engel Boles Thermodynamics An Engineering Approach Third Edition

Çengel Boles Thermodynamics An Engineering Approach Third Edition Yunus A. Çengel Michael A. Boles Third Edition WCB/Mc. Graw-Hill © © The Mc. Graw-Hill Companies, Inc. , 1998

1 CHAPTER Çengel Boles Thermodynamics Basic Concepts of Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
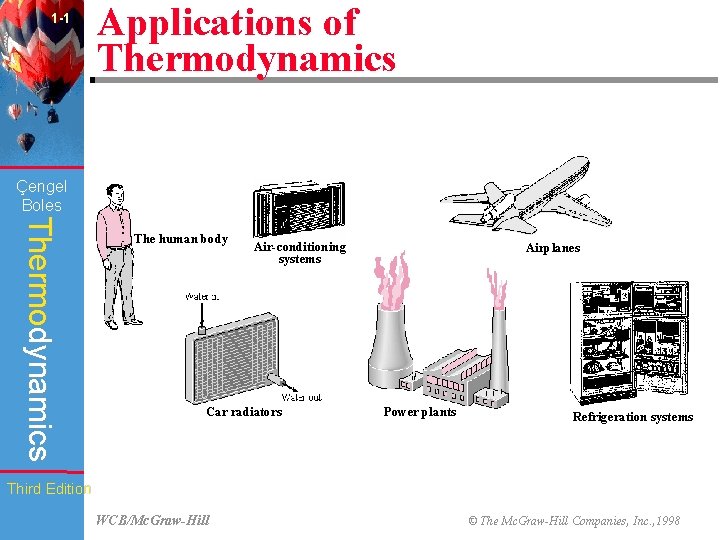
1 -1 Applications of Thermodynamics Çengel Boles Thermodynamics The human body Air-conditioning systems Car radiators Airplanes Power plants Refrigeration systems Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
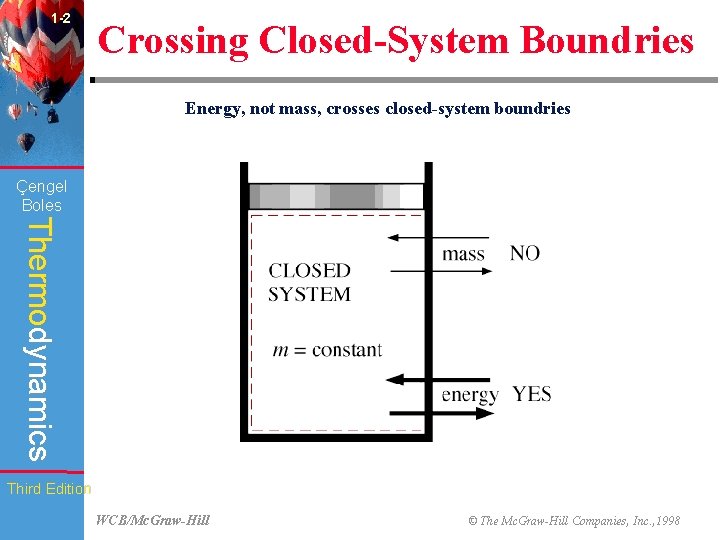
1 -2 Crossing Closed-System Boundries Energy, not mass, crosses closed-system boundries (Fig. 1 -13) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
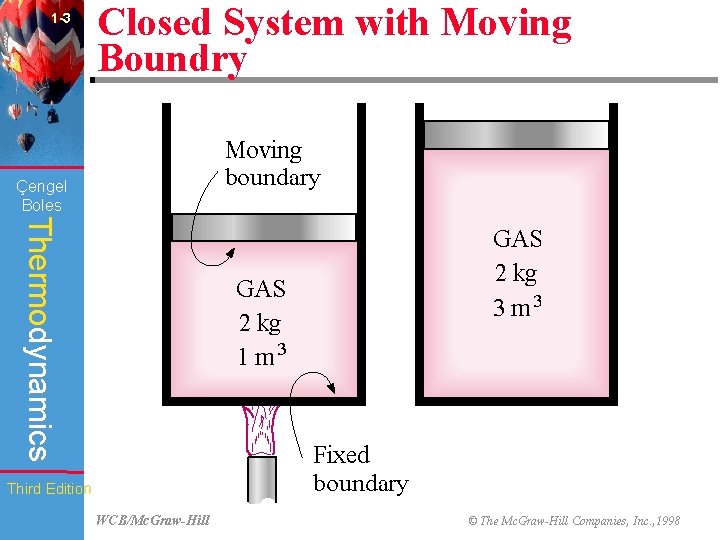
1 -3 Closed System with Moving Boundry Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -4 Crossing Control Volume Boundaries Mass and Energy Cross Control Volume Boundaries Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
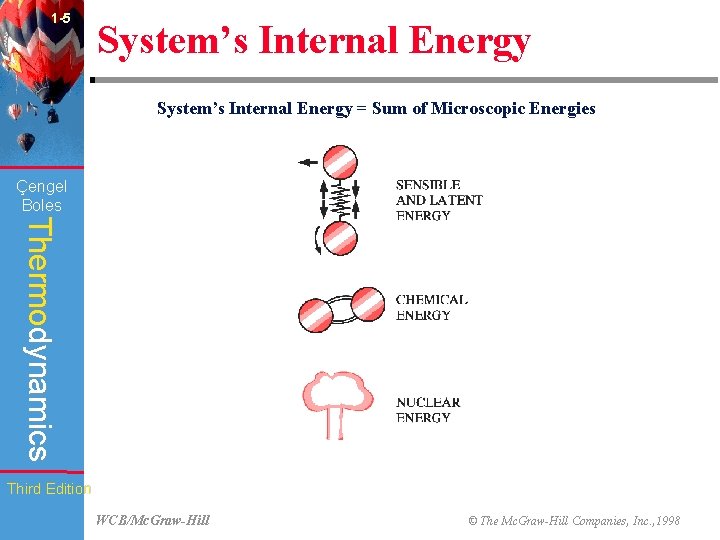
1 -5 System’s Internal Energy = Sum of Microscopic Energies (Fig. 1 -19) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -6 Quasi-Equilibrium, Work-Producing Devices Deliver the Most Work (Fig. 1 -30) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
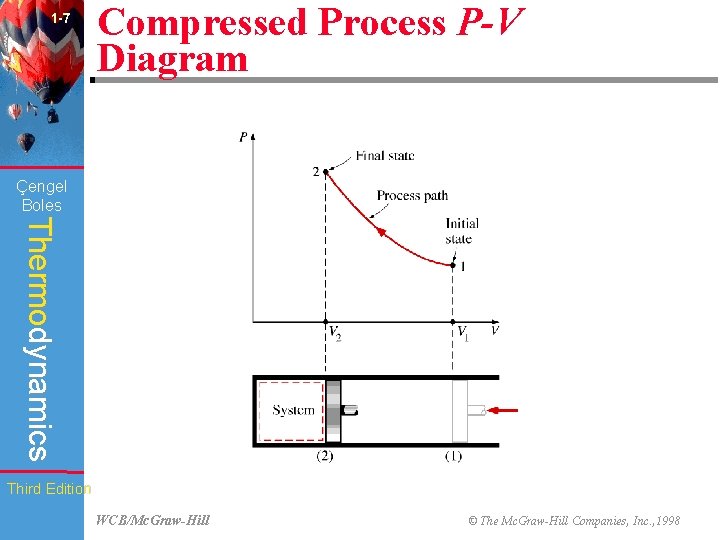
1 -7 Compressed Process P-V Diagram (Fig. 1 -31) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
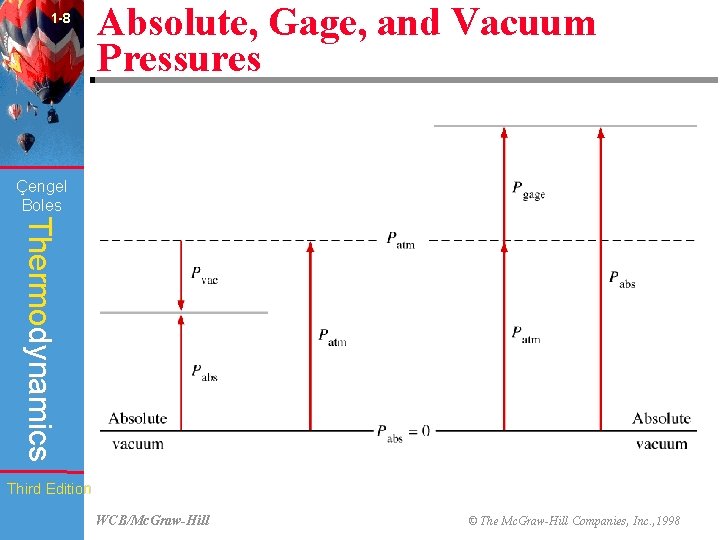
1 -8 Absolute, Gage, and Vacuum Pressures (Fig. 1 -36) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
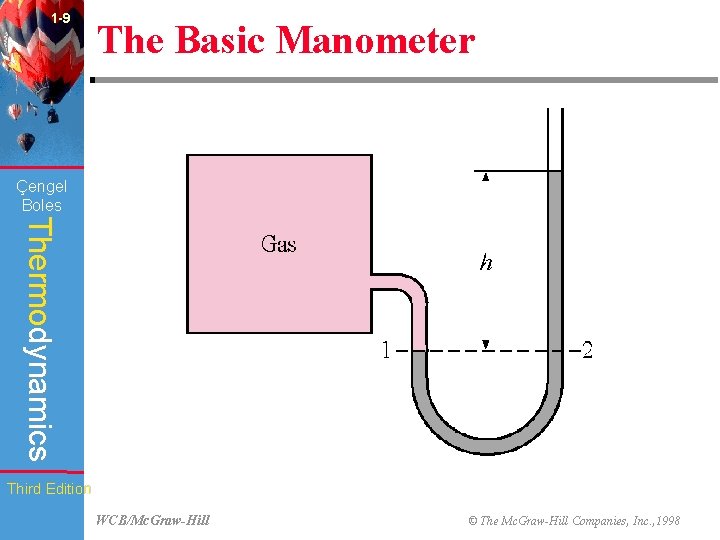
1 -9 The Basic Manometer Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
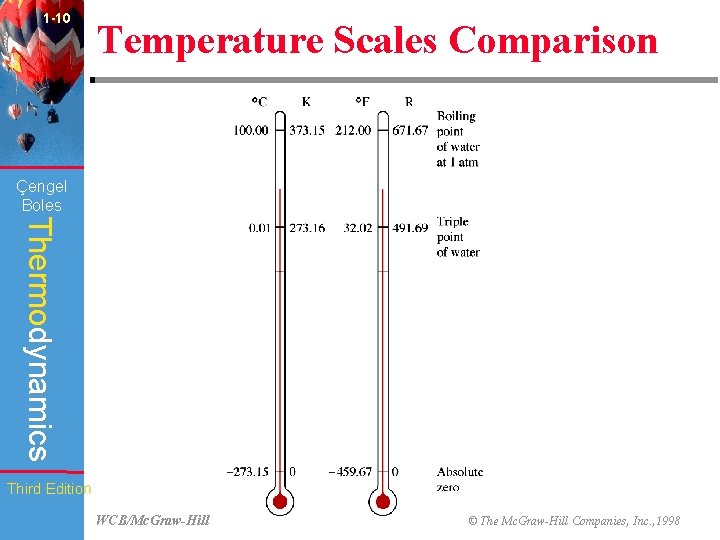
1 -10 Temperature Scales Comparison (Fig. 1 -48) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
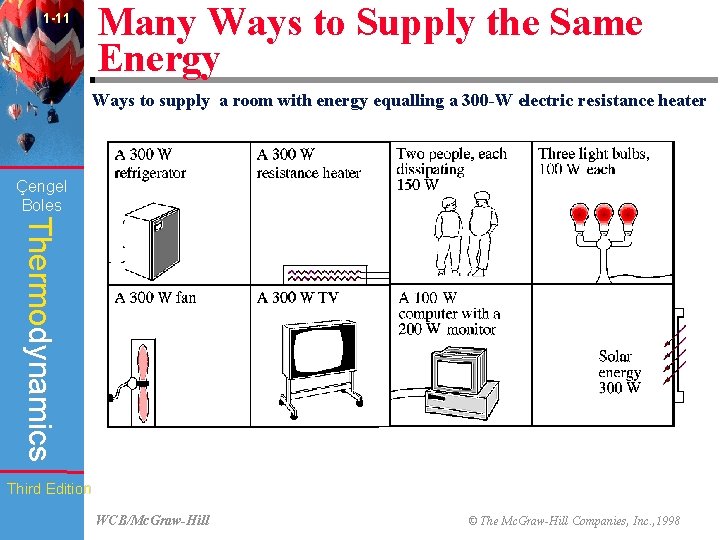
1 -11 Many Ways to Supply the Same Energy Ways to supply a room with energy equalling a 300 -W electric resistance heater (Fig. 1 -52) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
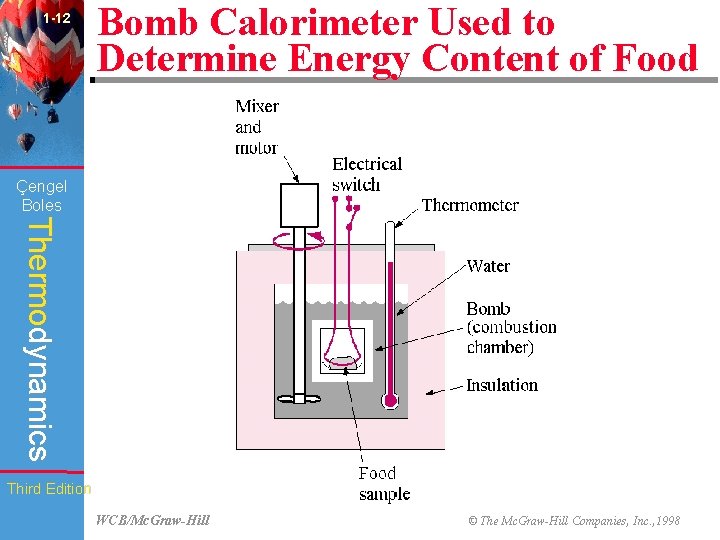
1 -12 Bomb Calorimeter Used to Determine Energy Content of Food (Fig. 1 -53) Çengel Boles Thermodynamics Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -13 Chapter Summary Çengel Boles Thermodynamics • Thermodynamics is the science that primarily deals with energy. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -14 Çengel Boles Chapter Summary Thermodynamics • The first law of thermodynamics is simply an expression of the conservation of energy principle, and it asserts that energy is a thermodynamic property. • The second law of thermodynamics asserts that energy has quality as well as quantity, and actual processes occur in the direction of decreasing quality of energy. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -15 Çengel Boles Chapter Summary Thermodynamics • A system of fixed mass is called a closed system, or control mass, and a system that involves mass transfer across its boundaries is called an open system, or control volume. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -16 Çengel Boles Chapter Summary Thermodynamics • The mass-dependent properties of a system are called extensive properties and the others, intensive properties. Density is mass per unit volume, and specific volume is volume per unit mass. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -17 Çengel Boles Chapter Summary Thermodynamics • The sum of all forms of energy of a system is called total energy, which is considered to consist of internal, kinetic, and potential energies. Internal energy represents the molecular energy of a system and may exist in sensible, latent, chemical, and nuclear forms. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -18 Çengel Boles Chapter Summary Thermodynamics • A system is said to be in thermodynamic equilibrium if it maintains thermal, mechanical, phase, and chemical equilibrium. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -19 Çengel Boles Chapter Summary Thermodynamics • Any change from one state to another is called a process. • A process with identical end states is called a cycle. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -20 Çengel Boles Chapter Summary Thermodynamics • During a quasi-static or quasi-equilibrium process, the system remains practically in equilibrium at all times. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -21 Çengel Boles Chapter Summary Thermodynamics • The state of a simple, compressible system is completely specified by two independent, intensive properties. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -22 Çengel Boles Chapter Summary Thermodynamics • Force per unit area is called pressure, and its unit is the pascal. The absolute, gage, and vacuum pressures are related by Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -23 Çengel Boles Chapter Summary Thermodynamics • Small to moderate pressure differences are measured by a manometer, and a differential fluid column of height h corresponds to a pressure difference of where is the fluid density and g is the local gravitational acceleration. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -24 Çengel Boles Chapter Summary Thermodynamics • The atmospheric pressure is measured by a barometer and is determined from where h is the height of the liquid column above the free surface. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -25 Çengel Boles Chapter Summary Thermodynamics • The zeroth law of thermodynamics states that two bodies are in thermal equilibrium if both have the same temperature reading even if they are not in contact. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -26 Çengel Boles Chapter Summary Thermodynamics • The temperature scales used in the SI and the English system today are the Celsius scale and the Fahrenheit scale, respectively. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -27 Çengel Boles Chapter Summary Thermodynamics • The absolute temperature scale in the SI is the Kelvin scale, which is related to the Celsius scale by Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -28 Çengel Boles Chapter Summary Thermodynamics • In the English system, the absolute temperature scale is the Rankine scale, which is related to the Fahrenheit scale by Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -29 Çengel Boles Chapter Summary Thermodynamics • The magnitudes of each division of 1 K and 1 0 C are identical, and so are the magnitude of each division of 1 R and 10 F. Therefore, and Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998

1 -30 Çengel Boles Chapter Summary Thermodynamics • An important application area of thermodynamics is the biological system. Most diets are based on the simple energy balance: the net energy gained by a person in the form of fat is equal to the difference between the energy intake from food and the energy expended by exercise. Third Edition WCB/Mc. Graw-Hill © The Mc. Graw-Hill Companies, Inc. , 1998
- Slides: 32