Enhanced ferrihydrite dissolution by a unicellular planktonic cyanobacterium

Enhanced ferrihydrite dissolution by a unicellular, planktonic cyanobacterium: a biological contribution to particulate iron bioavailability Or How does Synechocystis influence iron bioavailability? Chana Kranzler, Nivi Kessler, Nir Keren and Yeala Shaked (2017)
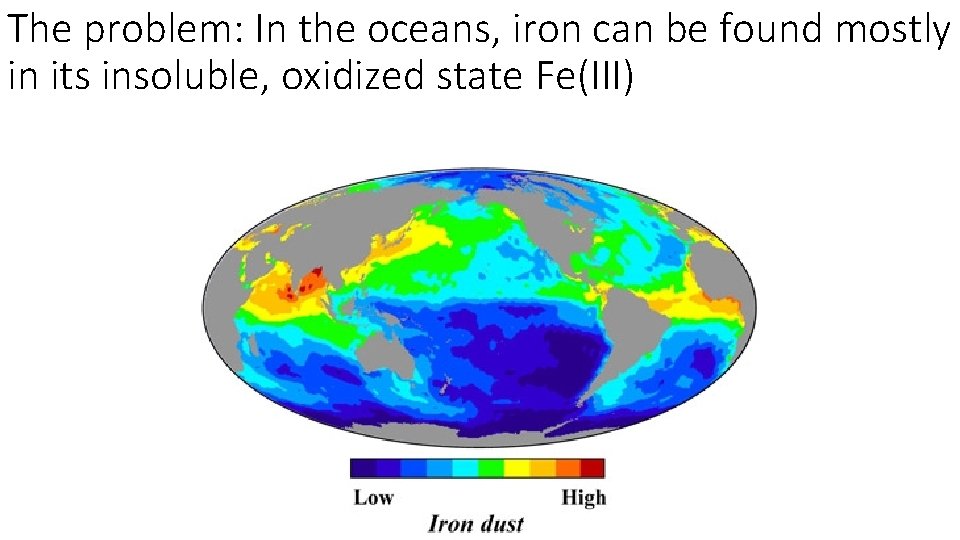
The problem: In the oceans, iron can be found mostly in its insoluble, oxidized state Fe(III)
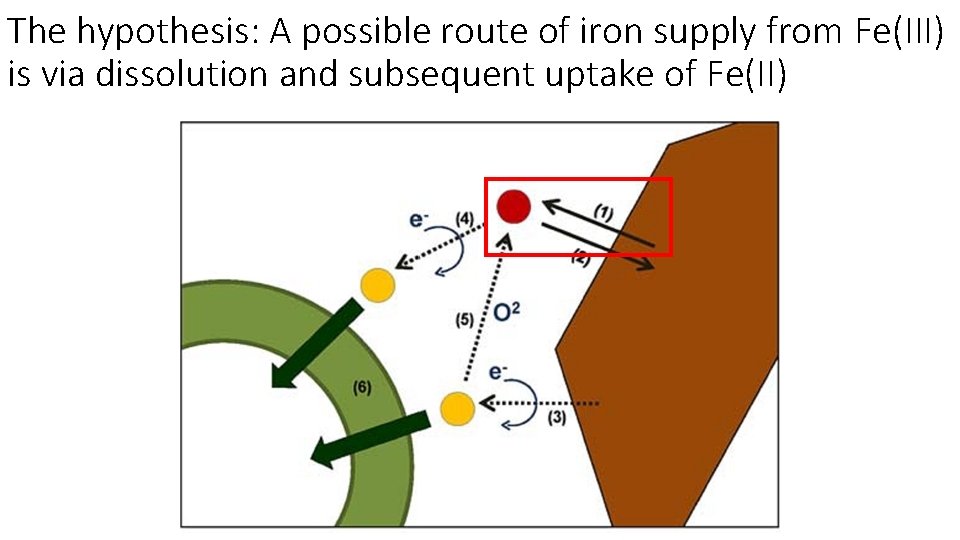
The hypothesis: A possible route of iron supply from Fe(III) is via dissolution and subsequent uptake of Fe(II)
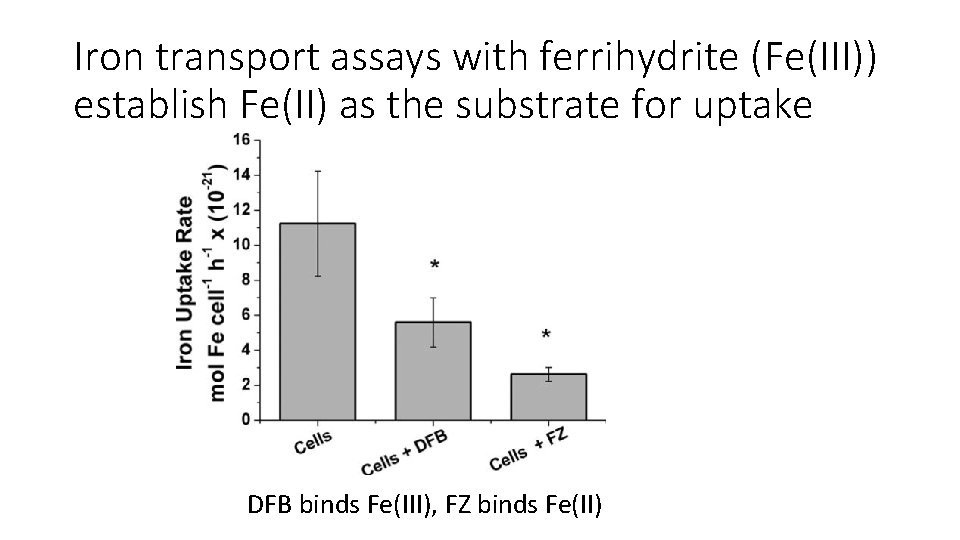
Iron transport assays with ferrihydrite (Fe(III)) establish Fe(II) as the substrate for uptake DFB binds Fe(III), FZ binds Fe(II)
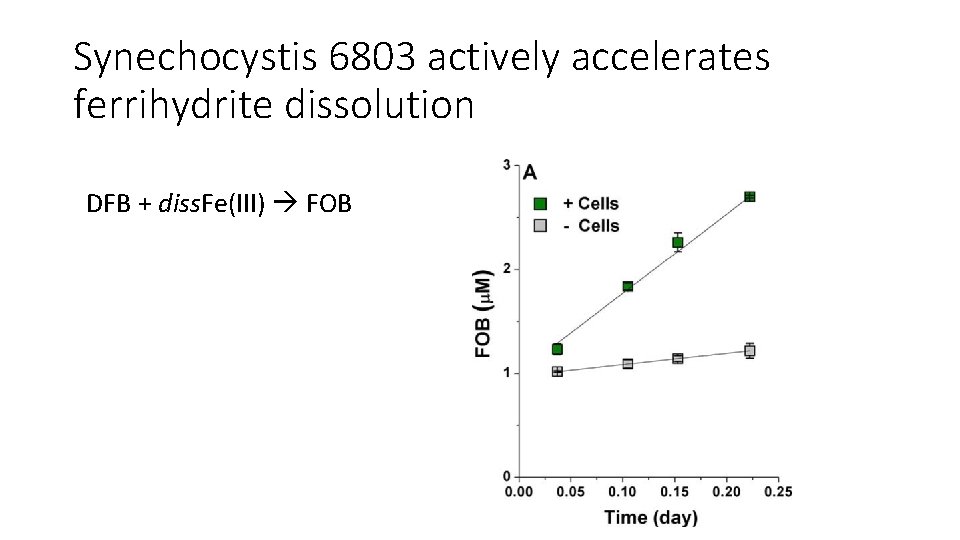
Synechocystis 6803 actively accelerates ferrihydrite dissolution DFB + diss. Fe(III) FOB
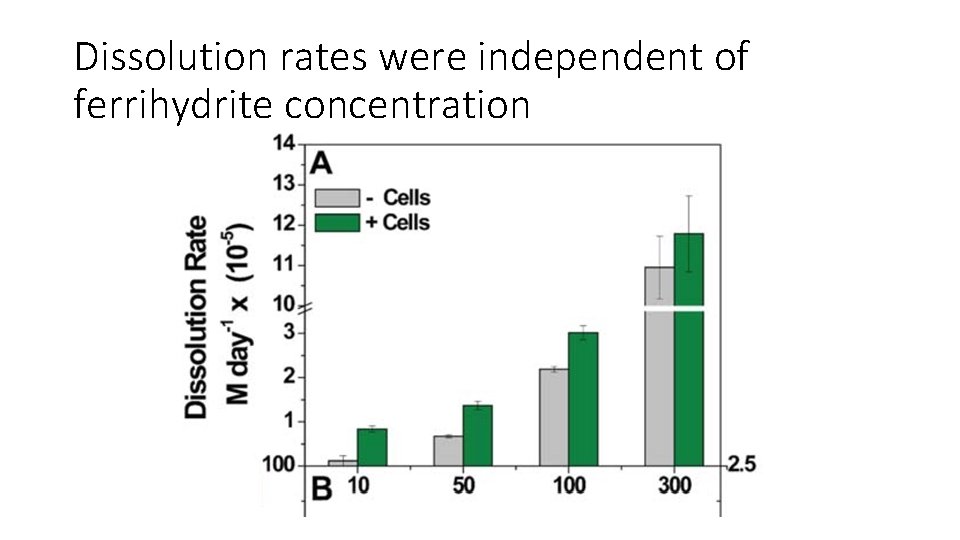
Dissolution rates were independent of ferrihydrite concentration

Biodissolution occurs via direct contact at the cellparticle interface or via close physical proximity ? ? ?

Biodissolution occurs via direct contact at the cellparticle interface or via close physical proximity
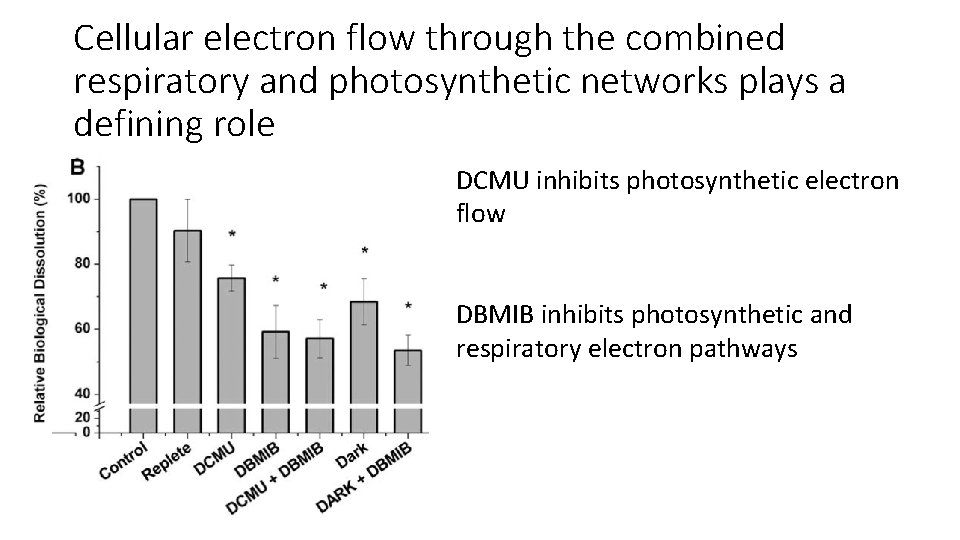
Cellular electron flow through the combined respiratory and photosynthetic networks plays a defining role DCMU inhibits photosynthetic electron flow DBMIB inhibits photosynthetic and respiratory electron pathways
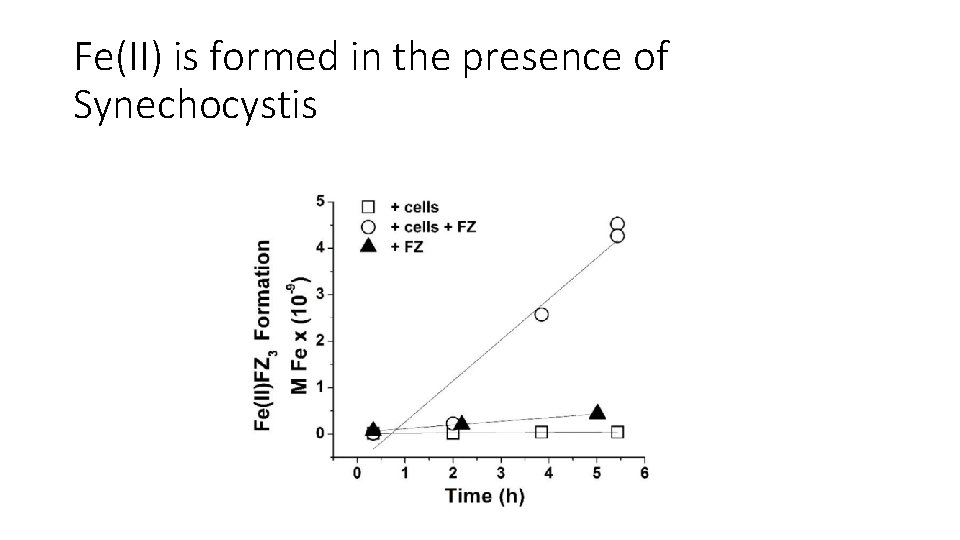
Fe(II) is formed in the presence of Synechocystis
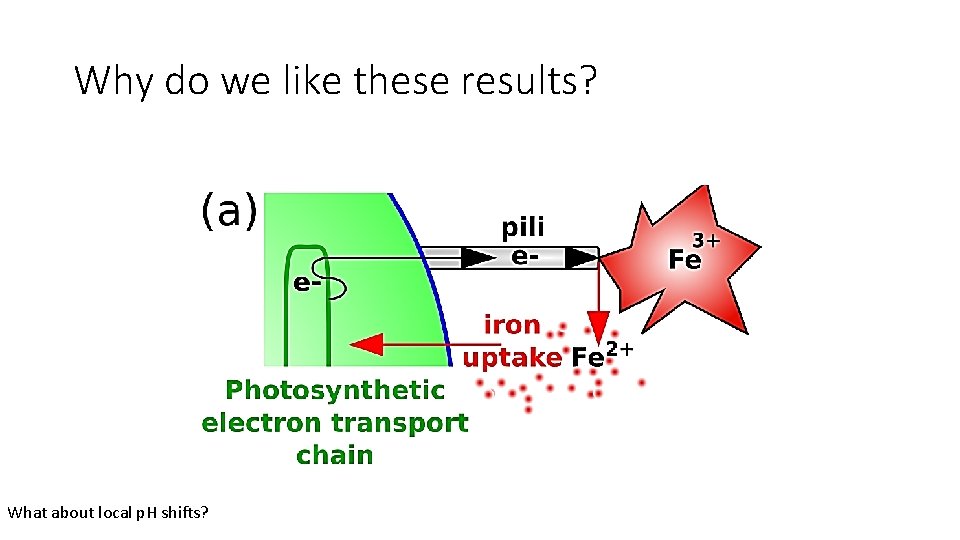
Why do we like these results? What about local p. H shifts?

Solubility is an endpoint. Dissolution is a process. [Remington Education: Physical Pharmacy, Pharmaceutical Press, 2016]
- Slides: 12