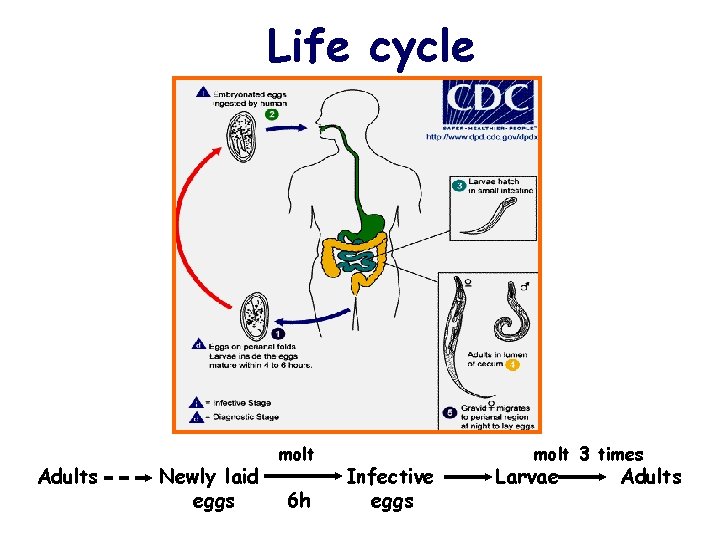
Life cycle Adults Newly laid eggs molt 6 h Infective eggs molt 3 times Larvae Adults

Characteristics of life cycle • Humans are the only host in nature • No intermediate host (direct life cycle) • No larval migration between organs
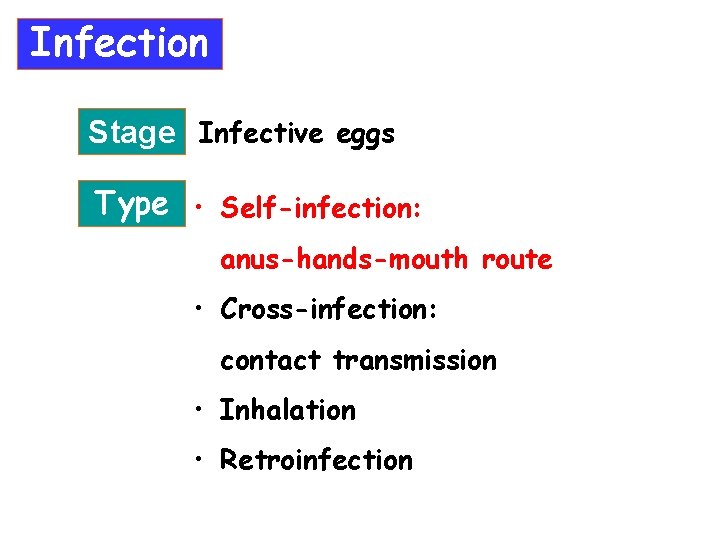
Infection Stage Infective eggs Type • Self-infection: anus-hands-mouth route • Cross-infection: contact transmission • Inhalation • Retroinfection
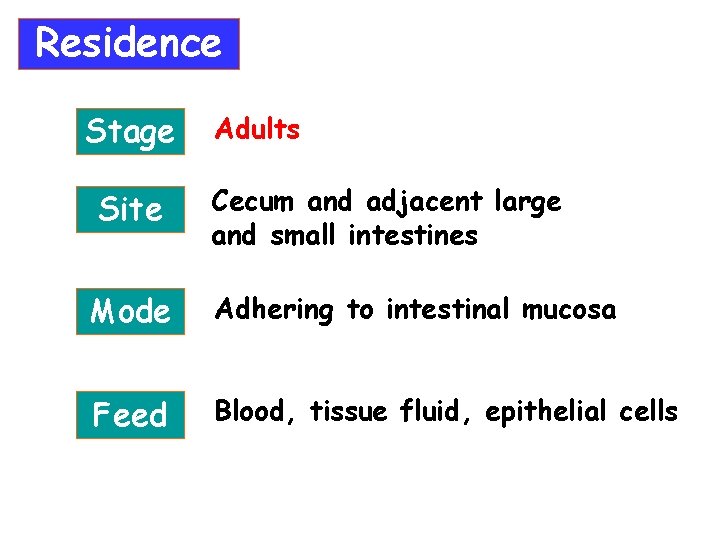
Residence Stage Site Adults Cecum and adjacent large and small intestines Mode Adhering to intestinal mucosa Feed Blood, tissue fluid, epithelial cells
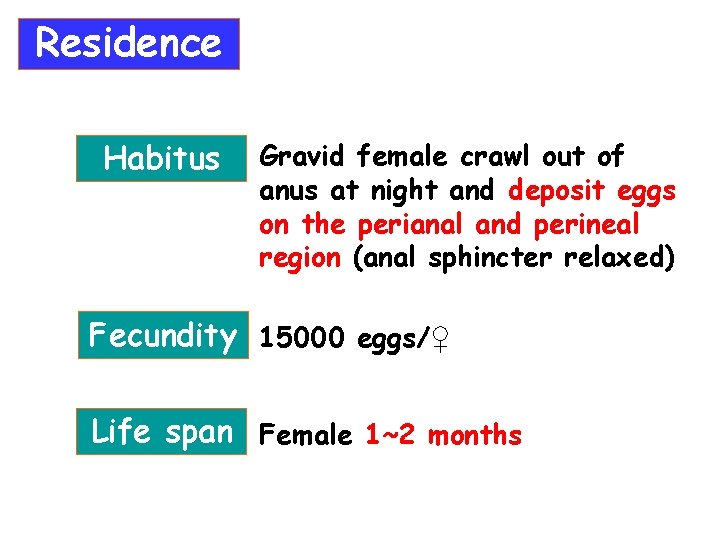
Residence Habitus Gravid female crawl out of anus at night and deposit eggs on the perianal and perineal region (anal sphincter relaxed) Fecundity 15000 eggs/♀ Life span Female 1~2 months

Discharge Stage Eggs Mode Female release eggs on the perianal zone

Pathogenesis • Enterobiasis is usually asymptomatic, the adults may cause slight irritation of the intestinal mucosa • The most typical symptom is perianal pruritus (itching and irritation), which associates with the nocturnal migration of the gravid females from the anus and deposition of eggs in the perianal folds of the skin, may lead to excoriations and bacterial superinfection

Pathogenesis • Heavy infection in children may result in restlessness, sleeplessness, anorexia, weigh loss, grinding of teeth, nervousness, irritability, abdominal pain and vomiting • Sometimes, pinworm may migrate up the female reproductive tract, cause vaginitis, endometritis and granuloma in uterus and fallopian tubes. Occasionally, invasion of the female to the appendix, the peritoneal cavity or the urinary bladder may occur
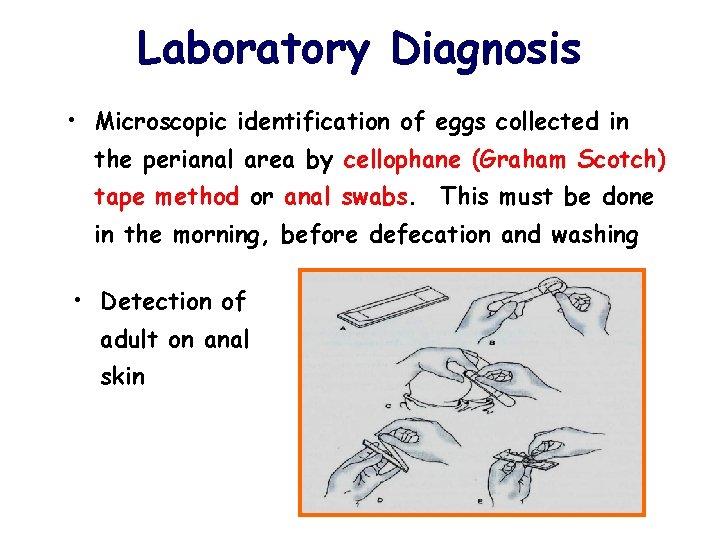
Laboratory Diagnosis • Microscopic identification of eggs collected in the perianal area by cellophane (Graham Scotch) tape method or anal swabs. This must be done in the morning, before defecation and washing • Detection of adult on anal skin

Epidemiology Geographical distribution • Geographical distribution—cosmopolitan in temperate zones with about 30~50% of the population infected, and more prevalent in children than adults. it is estimated that 500 million people are infected Worldwide • Enterobiasis is most common where people live under crowded conditions such as orphanages, kindergartens, and large families
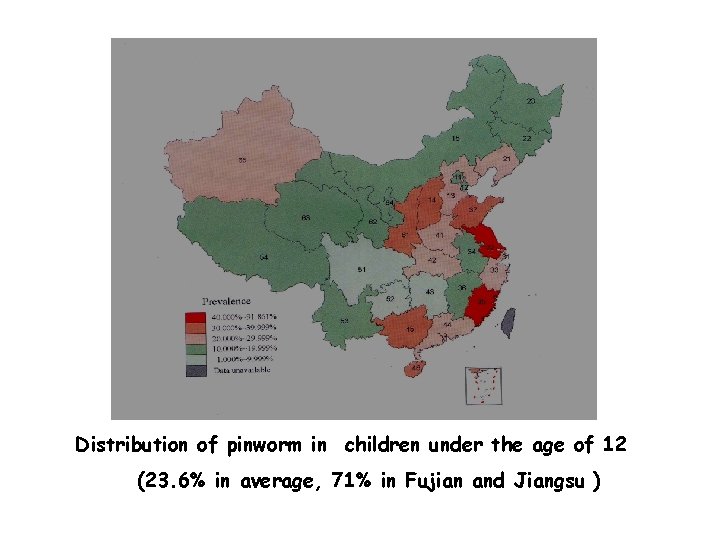
Distribution of pinworm in children under the age of 12 (23. 6% in average, 71% in Fujian and Jiangsu )

Source of infection Patients and carriers Prevalent features • Multiple ways of infection • Reinfection

Treatment and prevention • Albendazole/Mebendazole: 95% effective • Repeated retreatment may be necessary for a radical cure • Personal hygiene and eating habits • Sanitary disposal of clothing, bed linen, and environment • Health education

Trichinella spiralis

Introduction • Trichinella spiralis is capable of infecting all mammals, cause parasitic zoonosis--trichinellosis • Human beings acquire the food-borne trichinellosis by ingesting the raw or undercooked meat of pigs and other animals containing the Trichinella larvae • Larvae which inhabit the striated skeletal muscles are the main pathogenic stage • World-wide distribution

Classification Phylum Nemathelminthes Class Nematoda Order Trichurata Family Trichinellidae
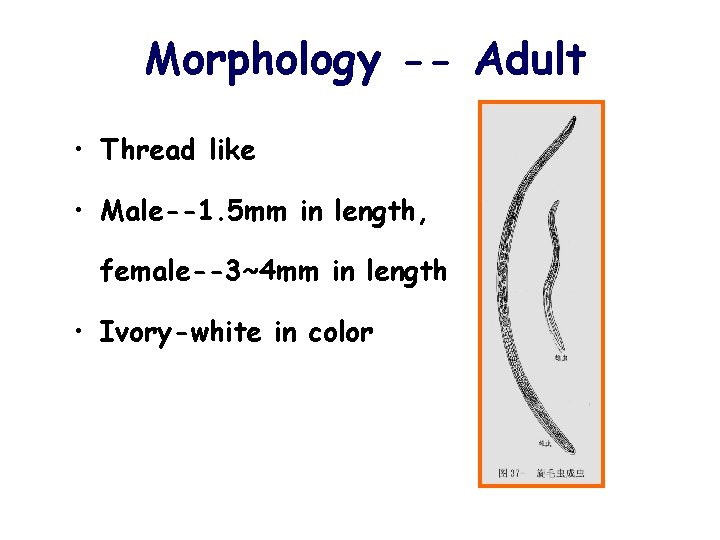
Morphology -- Adult • Thread like • Male--1. 5 mm in length, female--3~4 mm in length • Ivory-white in color
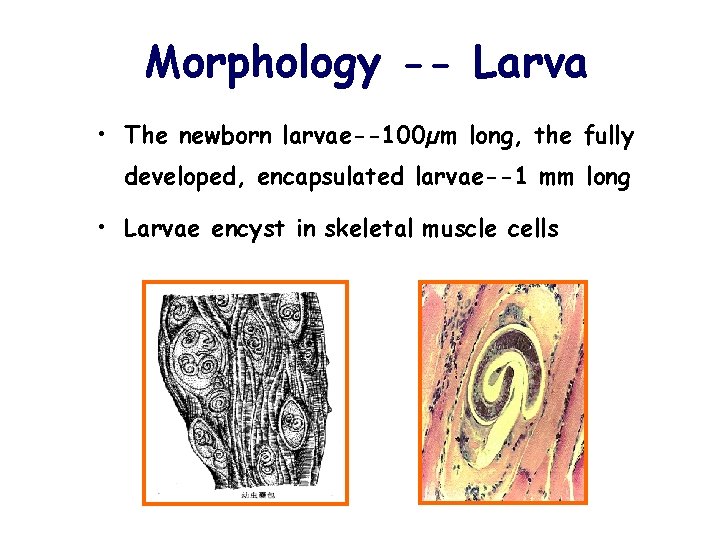
Morphology -- Larva • The newborn larvae--100µm long, the fully developed, encapsulated larvae--1 mm long • Larvae encyst in skeletal muscle cells
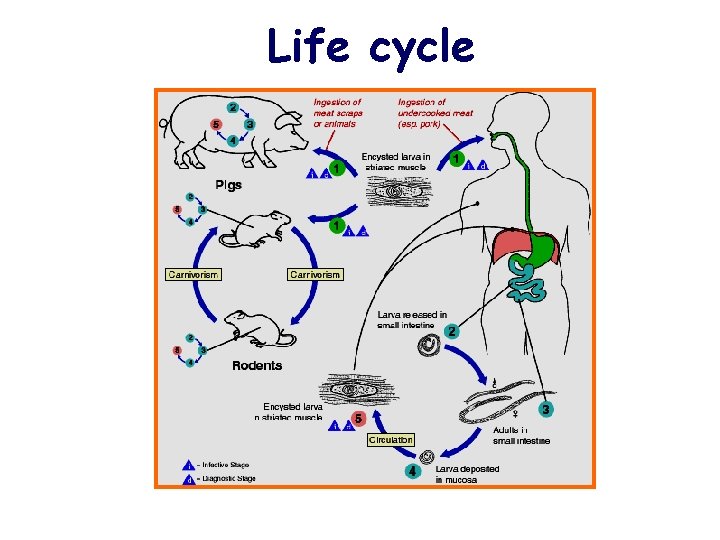
Life cycle
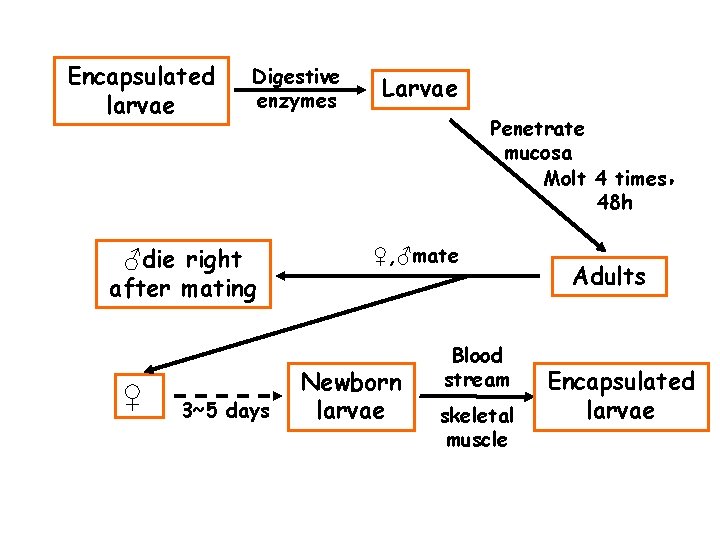
Encapsulated larvae Digestive enzymes ♂die right after mating ♀ 3~5 days Larvae Penetrate mucosa Molt 4 times, 48 h ♀, ♂mate Newborn larvae Blood stream skeletal muscle Adults Encapsulated larvae
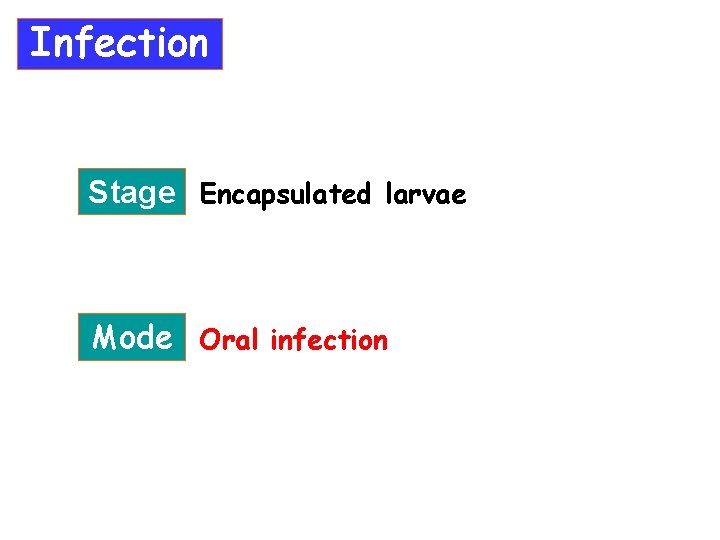
Infection Stage Encapsulated larvae Mode Oral infection

Residence Stage Adults, larvae Site Adults--duodenal and jejunal mucosa Larvae--skeletal muscles of the tongue, the deltoid, pectoral, and intercostal muscles, the diaphragm, and the gastrocnemius Life span Female adults--1~2 months Larvae--several to 30 years

Characteristics of life cycle • Adults and larvae live in the same host (not only final host but also intermediate host), alternation of hosts is needed to complete the whole life cycle • Ovoviviparity • No discharge or free-living stages • The only intracellular parasitic nematode
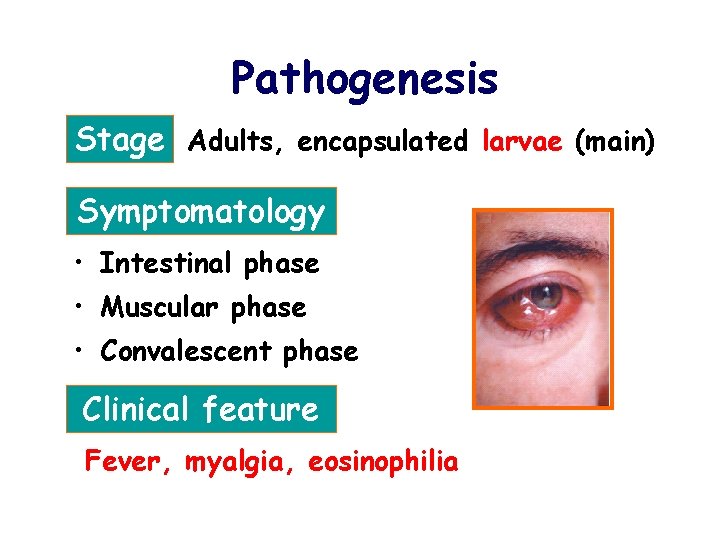
Pathogenesis Stage Adults, encapsulated larvae (main) Symptomatology • Intestinal phase • Muscular phase • Convalescent phase Clinical feature Fever, myalgia, eosinophilia
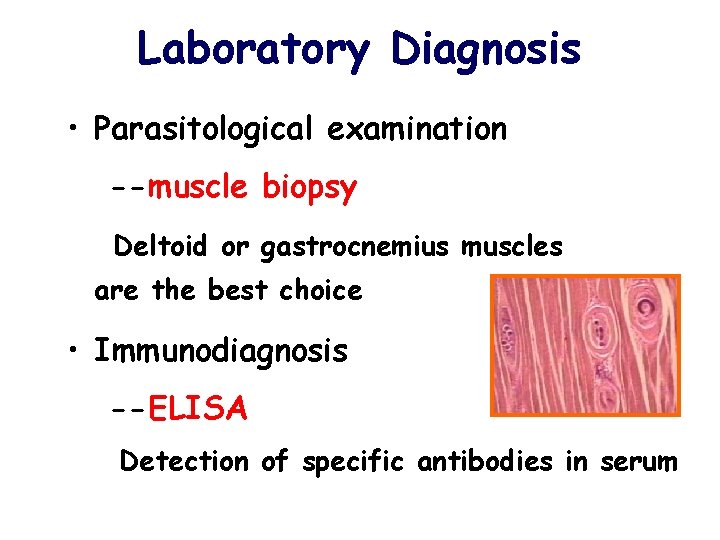
Laboratory Diagnosis • Parasitological examination --muscle biopsy Deltoid or gastrocnemius muscles are the best choice • Immunodiagnosis --ELISA Detection of specific antibodies in serum

Epidemiology Geographical distribution • Cosmopolitan The global prevalence of trichinellosis is estimated as many as 11 million people may be infected. More cases of human infection have been found in developed countries than in developing countries
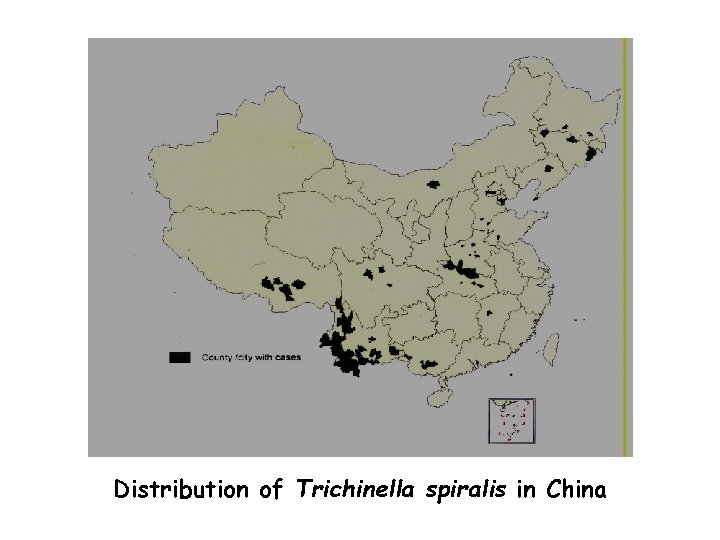
Distribution of Trichinella spiralis in China

Source of infection Pigs and other wild animals containing the Trichinella larvae Mode of infection Ingesting the raw or improperly prepared pork products

Treatment and prevention • Albendazole / Mebendazole • Development of industrialized pig farms • Improvement of pig feeding • Inspection of meat • Health education

Filaria

Introduction • Filariasis is a widely spread disease caused by different species of filariae • The adults of filariae inhabit the lymphatics, subcutaneous tissue, deep connective tissue, peritoneal or thoracic cavity • There are eight species of filariae, namely, Wuchereria bancrofti, Brugia malayi, Brugia timori, Loa loa, Onchocerca volvulus, Mansonella ozzardi, Dipetalonema perstans and Dipetalonema streptocerca in humans

• Larvae known as microfilariae appear in the circulating blood or tissue fluids • W. bancrofti and B. malayi which lie coiled in the lymphatic vessels are most commonly responsible for lymphatic filariasis and of more medical importance • Microfilariae circulate in peripheral blood once each day. Mosquito is essential vector and intermediate host

Classification Phylum Nemathelminthes Class Nematoda Order Filariata Family Dipetalonematidae

Wuchereria bancrofti and Brugia malayi

Morphology -- Adult • Slender, thread-like • White in color • Male -- 2. 5 -4 cm long and has a curved tail, female -- 5 -10 cm in length
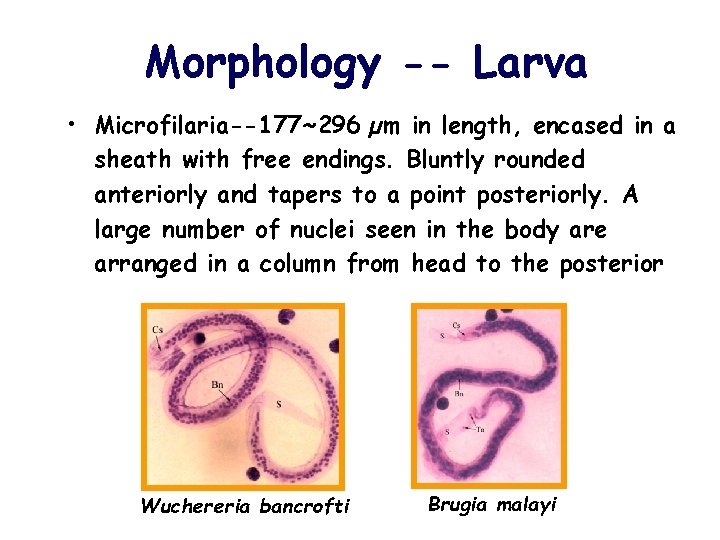
Morphology -- Larva • Microfilaria--177~296 µm in length, encased in a sheath with free endings. Bluntly rounded anteriorly and tapers to a point posteriorly. A large number of nuclei seen in the body are arranged in a column from head to the posterior Wuchereria bancrofti Brugia malayi
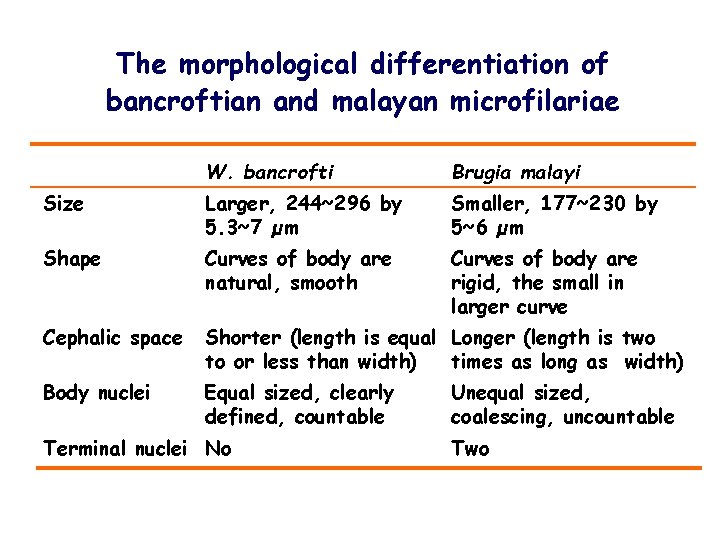
The morphological differentiation of bancroftian and malayan microfilariae W. bancrofti Brugia malayi Size Larger, 244~296 by 5. 3~7 µm Smaller, 177~230 by 5~6 µm Shape Curves of body are natural, smooth Curves of body are rigid, the small in larger curve Cephalic space Shorter (length is equal Longer (length is two to or less than width) times as long as width) Body nuclei Equal sized, clearly defined, countable Terminal nuclei No Unequal sized, coalescing, uncountable Two
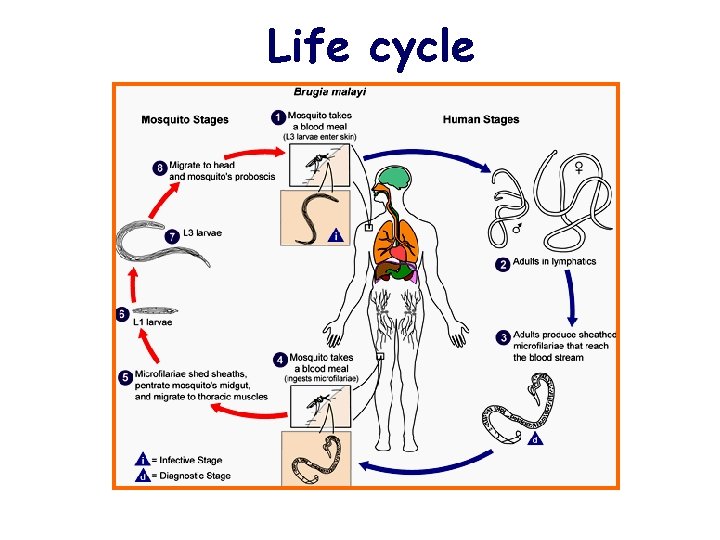
Life cycle
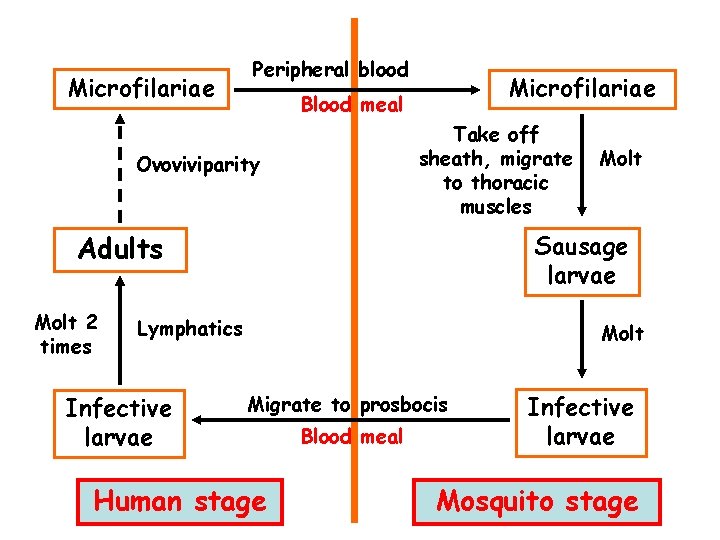
Microfilariae Peripheral blood Microfilariae Blood meal Take off sheath, migrate to thoracic muscles Ovoviviparity Adults Molt 2 times Sausage larvae Lymphatics Infective larvae Molt Migrate to prosbocis Human stage Blood meal Infective larvae Mosquito stage

Characteristics of life cycle • Host: mosquitoes as intermediate host, human as final host • Location: lymphatic vessels and lymph nodes • Infective stage: infective larvae • Transmission stage: microfilariae • Diagnostic stage: microfilariae • Ovoviviparity

Characteristics of life cycle • Different parasitic site between the two species of lymphatic filariae: W. bancrofti parasitizes in the superficial and deep lymphatic systems, including in the genitourinary lymphatic system; B. malayi parasitizes in the shallow lymphatic system only, especially in the lymphatics of limbs

Characteristics of life cycle • Nocturnal periodicity: the numbers of microfilariae present in the peripheral blood during daytime is very low in density, usually undetectable, but gradually increase from evening to midnight and reach the greatest density at 10 p. m. to 2 a. m. • The mechanism for this phenomenon is still not clear. It may be related to the change of oxygen tension in cerebral and the pulmonary vessels

Pathogenesis • Results from a complex interplay of the pathogenic potential of parasite, the immune response of the host, and external bacterial and fungal infections • Acute diseases--dilatation of the lymphatics / hyperplastic changes in the vessel endothelium / infiltration by lymphocytes, plasma cells and eosinophils / thrombus formation

Pathogenesis • Chronic lesion--the changes include granuloma formation, fibrosis, and permanent lymphatic obstruction • Repeated infections eventually result in massive lymphatic blockade • The skin and subcutaneous tissues become edematous, thickened, and fibrotic • Dilated vessels may rupture, spilling lymph into the tissue to cause lymphedema and elephantiasis

Clinical manifestations • Asymptomatic amicrofilariaemic • Asymptomatic microfilariaemic • Acute manifestation • Obstructive (Chronic) lesions

Clinical manifestations • Asymptomatic amicrofilariaemic--in endemic areas, a proportion of population does not show microfilariae or clinical manifestation even though they have some degree of exposure to infective larva similar to those who become infected. Laboratory diagnostic techniques are not able to determine whether they are infected or free

Clinical manifestations • Asymptomatic microfilariaemic-considerable proportions are asymptomatic for months and years, though they have circulating microfilariae. They are an important source of infection. They can be detected by night blood survey and other suitable procedures

Clinical manifestations • Acute manifestation--during initial months and years, there are recurrent episodes of acute inflammation in the lymph vessel/node of the limb & scrotum that are consisting of filarial fever, lymphangitis, lymphadinitis, epididymitis, orchitis
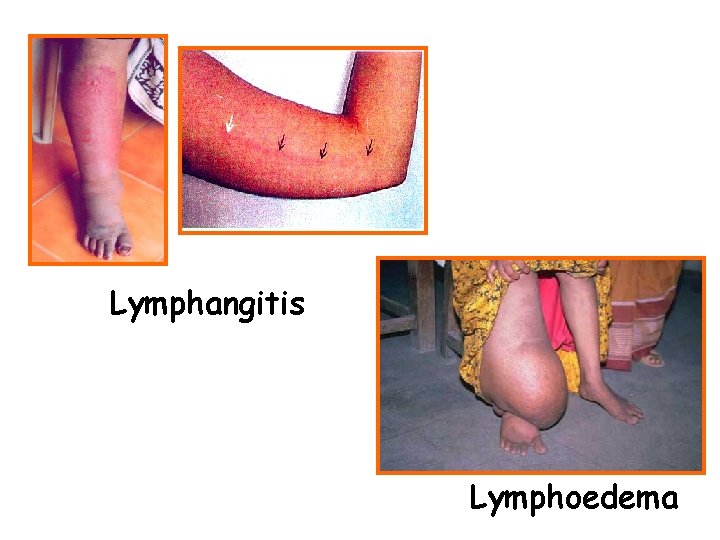
Lymphangitis Lymphoedema

Clinical manifestations • Obstructive (Chronic) lesions--takes 10 -15 years, main pathological change is lymph obstruction. The lymph circulation is disturbed and lymphedema occurs. The affected limb feels soft at first and becomes fibrotic after extensive growth of connective tissue as elephantiasis develops. e. g. hydrocoele (40~60%), elephantiasis of scrotum, penis, leg, arm, vulva, breast, and chyluria
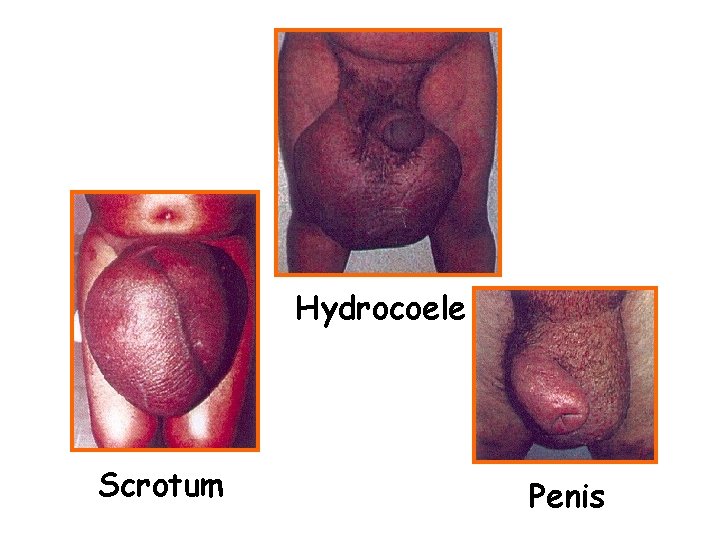
Hydrocoele Scrotum Penis
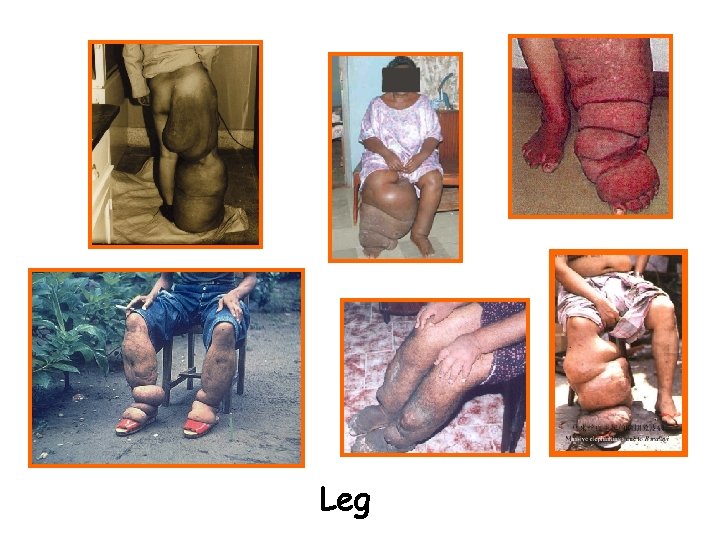
Leg
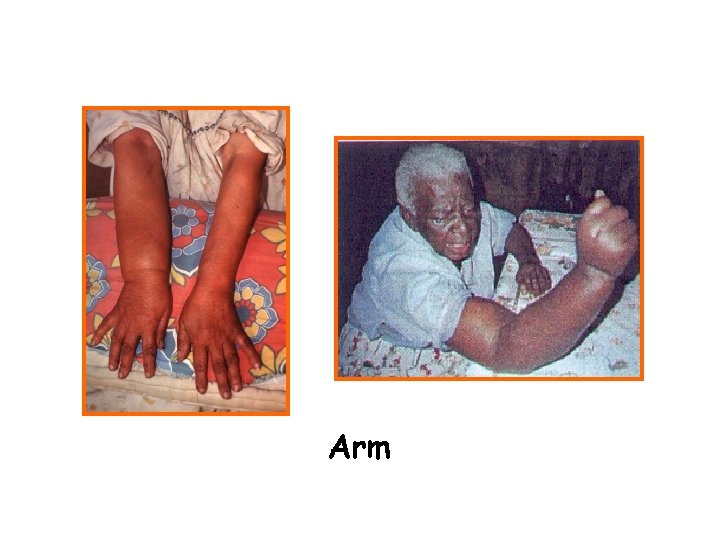
Arm
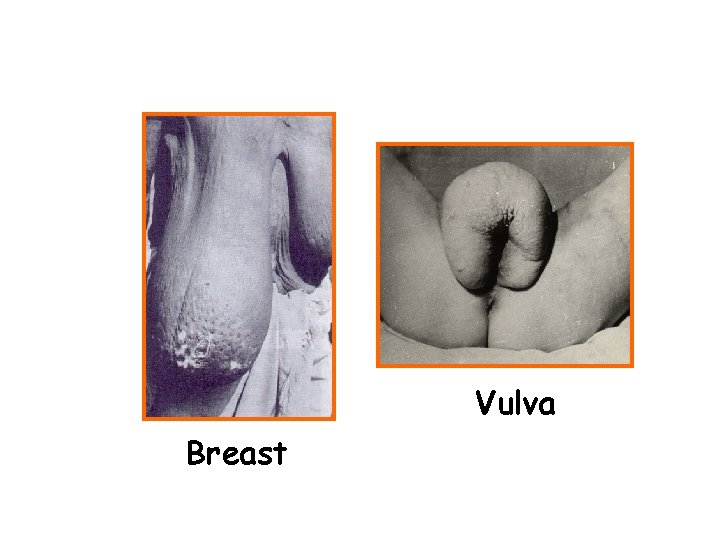
Vulva Breast
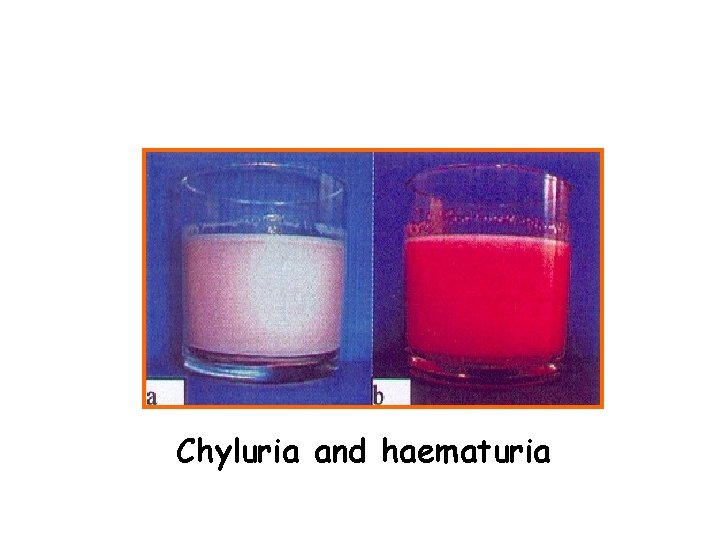
Chyluria and haematuria

Laboratory Diagnosis • Microfilariae: demonstration of microfilarae in the peripheral blood a. Thick blood smear: 2~3 drops of free flowing blood by finger prick method b. Membrane filtration method: 1~2 ml intravenous blood filtered through 3µm pore size membrane filter c. DEC provocative test (2 mg/Kg): after consuming DEC, mf enters into the peripheral blood in day time within 30~45 minutes • Adult: biopsy of the nodes or lymphatic vessels • Immunological tests: antibody or circulating filarial antigen (CFA)

Epidemiology Geographical distribution • • W. bancrofti is widely distributed throughout the tropics and subtropics. It is prevalent in Africa, Asia, and certain islands in the Pacific Ocean B. malayi has a distribution centering around the Malay peninsula. In addition to Malaysia, it occurs in India, Indonesia, New Guinea, Thailand, Vietnam, Korea, Japan and China
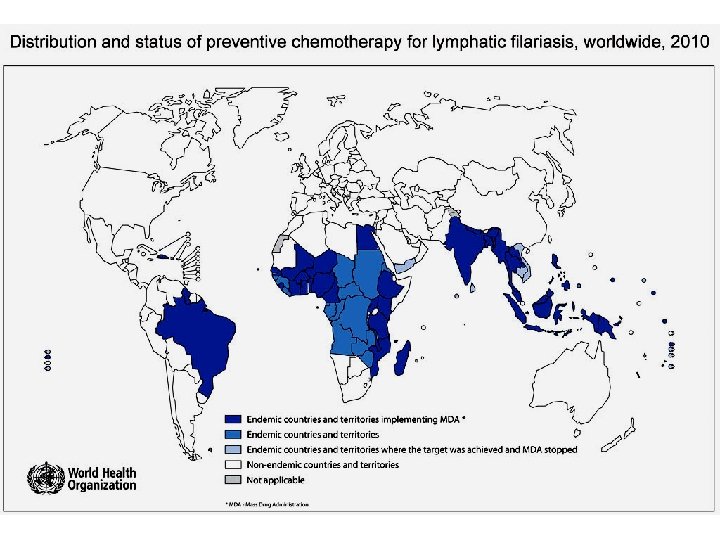

Epidemiology Global scenario • • • Population at risk: 1. 2 Billion No. of countries : > 80 Mf carriers: 76 Million Diseased: 44 Million Hydrocele: 27 Million Lymphoedema: 16 Million

Treatment and prevention • The source of infection should be eradicated by mass survey and treatment • All person with microfilariae should be treated with diethylcarbamazine (DEC) which is the low toxicity but most effective drug. 200 mg tid for seven days as one course. DEC added to table salt (3: 1000) and distributed in endemic areas over a period of six months, results in great reduction of microfilaria in the blood stream • Elimination of vectors and protection of the people from mosquito bites are important to control filariasis
- Slides: 66