Energy What is energy l The ability to

Energy

What is energy? l The ability to do work or cause change is called energy.

Energy Occurs in Many Forms: l l Potential Energy Kinetic Energy l l l Mechanical Energy Thermal Energy Chemical Energy Electromagnetic Energy Nuclear Energy

Kinetic Energy l l The energy of motion is called kinetic energy. The kinetic energy of an object depends on both its mass and its velocity. Kinetic energy increases as mass increases. Kinetic energy increases as velocity increases. If these two vehicle are going the same velocity, which one has the greater kinetic energy?

Potential Energy that is stored or held in readiness is called potential energy. Examples: food, a roller coaster at the top of the first hill

Elastic Potential Energy l l l The potential energy associated with objects that can be stretched or compressed is called elastic potential energy. Ex. a stretched rubber band A compressed spring

Gravitational Potential Energy l l Potential energy that depends on an object’s height above the ground is gravitational potential energy. Example: a boulder on the edge of a cliff What does this boulder have the potential to do?

Potential Energy Examples

Kinetic and Potential Energy

Mechanical Energy l l Mechanical energy is the energy associated with the motion or movement of an object. Examples: a frog leaping, a car moving

Thermal Energy l Thermal (or heat) energy is a measure of the energy of the movement of particles (molecules) in an object. Example: ice cream melting l

Chemical Energy l l Chemical energy is energy in the chemical bonds that hold compounds together. When those bonds are broken through chemical reactions, the energy is released. Ex. the food you eat, gasoline

Electrical Energy l l Electrical energy is energy associated with moving electric charges. Example: energy in power lines

Electromagnetic Energy Electromagnetic energy is a form or radiant energy that travels in waves. l Examples: visible light microwaves infrared radiation ultraviolet radiation l


Nuclear Energy Nuclear energy is stored in the nucleus of an atom and is released during nuclear reactions. Examples: nuclear power in a power plant, the sun and other stars

Transformations or Conversions Between Forms of Energy l l A change from one form of energy to another is called an energy conversion. Most forms of energy can be converted into any other form. Example: a toaster converts electrical energy into thermal energy

Kinetic and Potential Energy l One of the most common conversions is from kinetic to potential energy and back. l Ex. Energy Conversion in an Earthquake Ex. Energy Conversion in a Waterfall Ex. Energy Conversion in a Pole Vault Ex. Energy Conversion in a Pendulum l l l


Conservation of Energy l l According to the law of conservation of energy, energy cannot be created or destroyed. So what happens to the energy in a pendulum? Why does it slow down? The mechanical energy of the moving pendulum is transformed into heat energy due to friction. So, it is not lost, but converted.

A little change…. l Einstein changed the law of conservation of energy with his theory of relativity. He said that energy can sometimes be created, but in order to do so, matter must be destroyed. l. Therefore, scientists say that energy and matter together are always conserved.

More Energy Conversions: Photosynthesis l l The process through which plants make food is known as photosynthesis. Photo = “light” Synthesis = “putting together” During photosynthesis, white light (electromagnetic energy) is converted into food (chemical energy).

The Nature of Light l l l The sun is the source of light energy on Earth. There are three things that can happen to light. It can be: Transmitted (allowed to pass through) Reflected (bounced off) Absorbed (taken in)

Plants and Light l Plants absorb some colors of the visible spectrum and reflect others. They have pigments that absorb different colors of light. The colors that are not absorbed (green) are reflected. This is why plants usually appear green. l Chlorophyll is the pigment that gives plants their green color. It absorbs most blue and red light, and reflects the green part of the spectrum.
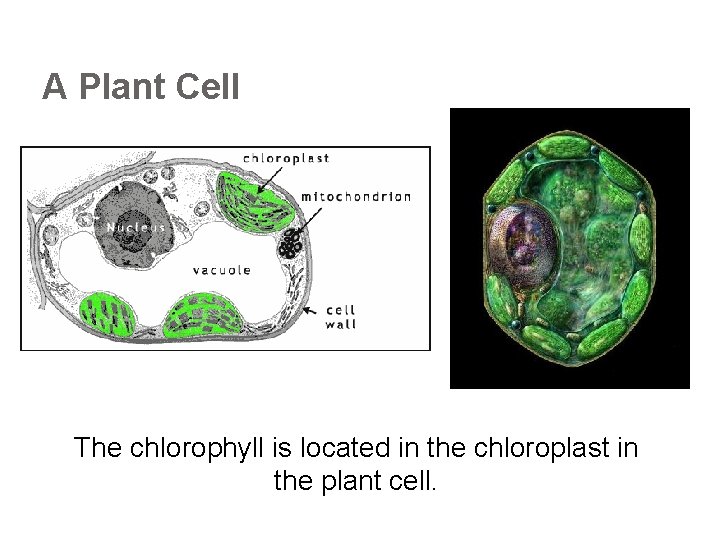
A Plant Cell The chlorophyll is located in the chloroplast in the plant cell.

Energy Conversions in Photosynthesis Capturing the Sun’s Energy Photosynthesis begins when chlorophyll in the chloroplast of the plant cells and other pigments capture the sun’s radiant energy.

Using Energy to Make Food l Water and Carbon dioxide are combined with the radiant energy to create a sugar that the plant uses for food. l The chemical formula for this is……
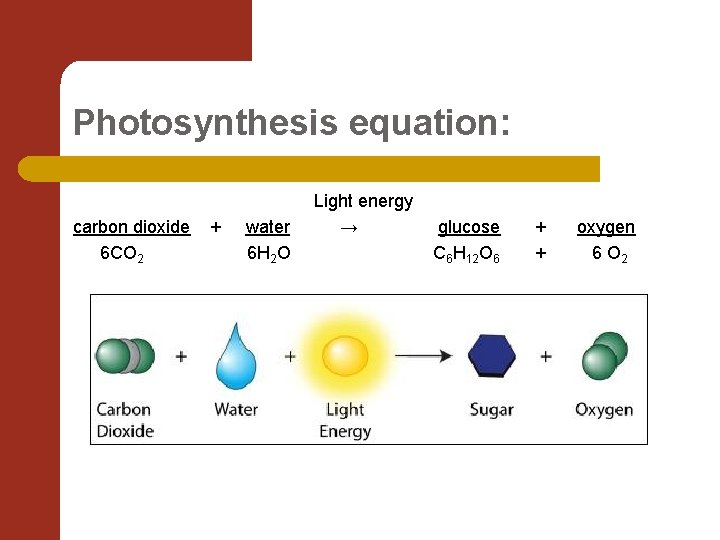
Photosynthesis equation: carbon dioxide 6 CO 2 + water 6 H 2 O Light energy → glucose C 6 H 12 O 6 + + oxygen 6 O 2

Fossil Fuels l l l A fuel is a material that releases energy when it burns. A fossil fuel is an energy-rich substance formed from the remains of organisms. This energy was stored as potential energy when the plants of that time converted the sun’s radiant energy into chemical Fossil ferns within a sheet of coal. energy.

Using Fossil Fuels l The process of burning fuels is known as combustion. Today in coal burning power plants, coal is burned to heat water, which creates steam that runs turbines. These turbines are connected to generators which produce electricity. Left: An oil rig - pumps oil from deep underground. Right: A coal mine - large quarry diggers carve out the resource.
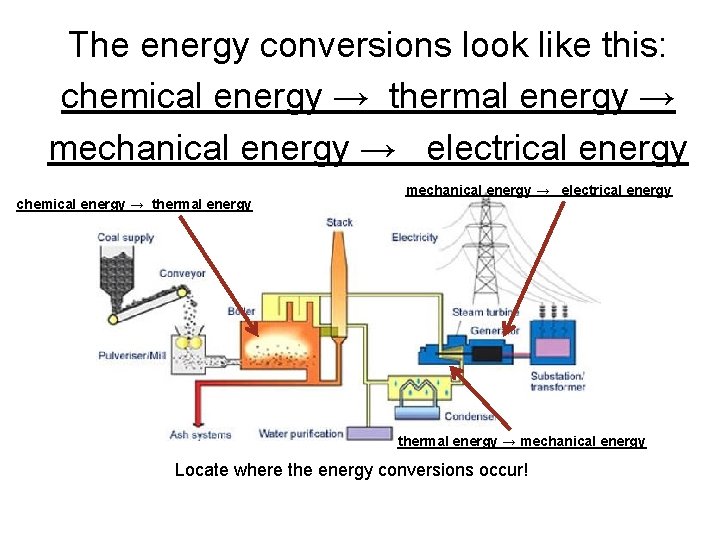
The energy conversions look like this: chemical energy → thermal energy → mechanical energy → electrical energy chemical energy → thermal energy mechanical energy → electrical energy thermal energy → mechanical energy Locate where the energy conversions occur!

Fossil Fuels
- Slides: 33