Enhanced Distillation Techniques Recommended also for Pressure Swing







- Slides: 22

Enhanced Distillation Techniques

(Recommended also for Pressure Swing & ED)

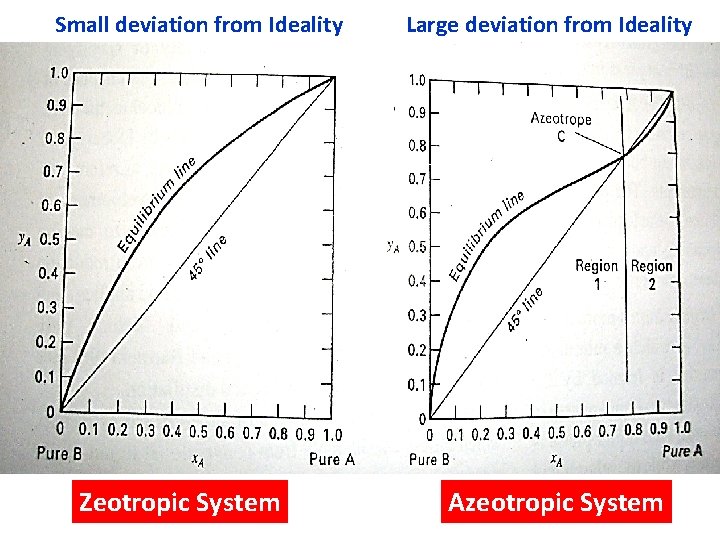
Small deviation from Ideality Zeotropic System Large deviation from Ideality Azeotropic System

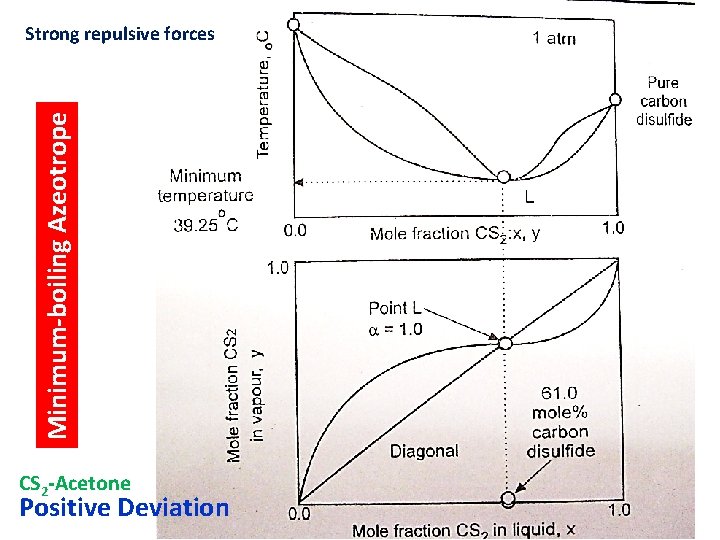
Minimum-boiling Azeotrope Strong repulsive forces CS 2 -Acetone Positive Deviation

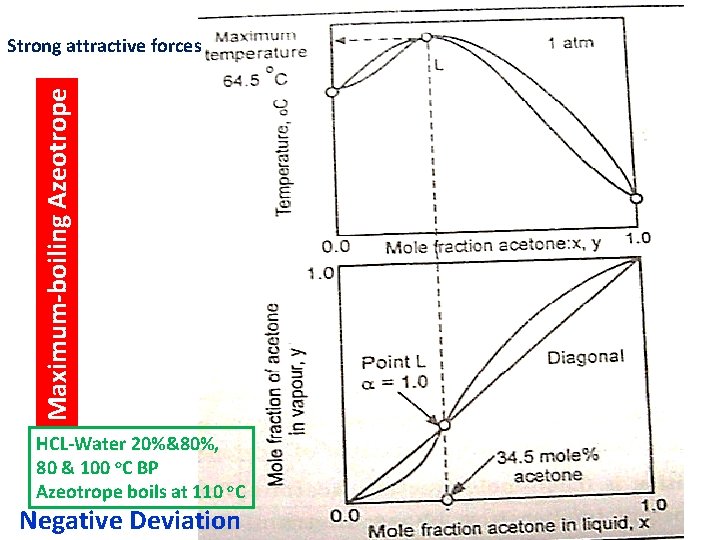
Maximum-boiling Azeotrope Strong attractive forces HCL-Water 20%&80%, 80 & 100 o. C BP Azeotrope boils at 110 o. C Negative Deviation

Ø Maximum-boiling Azeotropes are not separated using Azeotropic Distillation due to high energy requirement Ø Low-boiling Azeotropes can be separated using Azeotropic Distillation in two ways depending upon type of Entrainer: (a) Homogenous Low-boiling Azeotropic Distillation: In this distillation, Entrainer (E) breaks the azeotrope of feed (A&B) and form of new azeotrope (Entrainer+any one component of feed i. e. E&A) in distillation column. The E&A are available as a top product and B as a bottom Product. Here, E&A are miscible and has to be separated in next second distillation column. [EXTRA distillation & stripper columns are required for homogeneous solution of E&A] (b) Heterogeneous Low-boiling Azeotropic Distillation: In this distillation, Entrainer (E) breaks the azeotrope of feed (A&B) and form of new azeotrope (Entrainer+any one component of feed i. e. E&A) in distillation column. The E&A are available as a top product and B as a bottom Product. Here, E&A are immiscible so can be separated easily using decanter. [NO EXTRA distillation column is required for hetrogeneous solution of E&A]. In practice this type of Azeotropic Distillation is used due to economy.

Homogeneous AD: Benzene-Cyclohexane : 55&45%, Entrainer: 1 Butyl-3 -methylimidazolium thiocyanae For more examples of Azeotropes visit: https: //en. wikipedia. org/wiki/Azeotrope_tables

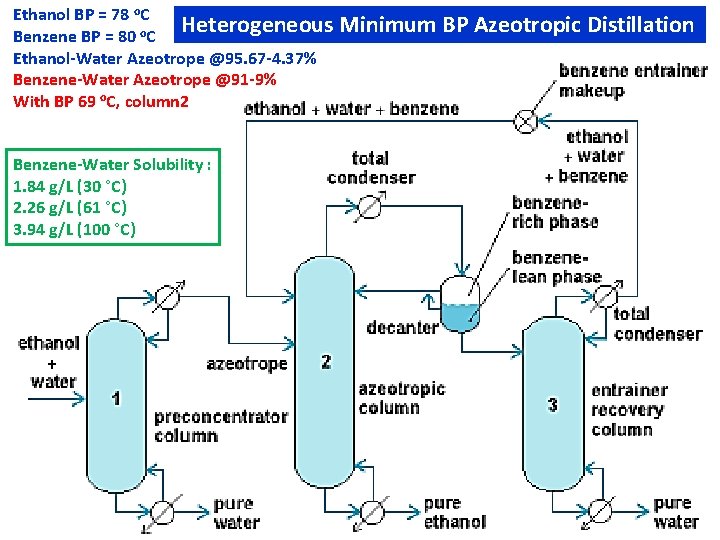
Ethanol BP = 78 o. C Heterogeneous Benzene BP = 80 o. C Ethanol-Water Azeotrope @95. 67 -4. 37% Benzene-Water Azeotrope @91 -9% With BP 69 o. C, column 2 Benzene-Water Solubility : 1. 84 g/L (30 °C) 2. 26 g/L (61 °C) 3. 94 g/L (100 °C) Minimum BP Azeotropic Distillation

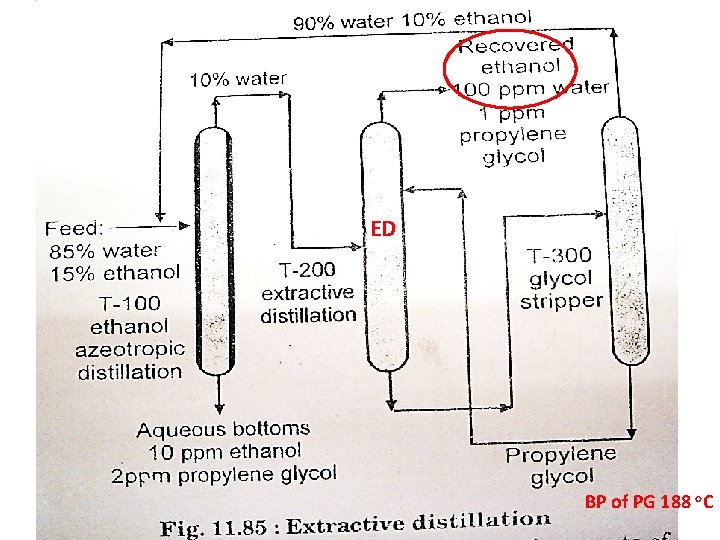
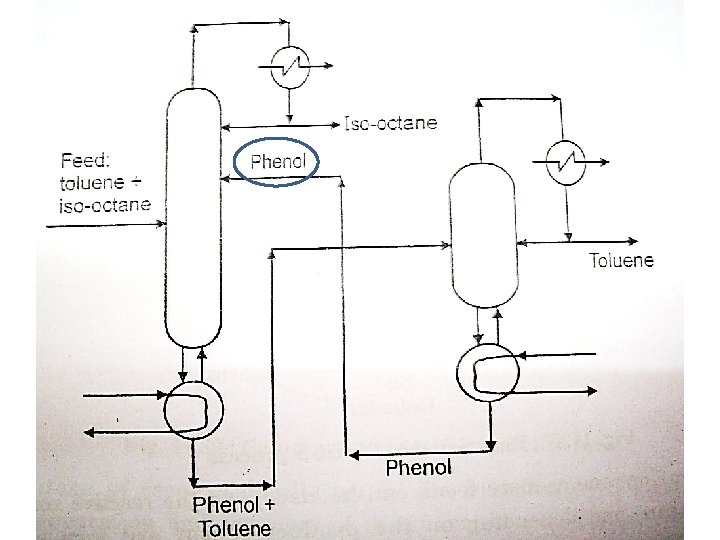
Extractive Distillation

ED BP of PG 188 o. C


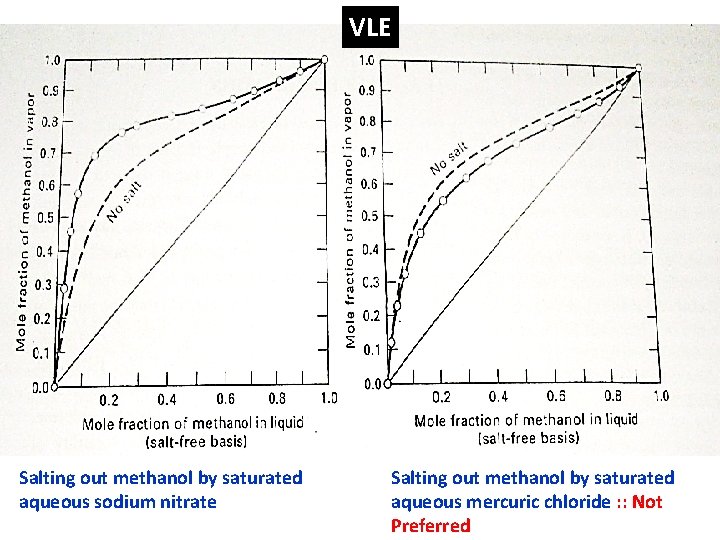
Salt Distillation One type of extractive distillation in which relative volatility of key components is altered by dissolving a soluble SALT in the top reflux

VLE Salting out methanol by saturated aqueous sodium nitrate Salting out methanol by saturated aqueous mercuric chloride : : Not Preferred

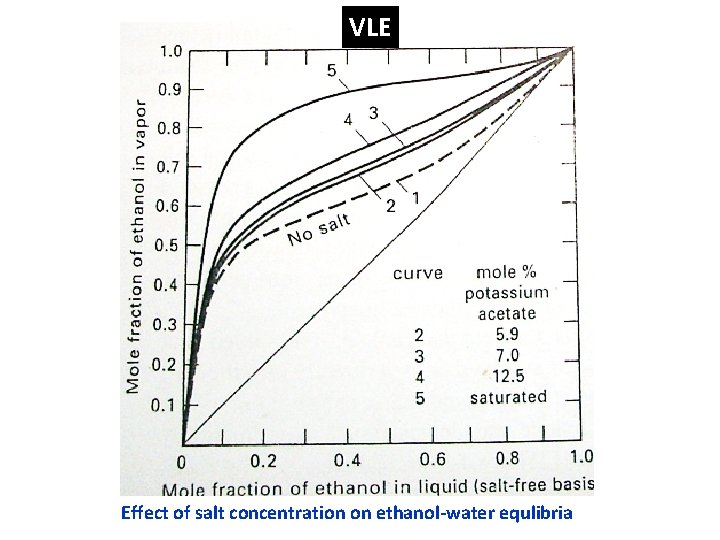
VLE Effect of salt concentration on ethanol-water equlibria

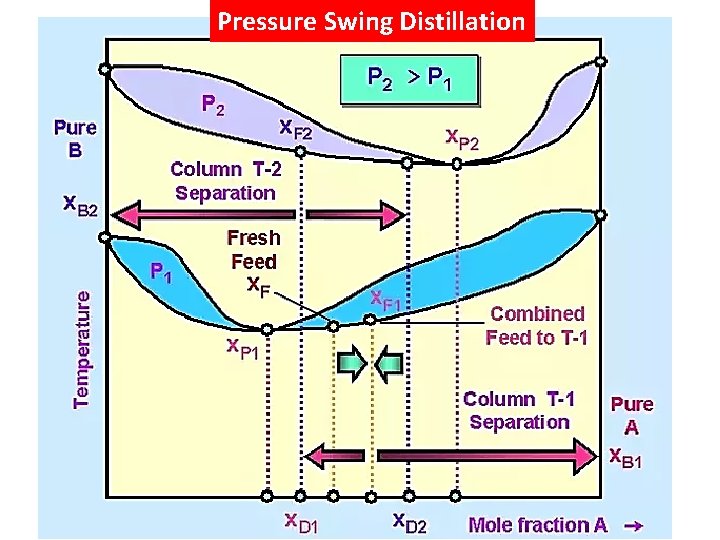
Pressure Swing Distillation

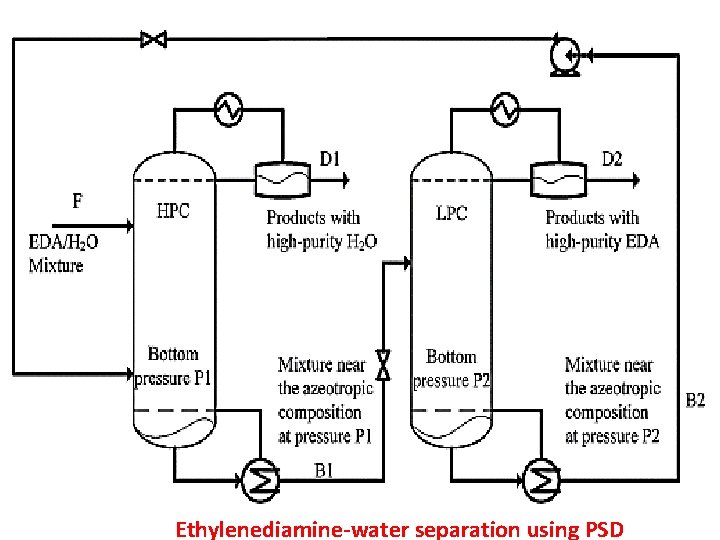
Ethylenediamine-water separation using PSD

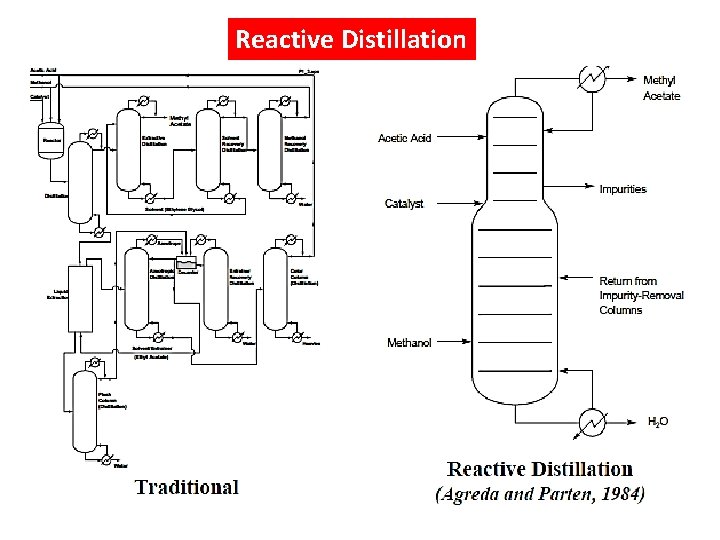
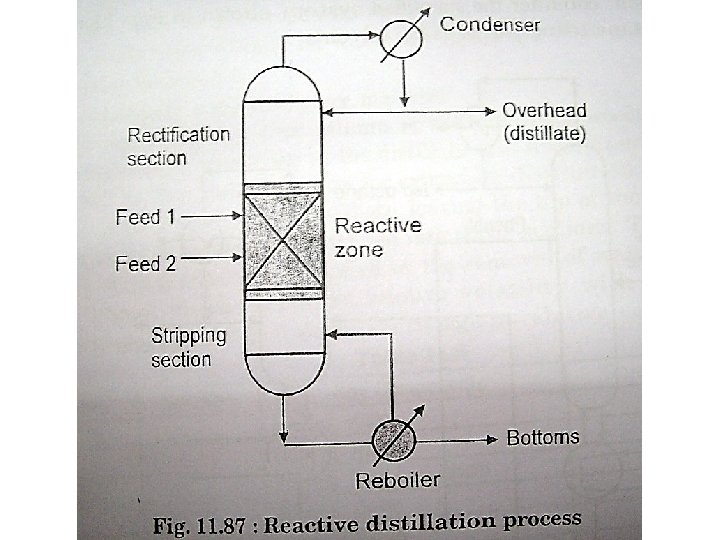
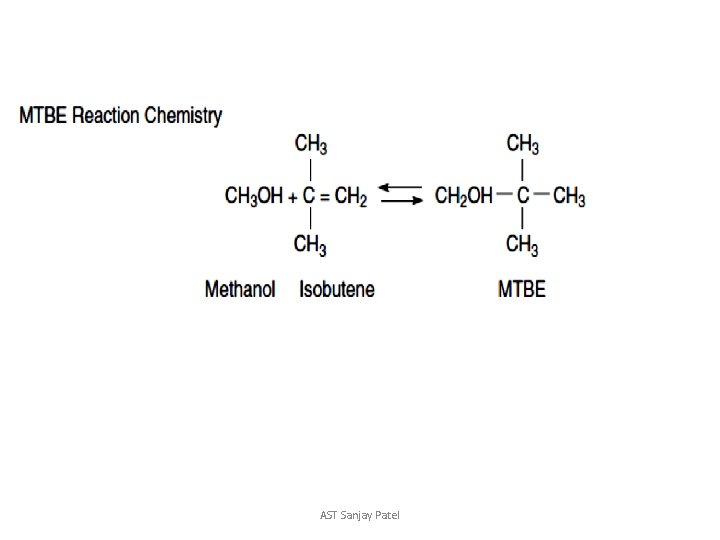
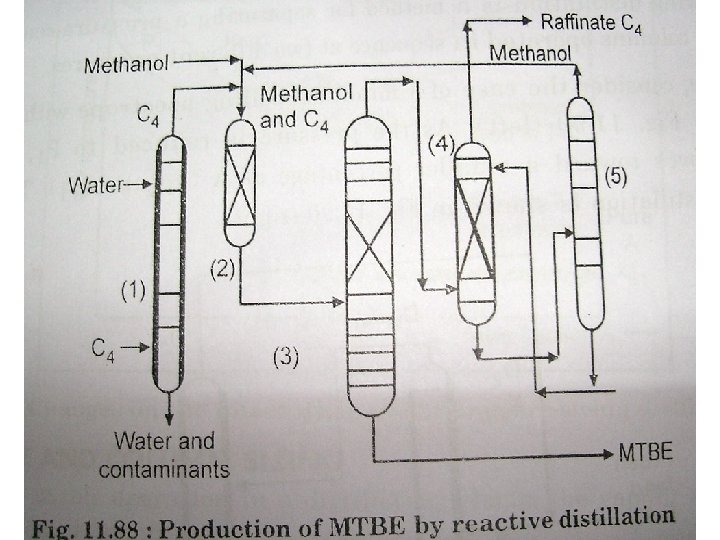
Reactive Distillation



AST Sanjay Patel


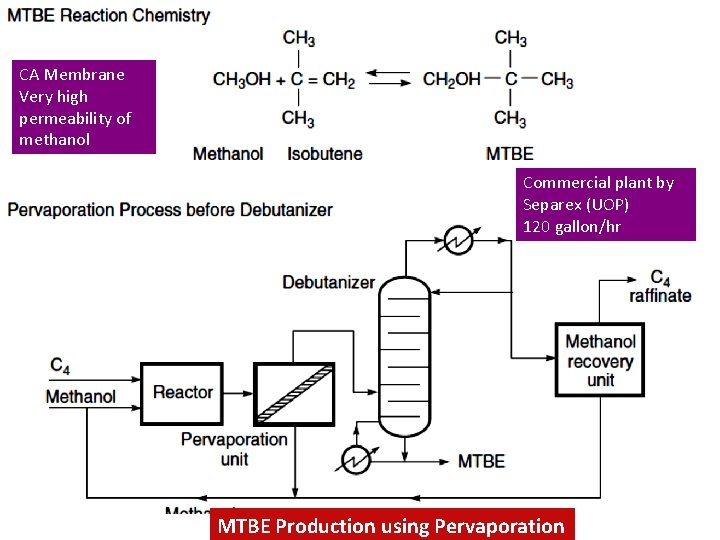
CA Membrane Very high permeability of methanol Commercial plant by Separex (UOP) 120 gallon/hr AST Sanjay Patel MTBE Production using Pervaporation