Engine emission control by Three way catalytic converter

Engine emission control by Three way catalytic converter system Unit-II

Catalytic converter (CATCON): • Pollutant gases are made of harmful molecules, but those molecules are made from relatively harmless atoms. So if we could find a way of splitting up the molecules after they leave a car's engine and before they get pumped out into the air, we could crack the problem of pollution. That's the job that a catalytic converter does.

Catalyst: • A catalyst is simply a chemical that makes a chemical reaction go faster without itself changing in the process. • It's a bit like an athletics coach who stands by the side of the track and shouts at the runners to go faster. The coach doesn't run anywhere; he just stands there, waves his arms about, and makes the runners speed up. • In a catalytic converter, the catalyst's job is to speed up the removal of pollution. The catalyst is made from platinum or a similar, platinum-like metal such as palladium or rhodium.

Catalytic converter construction: • A catalytic converter is a large metal box, bolted to the underside of the car, that has two pipes coming out of it. • One of them (the converter's "input") is connected to the engine and brings in hot, polluted fumes from the engine's cylinders (where the fuel burns and produces power). • The second pipe (the converter's "output") is connected to the tailpipe (exhaust). • As the gases from the engine fumes blow over the catalyst, chemical reactions take place on its surface, breaking apart the pollutant gases and converting them into other gases that are safe enough to blow harmlessly out into the air.

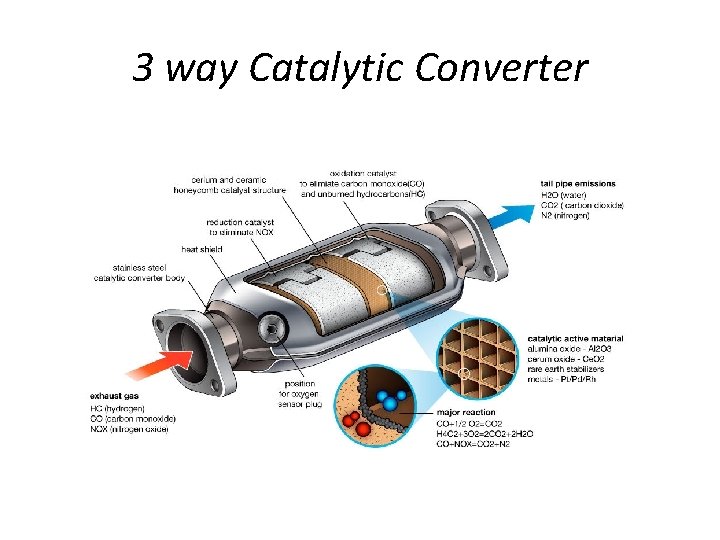
3 way Catalytic Converter
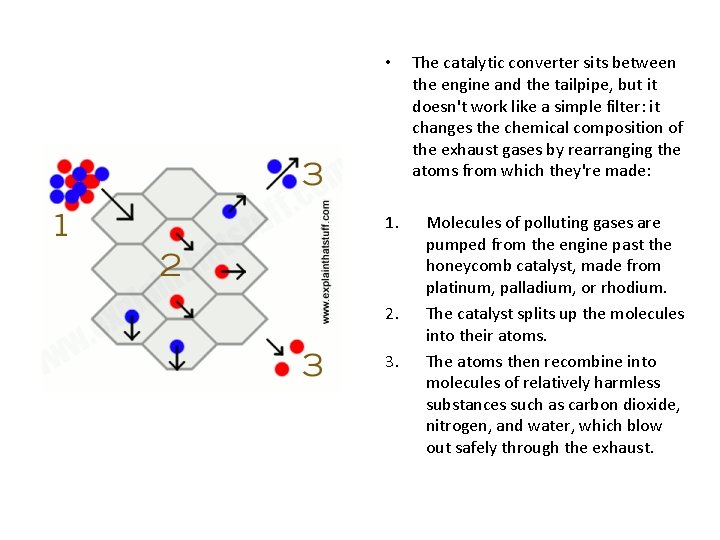
• 1. 2. 3. The catalytic converter sits between the engine and the tailpipe, but it doesn't work like a simple filter: it changes the chemical composition of the exhaust gases by rearranging the atoms from which they're made: Molecules of polluting gases are pumped from the engine past the honeycomb catalyst, made from platinum, palladium, or rhodium. The catalyst splits up the molecules into their atoms. The atoms then recombine into molecules of relatively harmless substances such as carbon dioxide, nitrogen, and water, which blow out safely through the exhaust.
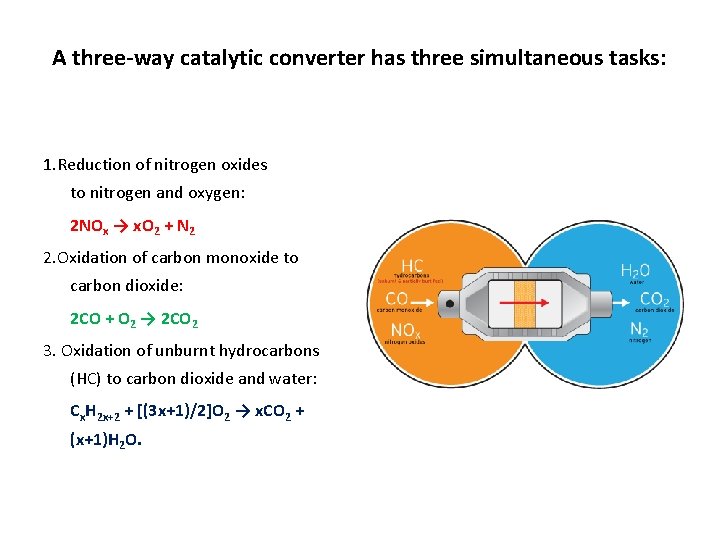
A three-way catalytic converter has three simultaneous tasks: 1. Reduction of nitrogen oxides to nitrogen and oxygen: 2 NOx → x. O 2 + N 2 2. Oxidation of carbon monoxide to carbon dioxide: 2 CO + O 2 → 2 CO 2 3. Oxidation of unburnt hydrocarbons (HC) to carbon dioxide and water: Cx. H 2 x+2 + [(3 x+1)/2]O 2 → x. CO 2 + (x+1)H 2 O.
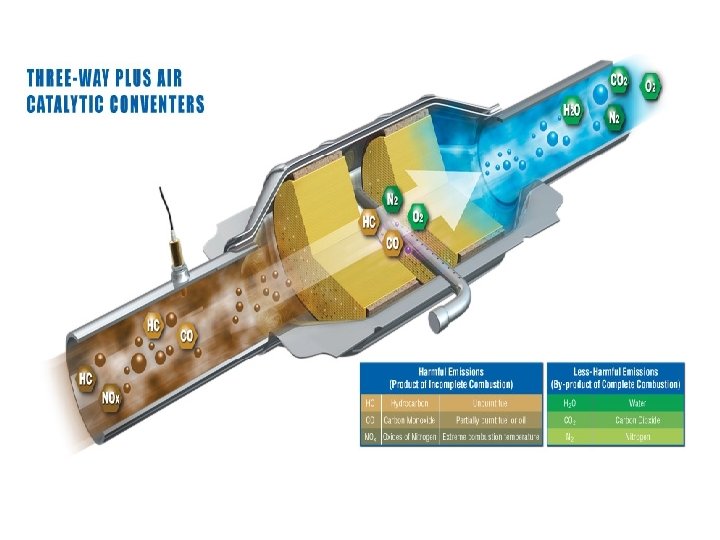

Additional Information: • Closed-loop engine control systems are necessary for effective operation of three-way catalytic converters because of the continuous balancing required for effective NOx reduction and HC oxidation. The control system must prevent the NOx reduction catalyst from becoming fully oxidized, yet replenish the oxygen storage material so that its function as an oxidation catalyst is maintained. • Three-way catalytic converters can store oxygen from the exhaust gas stream, usually when the air–fuel ratio goes lean. When sufficient oxygen is not available from the exhaust stream, the stored oxygen is released and consumed. • A lack of sufficient oxygen occurs either when oxygen derived from NOx reduction is unavailable or when certain maneuvers such as hard acceleration enrich the mixture beyond the ability of the converter to supply oxygen.
- Slides: 10