Engineering Chemistry revised edition ISBN 978 81 265

Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Introduction Nanotechnology can be defined as the manipulation of atoms and molecules at nano (one billionth) scale (1 -100 nm) to produce devices, structures or systems having at least one novel or superior property. Nanomaterials: . The materials having at least one dimension in the nano scale are called Nanochemistry or Nanotechnology related with the production and the reactions of nanoparticles and their compounds. ØConcept was given by Richard Feynmann in 1959.
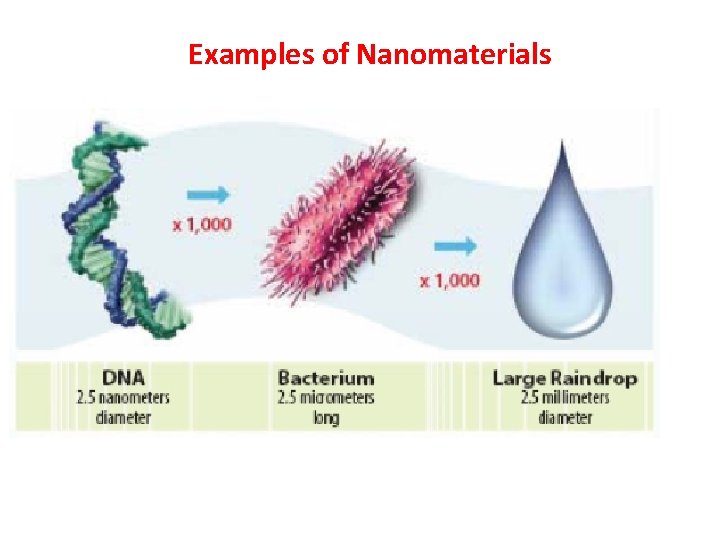
Examples of Nanomaterials




Properties of Nanomaterials are different from bulk material because: 1. The surface area to volume ratio of the nanomaterials is relatively larger than that of bulk materials of the same mass. This increases the chemical reactivity and affects strength and electrical properties of the material. 2. The quantum confinement is observed at nanometer sizes that changes the optical, electronic and magnetic properties of the material. The band gap increases as the size of the material is reduced to nanometer range. This effect is caused by the phenomenon resulting from electrons and electron holes being constricted into a dimension which approaches the critical quantum measurement, known as the exciton Bohr radius.



This science use methodologies from the synthetic chemistry and the material's chemistry to obtain nanomaterials with specific sizes, shapes, surface properties, defects, self-assembly properties, designed to accomplish specific functions and uses. The combination of number of possible materials coupled with the concepts of nanochemistry is very amazing. A few of them are: 1. Nanoparticles of various sizes and shapes such as, gold, silver nanoparticles. 2. Nanooxides such as iron oxide, cadmium oxide, Ti. O 2 3. Carbon nanotubes and fullerenes 4. Nanopolymers and nanomembranes 5. Nanocatalysts 6. Nanoformulations for cosmetics, example, in sunscreens, anti-aging creams, etc. 7. Nanoceramics Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

These materials have, in turn, applications in various domains: 1. In semiconductor devices, such as nanocapacitors 2. As therapeutic molecules (nanomedicine) for diseases such as cancer. 3. In targeted drug delivery 4. In agriculture 5. In electronics 6. As taste enhancers (nanofoods) and for packaging in food industry 7. In fuel cells 8. In chemical and biosensors 9. In fabric technology 10. In solar cells 11. Nanooptics 12. For manufacture of self-cleaning surfaces 13. In waste water treatment 14. Green nanotechnology Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
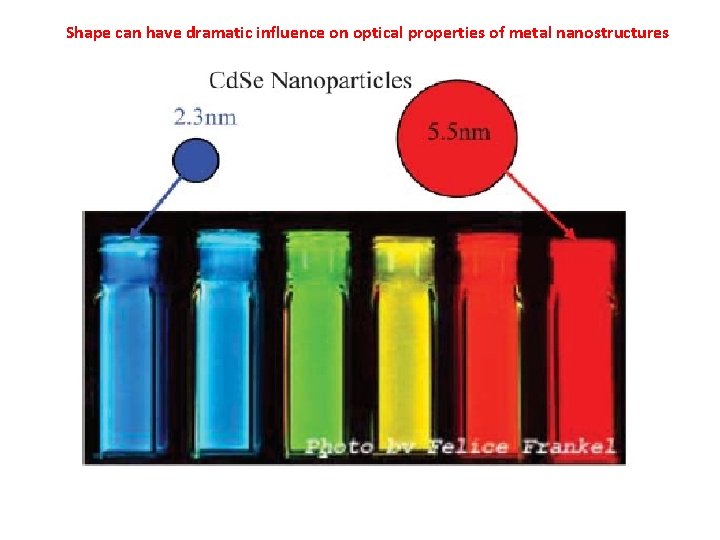
Shape can have dramatic influence on optical properties of metal nanostructures

Size-Dependent Properties Particle-Size vs. Surface Area If a macroscopic object is divided into smaller parts, the ratio of surface atoms to interior atoms becomes a significant number of the total fraction of atoms. There is inverse relation between Particle. Size and Surface Area. Surface Chemistry of Materials Due to increased surface area, nanomaterials possess greater surface energy and are thermodynamically less stable. The atoms or molecules that exist on the surface or the interface are somewhat different from atoms/molecules in the bulk/interior of the material. These have enhanced reactivity and greater tendency to agglomerate. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Electronic Properties Electrical Properties of Nanoparticles” discuss about fundamentals of electrical conductivity. Various physical properties such as thermal, optical and magnetic properties are dependent on the electronic state of the material. Some characteristics of electronic structure of solids are: 1. In an isolated solid, the electrons move in quantified energy levels. When the distance between atoms is less, electron orbitals interact with each other, which leads to broadening of energy levels to form energy bands. 2. The inner-shell electrons form narrow bands called internal bands and electrons in external shell form valence bands. The electrons in excited state form conduction band. 3. The difference of energy between valence band conduction band is called energy gap. In metals (or conductors) this energy gap is zero; it is small in case of semiconductors and large in case of insulators. 4. The maximum energy for electrons at absolute zero (0 K) is called Fermi level or Fermi energy. The physical properties of materials are mainly governed by electrons that have energy larger than Fermi energy.
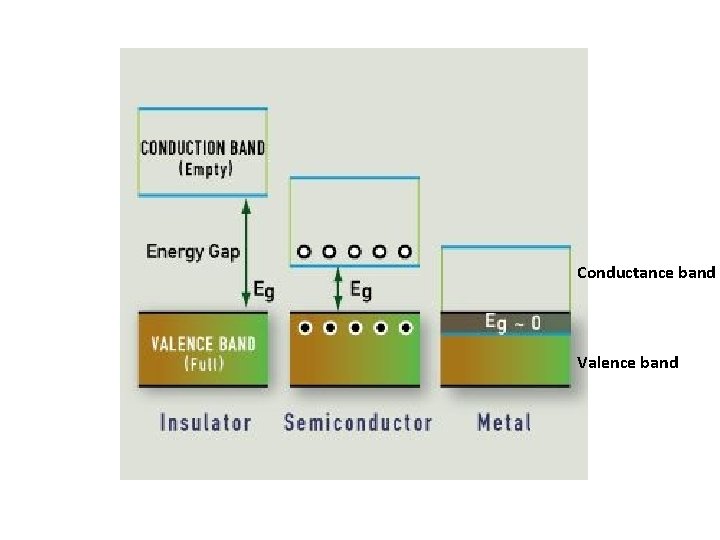
Conductance band Valence band

Mechanical Properties Nanocrystalline materials have an average crystallite size in the range 1 to 100 nm and are characterized by numerous grain boundaries due to the small size of the grain. The mechanical properties of nanocrystalline materials are determined by their small grain size and the grain boundary. These properties are enhanced by reducing the grain size, as grains of nano-size have no defects inside, unlike micro -grains of relatively larger size. For example, nanocrystalline copper is found to be three times more resistant to applied stress than normal copper crystals and deformed homogeneously. The crystallinity of the grain structure is maintained right up to the grain boundary. The fraction of atoms out of the total volume present at the grain boundaries is large for small size grains and decreases with increase in their size.

Fullerenes Two forms of fullerenes: (a) Bucky ball form; (b) nanotube form. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Characteristics of Fullerenes (C 60) • It is fine black powder • Its density is 1. 63 g/cm 3 • It sublimes at 800 K • It is insoluble in water. It is sparingly soluble in organic solvent such as benzene, toluene, carbon disulphide etc. • It different solvents in impart different colour e. g. in toluene it gives bright red colour • It is insulator as movements of electron from one molecule to another molecule is not posible. Because there are only weak van der waal’s force of attractions exist between different molecules. • Chemically fullerenes are stable; breaking the balls requires temperatures over 1000°C.

Characteristics of Fullerenes (C 60) (Contd. . ) • However, fullerenes participates in chemical reactions as they possess Π -electrons which are free to localise or delocalise in different chemical situations, reactions such as addition reactions and redox. • An important characteristic of C 60 molecule is its high symmetry with 120 symmetrical operations which map the molecule onto itself. Each carbon atom bonds with three other adjacent atoms using sp 2 hybridization. • Fullerenes themselves are non-toxic but some of the derivatives of fullerenes could be harmful to the health. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Applications of Fullerenes 1. Oxidation involving free radicals causes cell damage, food spoilage, plastics deterioration, metal corrosion, etc. Therefore, fullerenes due to powerful antioxidant property (they reportedly react readily at a high rate with free radicals), can find application in health and personal care areas. 2. Fullerene nanotubes are promising as chemical molecular sensors, due to their small size, room temperature operation and high sensitivity. They find application as coating materials on some chemical sensors, such as quartz crystal microbalance and surface acoustic wave sensors (SAW). 3. Fullerenes and its modified forms are capable of targeted and controlled delivery of drugs and genes into cells. 4. Other applications of fullerenes like catalysts in certain organic reactions, water purification, fuel cell membrane material are also being explored. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
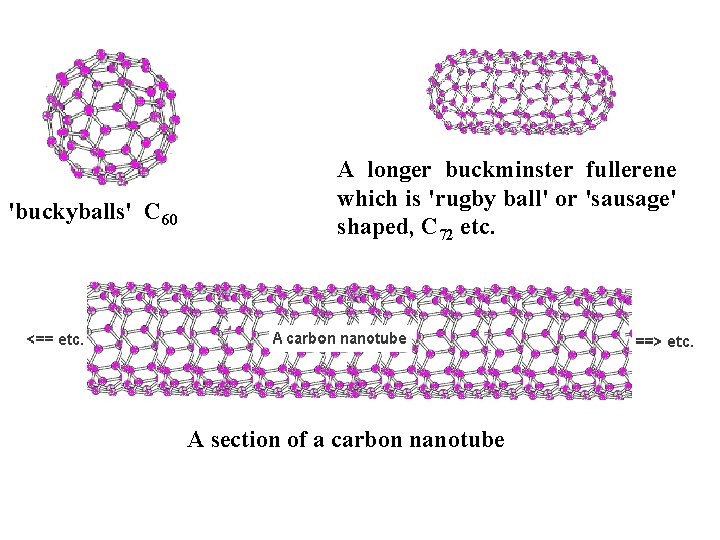
'buckyballs' C 60 A longer buckminster fullerene which is 'rugby ball' or 'sausage' shaped, C 72 etc. A section of a carbon nanotube
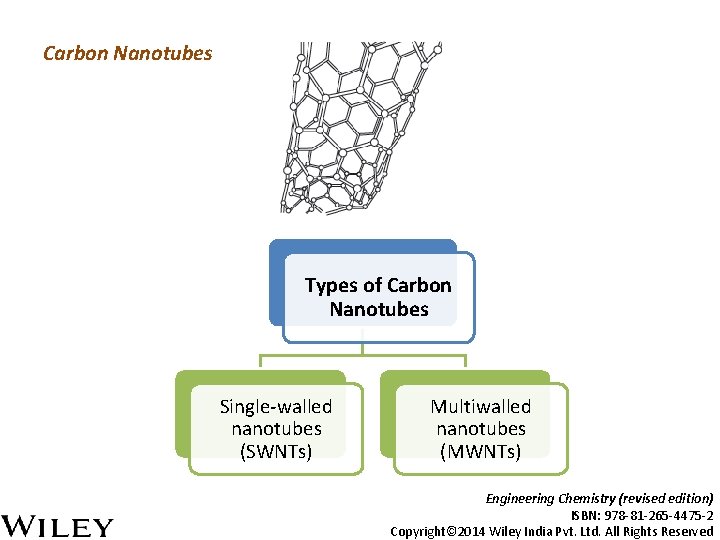
Carbon Nanotubes Types of Carbon Nanotubes Single-walled nanotubes (SWNTs) Multiwalled nanotubes (MWNTs) Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

A "zig-zag" carbon nanotube An "armchair" carbon nanotube.
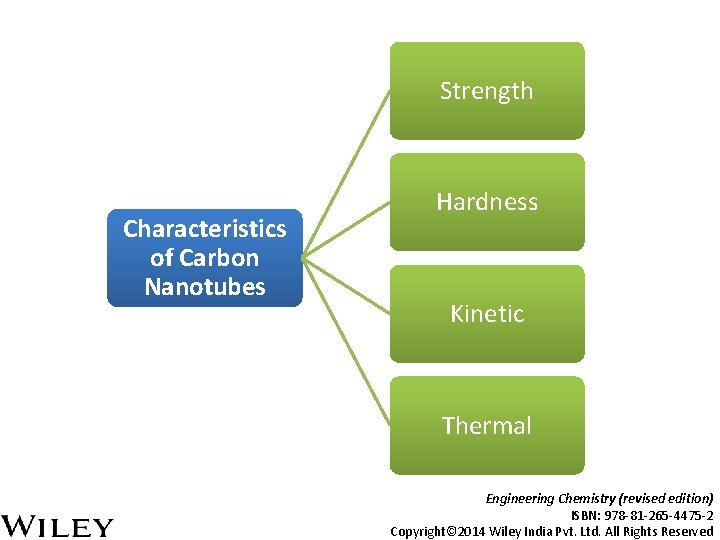
Strength Characteristics of Carbon Nanotubes Hardness Kinetic Thermal Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

General Applications of Nanomaterials The impact of nanotechnology and use of nano-materials extends from healthcare, medicine, energy, agriculture, food to fields such as electronics, communication, optics, chemistry and computing. In Medicine The size of nanomaterials is very similar to that of biological molecules and structures; therefore, these find extensive use in both in vivo and in vitro biomedical applications. The integration of nanomaterials with biology has led to development of various diagnostic devices, contrast agents, analytical tools, physical therapy applications and drug delivery vehicles. 1. Diagnostics: 2. Drug delivery 3. Tissue repair 4. Transdermal drug delivery Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

In Electronics and Communication Nanotechnology has brought revolution in telecommunication, computing and networking industries. The principle aim of nanotechnology is to produce three dimensional confined quantum structured electronic devices such as quantum wires and quantum dot devices. Some successful devices are: 1. Quantum well lasers for telecommunication. 2. High electron mobility transistors (HEMT) with low noise. 3. High gain microwave application and vertical cavity surface emitting lasers for data communication and sensor coding. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Some consumer products using nanomaterials are: 1. Computer hardware 2. Display devices. 3. Mobile and communication products. 4. Audio products. 5. Cameras and films, etc. Nanomaterials are used to develop semiconductors with much advanced properties. These include: 1. Transistors from carbon nanotubes with nanometer dimensions. 2. Memory chips with density of one tetrabyte per square inch. 3. Very high speed transistors from single atom thick graphene film. 4. Lightweight nanoemissive display panel using carbon nanotubes. 5. Nomfet (nanoparticle organic memory field-effect transistor) obtained by combining gold nanoparticles with organic molecules. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

In Energy Science Nanomaterials, particularly nanocomposites, are increasingly being used to replace conventional materials for conversion of energy. Some examples of their use are: 1. Non-porous crystalline solids called zeolites are used for refining of oil. 2. Quantum dots can be used as sources of light emissions. 3. Electrode materials can be changed by nano-structuring, that is, can be imparted nano-texture to improve electrical performance of batteries. 4. Energy production can be brought about using nanomaterials as clean and high efficiency energy sources. 5. Solar cells can be integrated from nanoscale crystals of semiconductors coated with light-absorbing dye-emitting electrons and from nanostructured diamond thermal cells that capture heat and light from the lattice and emit high-energy electrons. 6. Ultraporous nanomaterials are being explored for storing hydrogen at high -densities for fuel-cell powered cars. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

In Catalysis Heterogeneous catalysts used in industry today, consist of small particles (1 -10 nm) of catalytically active material attached to porous support. Nanoparticles of metals, semiconductors, oxides, and other compounds are widely used for important chemical reactions. There are four main categories of nano-catalysts based on the composition. These include: 1. Gold-based (e. g. , Au/Ti. O 2, Au/Mg. O) 2. Other metal based (e. g. , Cu, Rh, ) 3. Platinum or palladium based (e. g. , Pt/Ru/Ni) 4. Nanosize metal particles dispersed Pt/polypyrrole) in polymers (e. g. , Cu/polyaniline, Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved

Miscellaneous Applications 1. 2. 3. 4. 5. 6. 7. Nano zinc oxide and titanium oxide are used in sunscreen creams, lotions and other cosmetics. These nanoparticles become transparent and retain their ability to absorb UV radiation. Nanoparticles of tungsten-carbide–cobalt provide a hard coating. This results in a greater resistance to wear in applications such as drill bits, cutting tools and jet engine parts. Nano coating over glass produces self-cleaning glass, which becomes transparent or opaque according to the current applied. Nanotechnology finds applications in energy storage and its conversion. Nanomaterials are investigated as electrode materials in batteries and fuel cells. Nanotubes also offer enhanced hydrogen storage capability for the use in fuel cells. Nanotechnology finds applications in the production, processing, safety and packaging of food stuffs. Nanofibers in clothes make waterproof and stain-repellant or wrinkle-free and can be washed less frequently. Engineering Chemistry (revised edition) ISBN: 978 -81 -265 -4475 -2 Copyright© 2014 Wiley India Pvt. Ltd. All Rights Reserved
- Slides: 30