Engineering Polymers Prof Dr ElSayed Abdel Bary Assessment

Engineering Polymers Prof. Dr. El-Sayed Abdel Bary

Assessment Final examination, Exercises and Quizzes in Tutorials, and Reports • • • 15 3 quizzes 10 2 reports 10 ASSIGNMENT 25 Midterm exam 40 Final exam

Lecture 1 Introduction to Rubber Manufacturing and Technology

Contents • Origin of Rubber • Rubber Products • Some Common Elastomers • Requirements for Elastomers & Rubber
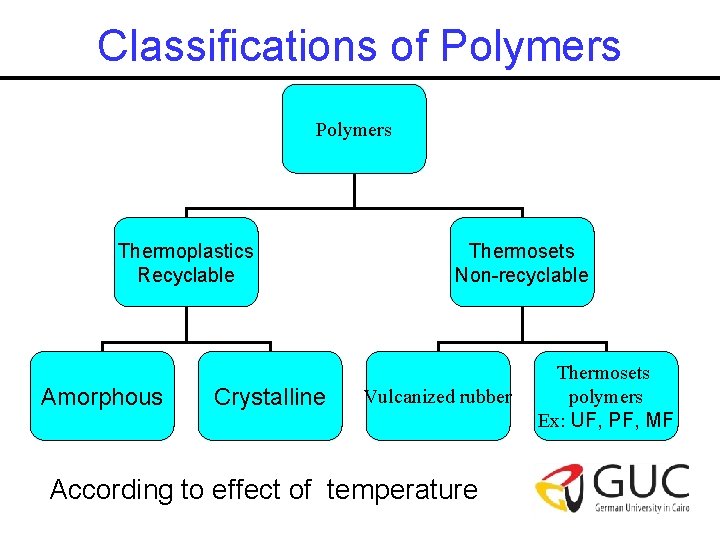
Classifications of Polymers Thermoplastics Recyclable Amorphous Crystalline Thermosets Non-recyclable Vulcanized rubber According to effect of temperature Thermosets polymers Ex: UF, PF, MF
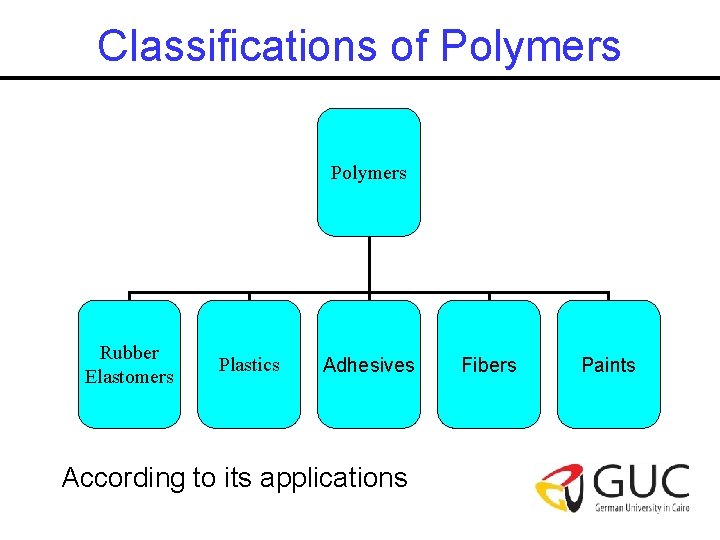
Classifications of Polymers Rubber Elastomers Plastics Adhesives According to its applications Fibers Paints
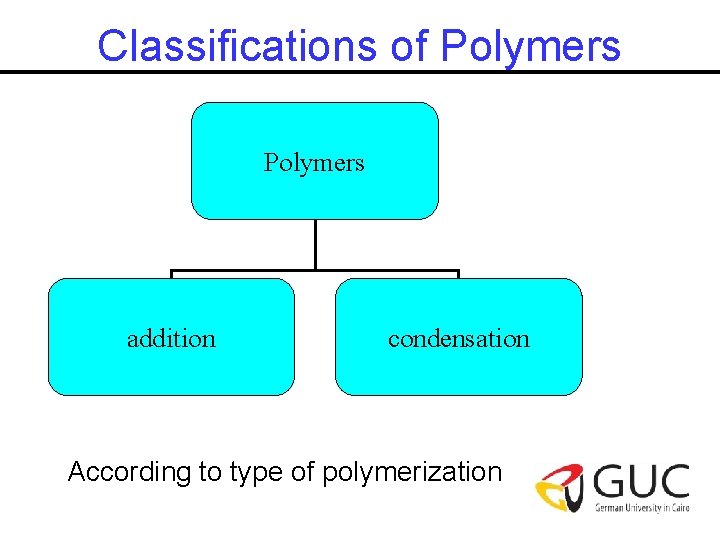
Classifications of Polymers addition condensation According to type of polymerization

Mechanical Properties of Polymers • In order to have a polymer with reasonable mechanical properties at room temperature, at least one of the following conditions should be fulfilled: • 1 -Tg of the polymer is above room temperature • 2 -Have a reasonable degree of crystallinity. • 3 -Crosslinked

Rubber • Rubber or elastomer is a unique material. It can be extended ten times its original length. • Its Tg is below room temperature and is amorphous. • Accordingly it should be in its cross-linked state to possess reasonable mechanical properties.

Requirements for Elastomers & Rubber ØCompletely amorphous, used above their Tg. ØLow intermolecular forces allow for flexibility. ØHigh modulus and strength when stretched often crystallize when stretched. ØLarge reversible extensions (several hundred percent) Ø High localized chain movements, low overall movement of chains relative to one another cross-linked chains prevent slippage.

Requirements for Elastomers & Rubber Ø Possess high molar mass to allow chain entanglements or are cross-linked Ø Gives dimensional stability Ø Cross-linked with sulfur Ø Reaction at allylic hydrogens( hydrogen of CH 2 group in the alpha position of double bond)

Rubber Products

Origin of Natural Rubber Hevea Rubber Tree: Hevea brasiliensis, Euphorbiaceae

Natural Rubber • Biological polymerization • Tg = -70 o. C • Primary use is auto tires, accounting for around 56% total consumption in 2005 • 2. 2 billion lb/yr in USA, all imported • hoses, belts and dampers for the automobile industry • Dipole moment, 1018 dyne ½. cm =0
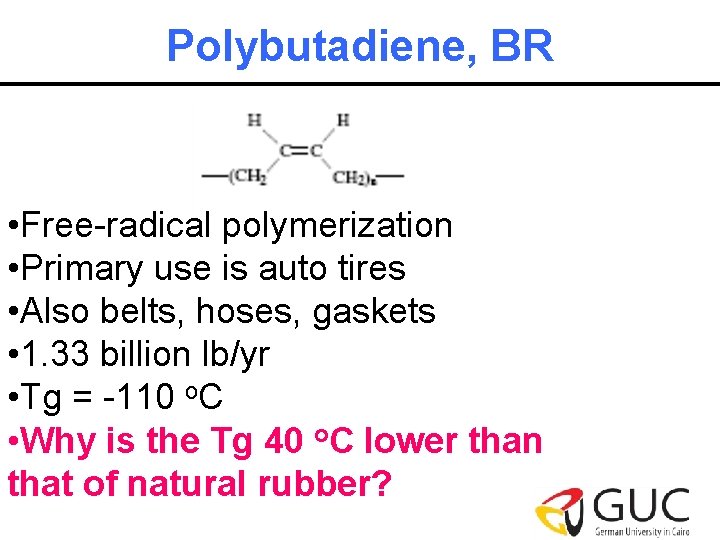
Polybutadiene, BR • Free-radical polymerization • Primary use is auto tires • Also belts, hoses, gaskets • 1. 33 billion lb/yr • Tg = -110 o. C • Why is the Tg 40 o. C lower than that of natural rubber?

Polychloroprene (CR) Applications • automotive products (fuel hoses, wire and cable covers), protective boots, bridge pads, roofing membranes, fabric coating and mechanical goods. • It is also flame-resistant. • The structure of CR is mostly trans-1, 4 and it crystallises upon standing or straining. The polar C-Cl bond enhances interchain interaction and promotes crystallisation. Because of its polar structure, polychloroprene has a relatively good resistance to oils. • Dipole moment, 1018 dyne ½. cm = 2. 01 The strain-crystallisation associated with its molecules is responsible for its excellent mechanical properties, comparable to those of natural rubber, however, crystallisation can also be a problem in prolonged exposure to low

Styrene-Butadiene Rubber, SBR • • • Free radical polymerization Mechanism? ? ? Tg = -65 o. C Primary use is auto tires Competes with NR

Acrylonitrile-butadiene rubber (nitrile rubber)(NBR) • Applications include fuel and oil tubing, • hoses, gaskets and seals, conveyor belting, printing rolls and pads. • oil-resistant rubber. The presence of polar a nitrile group (C=N) is responsible for the drastically increased resistance to oils, fuels, and solvents, which is superior to that of polychloroprene (CR). Furthermore, with increased acrylonitrile content, there is an increase in Tg, reduction in resilience, less die swell, decreased gas permeability, increased heat resistance, and increased strength. Heat ageing is quite good, and abrasion resistance is high. Dipole moment, 1018 dyne ½. cm = 3. 60
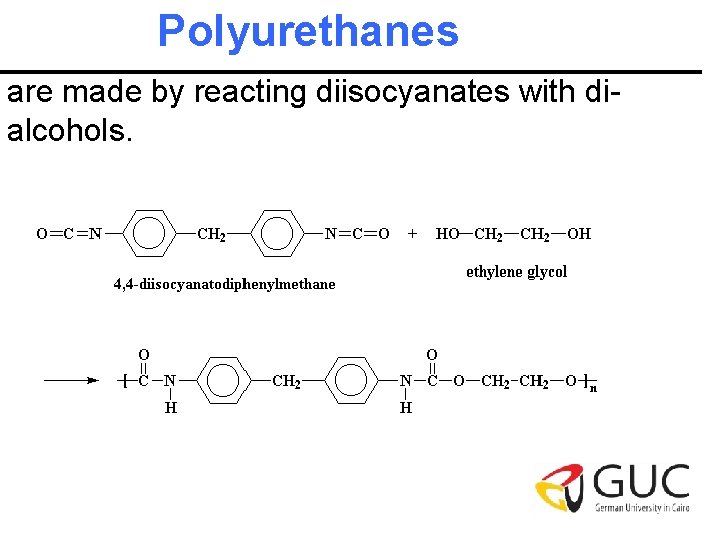
Polyurethanes are made by reacting diisocyanates with dialcohols.

Poly(styrene-butadiene-styrene)SBS Block copolymer • A hard rubber known as Th. ERMOPLASTIC ELASTOMER • Used for the soles of shoes, and other places where durability is important. The temperature during use should not exceed the Tg of polystyrene block. • It's a type of copolymer called a block copolymer.
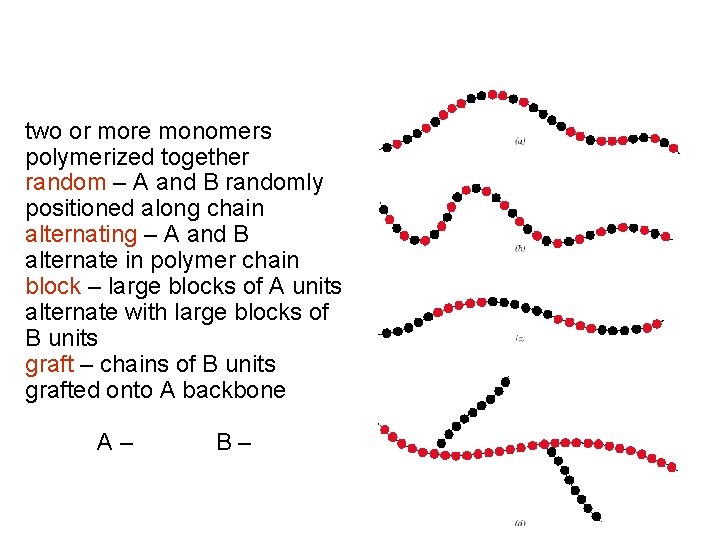
two or more monomers polymerized together random – A and B randomly positioned along chain alternating – A and B alternate in polymer chain block – large blocks of A units alternate with large blocks of B units graft – chains of B units grafted onto A backbone A – B –

Ethylene-propylene rubber (EPM, EPDM) EPMs have excellent resistance to weathering and ozone and good heat stability, but they do not allow conventional vulcanization with sulfur-based systems. While both polyethylene (PE) and polypropylene (PP) are crystallising plastics, random copolymers of ethylene and propylene are noncrystallising, and the low glass transition temperature of the copolymer is suitable for its use as a rubber. The percentage of ethylene and propylene repeat units is one of the factors determining the properties of the copolymers (reported values are range from 75/25 to 50/50 for PE and PP). Typical Tg is -60 o. C.

Ethylene-propylene rubber (EPM, EPDM) • The limitations of EPMs is mostly associated with low resistance to oils and fuels, poor adhesion to many substrates or reinforcements. They improve, in particular, low temperature properties, resilience, and abrasion or wear resistance of compounds.

Applications of EPDM

Silicon Rubber • in silicone grease and other silicone based lubricants, • defoaming agents, damping fluids, • heat transfer fluids, • cosmetics, hair conditioner. • PDMS has also been used as a filler fluid in breast implants, although this practice has decreased somewhat, due to safety concerns. • It continues to be used in knuckle replacement implants, with good results.
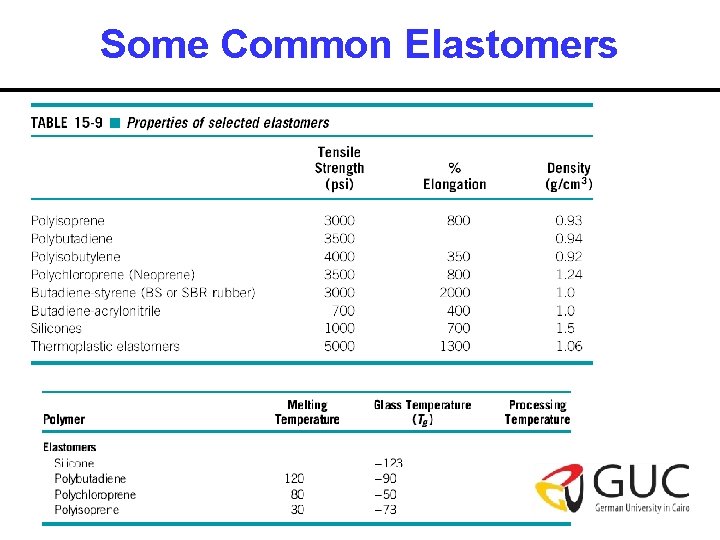
Some Common Elastomers
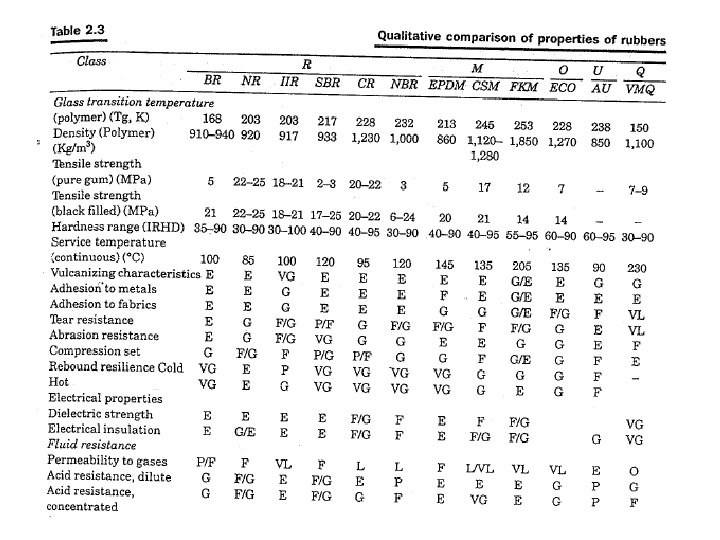
- Slides: 27